Two approaches Sediment accumulation rates Two basic approaches

Two approaches Sediment accumulation rates Two basic approaches: -a sediment component that changes at a known rate after deposition (constant, or known, input) Radioactive decay – U series, radiocarbon - a sediment component with a known, time-dependent input function (constant, or known change, after deposition) excess 230 Th , 18 O chronostratigraphy

Radiometric dating Dating principles – covered in Isotope Geochemistry (Faure) Two “simple” approaches: Average slopes from age vs. depth plots Absolute 14 C dates foraminiferal abundance maxima Normalization to constant 230 Th flux Sediment focusing / winnowing Point-by-point mass accumulation rates

Terminology Radioactive parent daughter + - (electron) or (He nucleus) (or + or emission or e – capture or…) Isotopes: Same number of protons, differing numbers of neutrons chemically similar, different mass (kinetics), different radioactive properties

Decay rate Radioactive decay rate proportional to number of atoms present N is the number of parent atoms in the sample λ is the decay constant (units t-1) λN gives the activity (disintegrations/time)

Half life (Radioactive decay equation)

Half life The time for 50% of atoms present to decay. Rule of thumb = radioisotope activity can be measured for about 5 half-lives
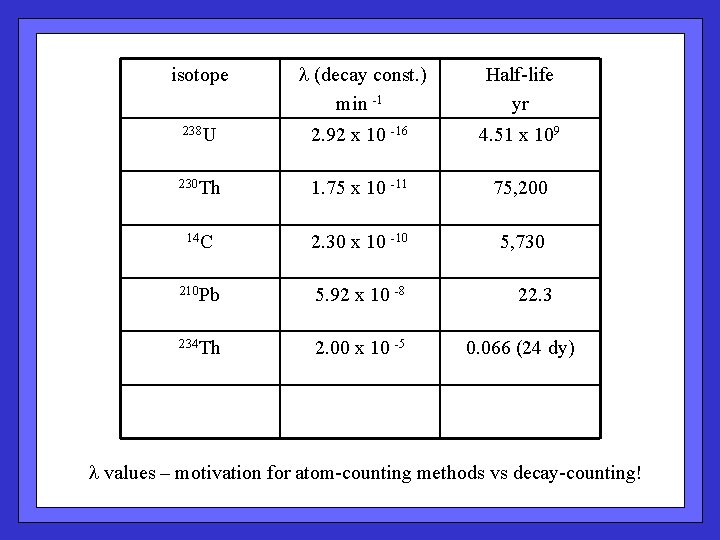
isotope l (decay const. ) min -1 Half-life yr 238 U 2. 92 x 10 -16 4. 51 x 109 230 Th 1. 75 x 10 -11 75, 200 14 C 2. 30 x 10 -10 5, 730 210 Pb 5. 92 x 10 -8 22. 3 234 Th 2. 00 x 10 -5 0. 066 (24 dy) l values – motivation for atom-counting methods vs decay-counting!
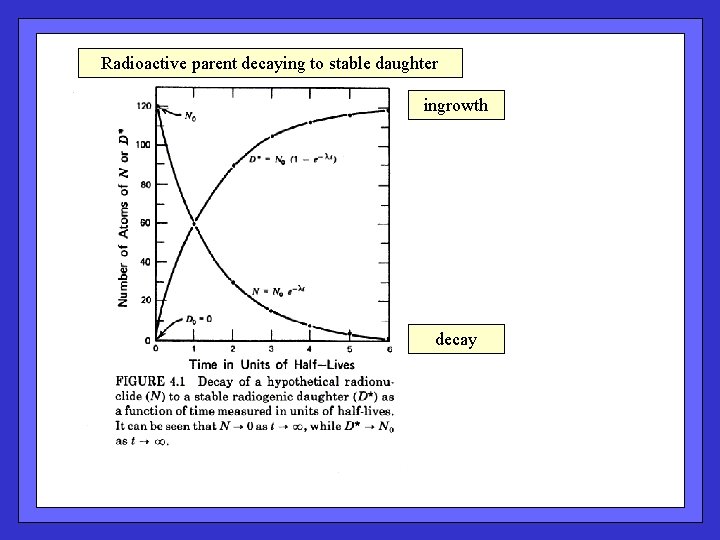
Radioactive parent decaying to stable daughter ingrowth decay
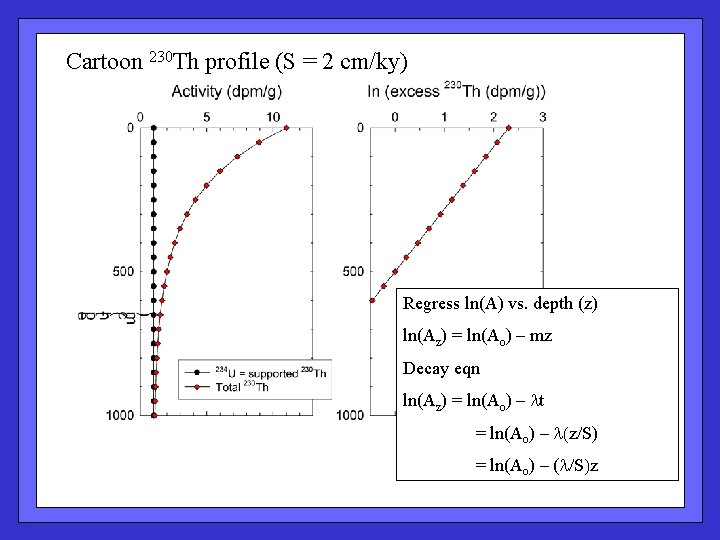
Cartoon 230 Th profile (S = 2 cm/ky) TH 230 cartoon Regress ln(A) vs. depth (z) ln(Az) = ln(Ao) – mz Decay eqn ln(Az) = ln(Ao) – lt = ln(Ao) – l(z/S) = ln(Ao) – (l/S)z

Assumptions for regressions of age vs. depth Accumulation without mixing below the mixed layer The isotope is immobile in the sediment Constant input activity (reservoir age), or known as a function of time Recall activity at time = 0 in the decay equation: How well do we know N(o) in the past?
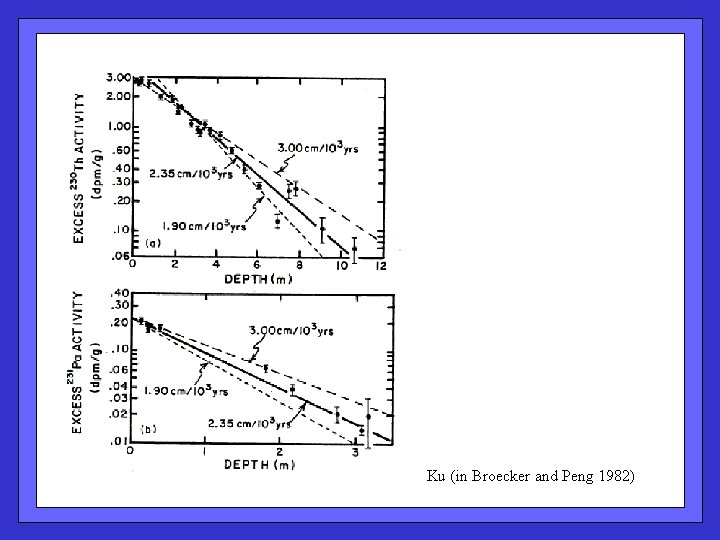
Ku (in Broecker and Peng 1982)
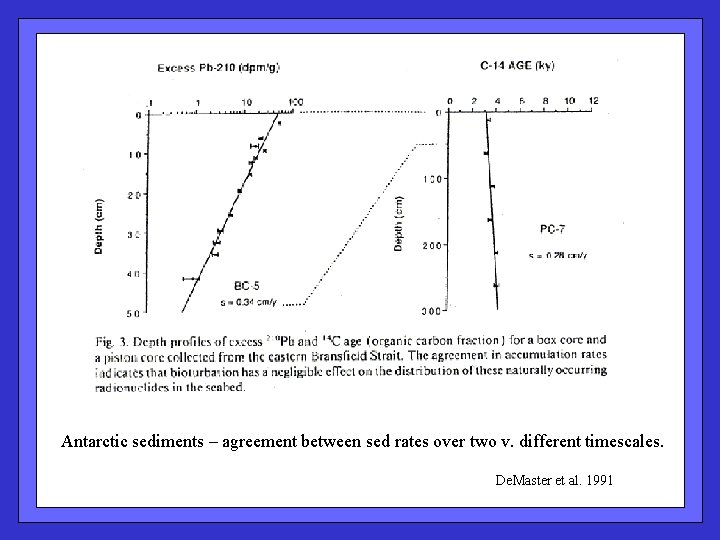
Antarctic sediments – agreement between sed rates over two v. different timescales. De. Master et al. 1991

Radiocarbon: Produced where? How? Natural variability in production? Natural variability in atmospheric 14 C content? Human impacts on 14 C budgets?
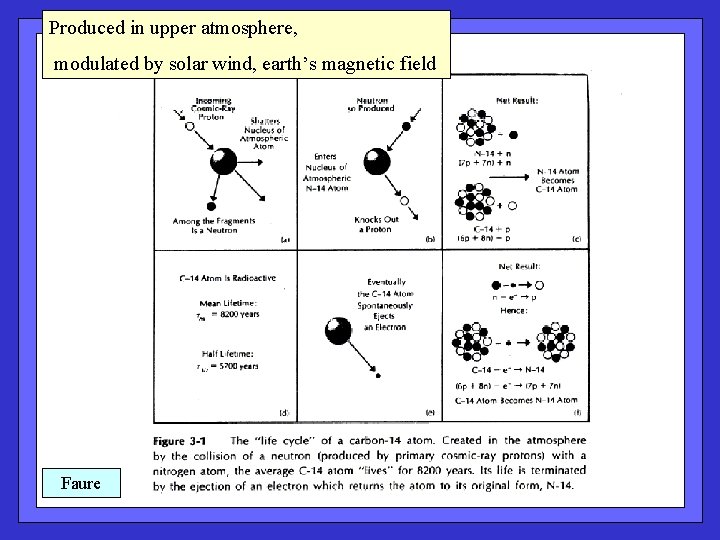
Produced in upper atmosphere, modulated by solar wind, earth’s magnetic field Faure

Natural variability in atmospheric 14 C content? YES! Production variations (solar, geomagnetic) Carbon cycle (partitioning between atmosphere, biosphere, and ocean) At steady state, global decay = global production But global C cycle not necessarily at steady state, and 14 C offsets between C reservoirs not constant.

Human impacts on 14 C budgets? Seuss effect (fossil fuel dilution of 14 C(atm)) Bomb radiocarbon inputs

14 C-free 14 C produced in atmosphere, but most CO 2 resides in (and decays in) the ocean

Radiocarbon dating of sediments. Bulk Ca. CO 3, or bulk organic C standard AMS sample 25 mol C (2. 5 mg Ca. CO 3) Specific phases of known provenance: Planktic, benthic foraminifera Specific (biomarker) compounds (5 mol C) Dating known phases (e. g. , foraminifera), at their abundance maxima, improves the reliability of each date. No admixture of fossil (14 C-free) material. Minimizes age errors caused by particle mixing and faunal abundance variations. But, reduces # of datable intervals.

Radiocarbon reporting conventions are convoluted! 14 C data reported as Fraction modern, or age, or Δ 14 C t 1/2 ~ 5730 y (half life) λ= 0. 00012097 / yr (decay constant) (about 1% in 83 years) Account for fractionation, normalize to 13 C = -25 Activity relative to wood grown in pre-bomb atmosphere Stable carbon isotopic composition
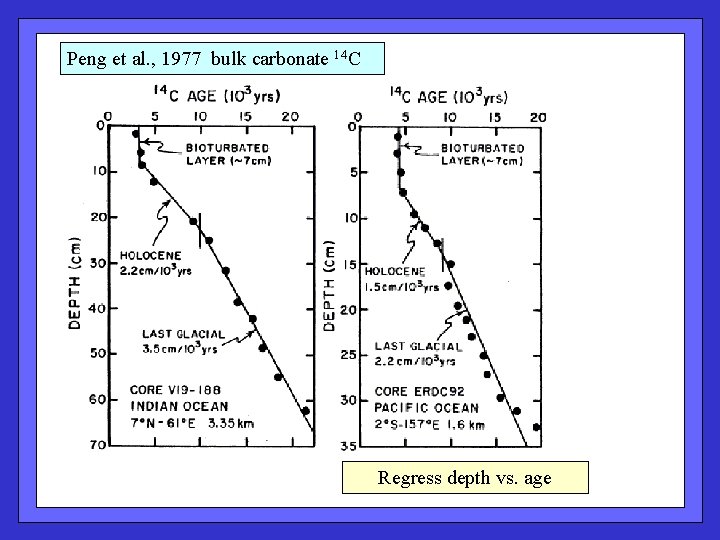
Peng et al. , 1977 bulk carbonate 14 C Regress depth vs. age
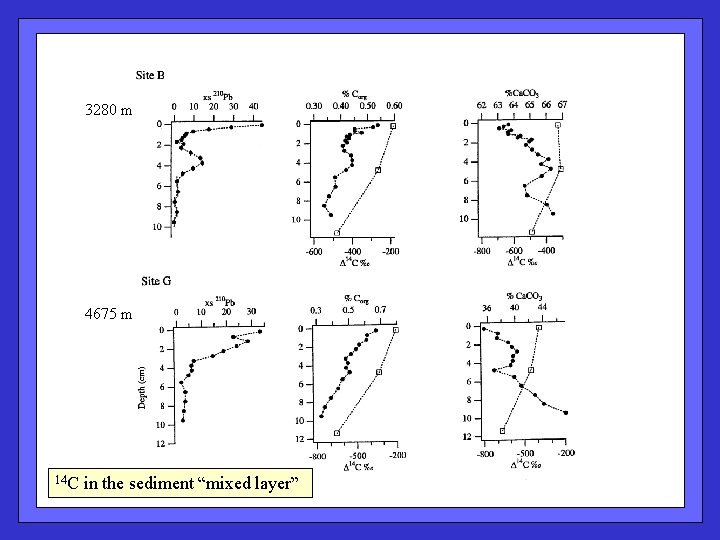
3280 m 4675 m 14 C in the sediment “mixed layer”
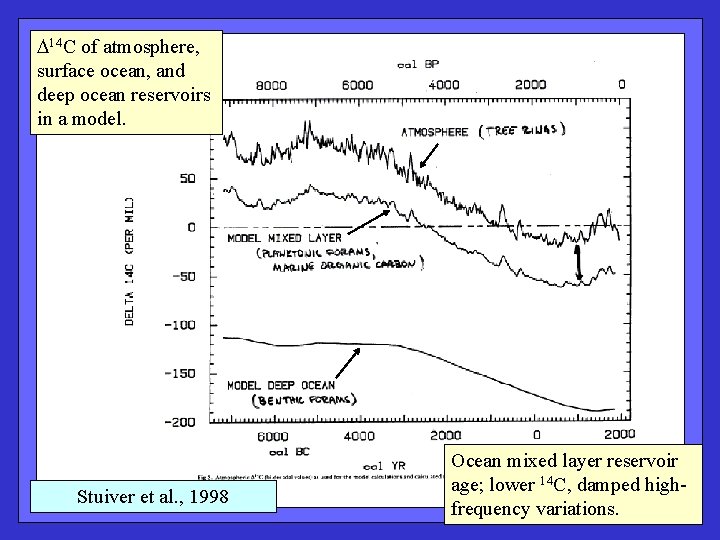
14 C of atmosphere, surface ocean, and deep ocean reservoirs in a model. Stuiver et al. , 1998 Ocean mixed layer reservoir age; lower 14 C, damped highfrequency variations.
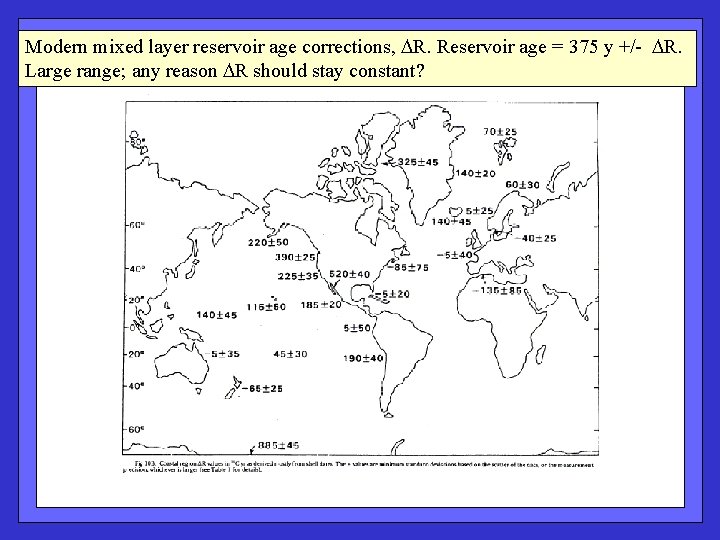
Modern mixed layer reservoir age corrections, R. Reservoir age = 375 y +/- R. Large range; any reason R should stay constant?
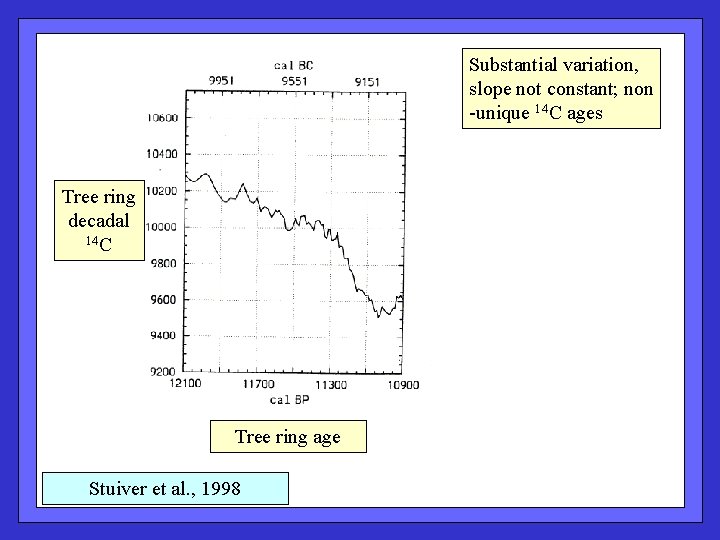
Substantial variation, slope not constant; non -unique 14 C ages Tree ring decadal 14 C Tree ring age Stuiver et al. , 1998
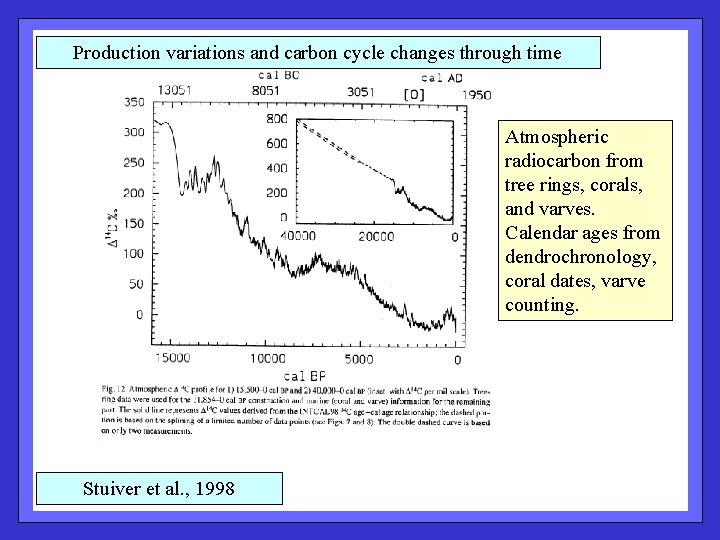
Production variations and carbon cycle changes through time Atmospheric radiocarbon from tree rings, corals, and varves. Calendar ages from dendrochronology, coral dates, varve counting. Stuiver et al. , 1998
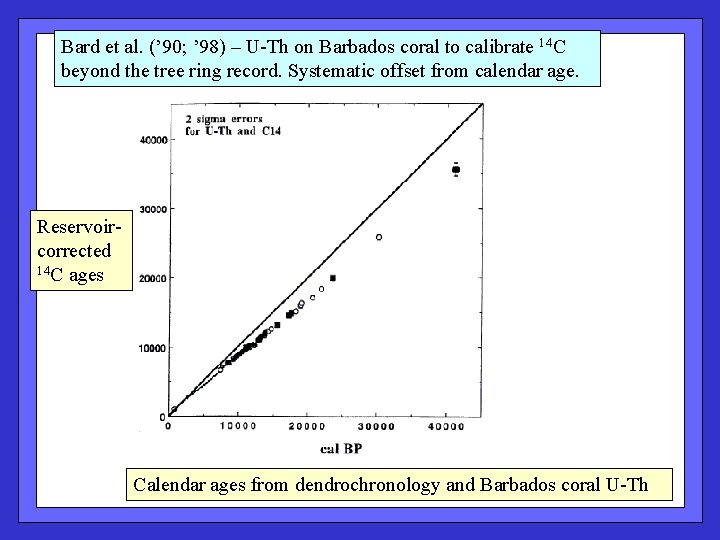
Bard et al. (’ 90; ’ 98) – U-Th on Barbados coral to calibrate 14 C beyond the tree ring record. Systematic offset from calendar age. Reservoircorrected 14 C ages Calendar ages from dendrochronology and Barbados coral U-Th

The product of these radiocarbon approaches is an age-depth plot. Regression gives a sedimentation rate; linearity gives an estimate of sed rate variability. Typically, sedimentation rates do vary. How many line segments do you fit to your data? How confident are you in each resulting rate estimate? To estimate mass accumulation rates (MARs) Calculate average sedimentation rates between dated intervals, and multiply by dry bulk density and concentration. But: Average sed rates can’t be multiplied by point-by-point dry bulk density and concentration to yield time series. The solution – 230 Th-normalized accumulation rates

Two approaches Sediment accumulation rates Two basic approaches: -a sediment component that changes at a known rate after deposition (constant, or known, input) Radioactive decay – U series, radiocarbon - a sediment component with a known, time-dependent input function (constant, or known change, after deposition) excess 230 Th , 18 O chronostratigraphy

Flux estimates using excess 230 Th in sediments (M. Bacon; R. Francois) Assume: 230 Th sinking flux = production from 234 U parent in the water column = constant fn. of water depth (uranium is essentially conservative in seawater) Correct sediment 230 Th for detrital 230 Th using measured 232 Th and detrital 232 Th/238 U. Correct sediment 230 Th for ingrowth from authigenic U (need approximate age model). Use an age model to correct the remaining, “excess” 230 Th for decay since the time of deposition.

Two applications: 1. Integrate the xs 230 Th between known time points (14 C, 18 O). Deviations from the predicted (decay-corrected) xs 230 Th inventory reflect sediment focusing or winnowing. 2. Sample by sample, normalize concentrations of sediment constituents (Ca. CO 3, organic C, etc. ) to the xs 230 Th of that sample. Yields flux estimates that are not influenced by dissolution, dilution.

Point by point normalization: Activity(230) (dpm g-1) = Flux(230) (dpm m-2 y-1) Bulk flux (g m-2 y-1) So: Bulk flux (g m-2 y-1) = Flux(230) (dpm m-2 y-1) Activity(230) (dpm g-1) = Prod(230) (dpm m-3 y-1) x (water depth) Activity(230) (dpm g-1) And: Component i flux (g m-2 y-1) = Bulk flux (g m-2 y-1) x (wt % i)

Simple examples (without focusing changes): If % C org increases in a sample, but Activity(xs 230 Th) increases by the same fraction, then no increase in C org burial – just a decrease in some other sediment component. If % C org stays constant relative to samples above and below, but Activity(xs 230 Th) decreases, then the C org flux (and the bulk flux) both increased in that sample (despite lack of a concentration signal). But: To assess changes in focusing, we’re stuck integrating between (dated) time points.

Chronostratigraphy based on foraminiferal 18 O values Two premises (observed): The 18 O of seawater responds to changes in global ice volume - high-latitude precipitation is strongly depleted in 18 O - more ice on continents => higher seawater 18 O Foraminiferal 18 O reflects seawater 18 O - (but also temperature) Foraminiferal 18 O provides a global stratigraphy Dating (radiometric, or orbital tuning) provides timescale
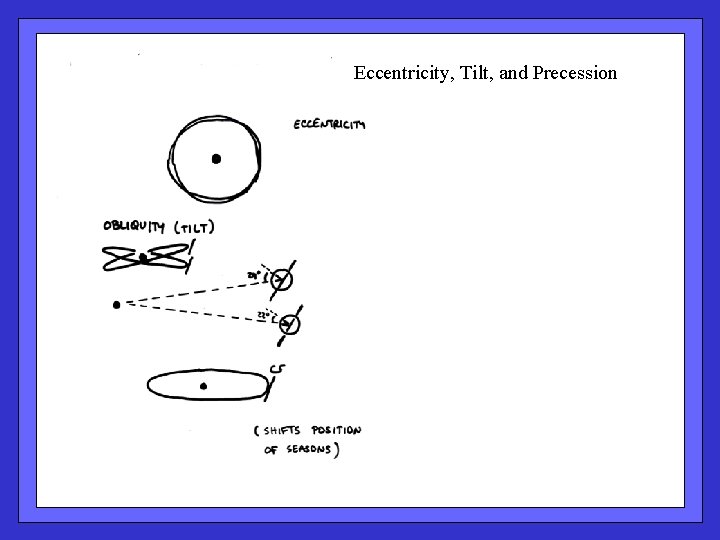
Eccentricity, Tilt, and Precession
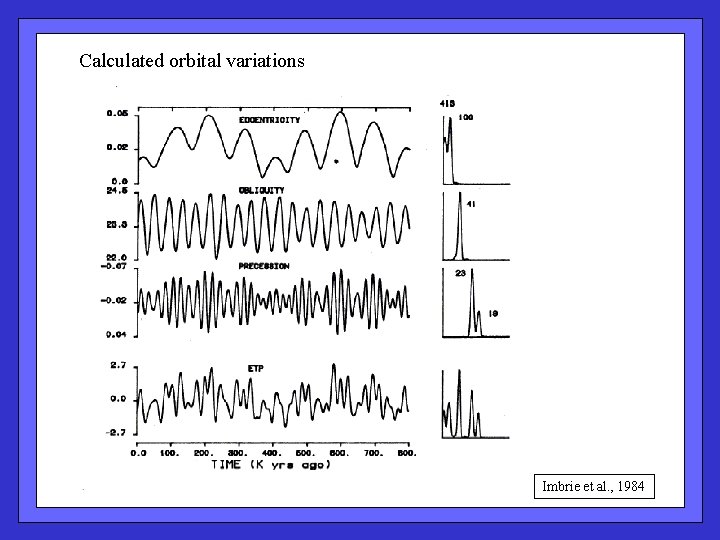
Calculated orbital variations Imbrie et al. , 1984
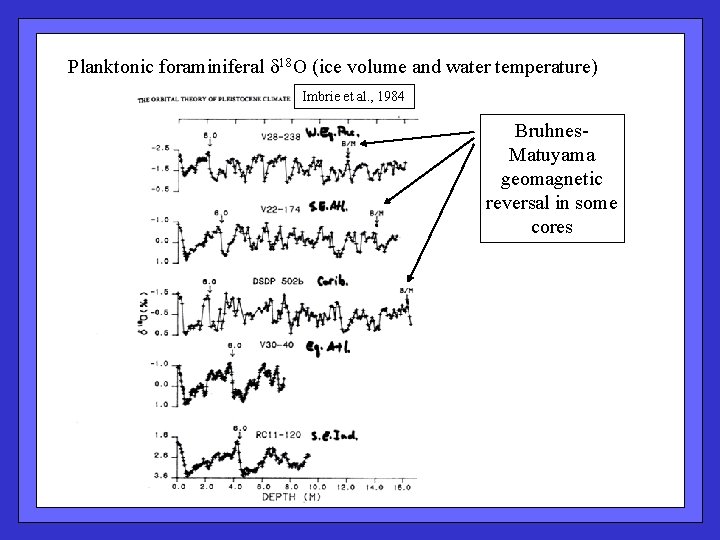
Planktonic foraminiferal 18 O (ice volume and water temperature) Imbrie et al. , 1984 Bruhnes. Matuyama geomagnetic reversal in some cores

18 O vs. time Align control points (“wiggle matching”) to put cores on same time-scale. Assumption – the d 18 O time series reflect global signal (ice volume, and SST)
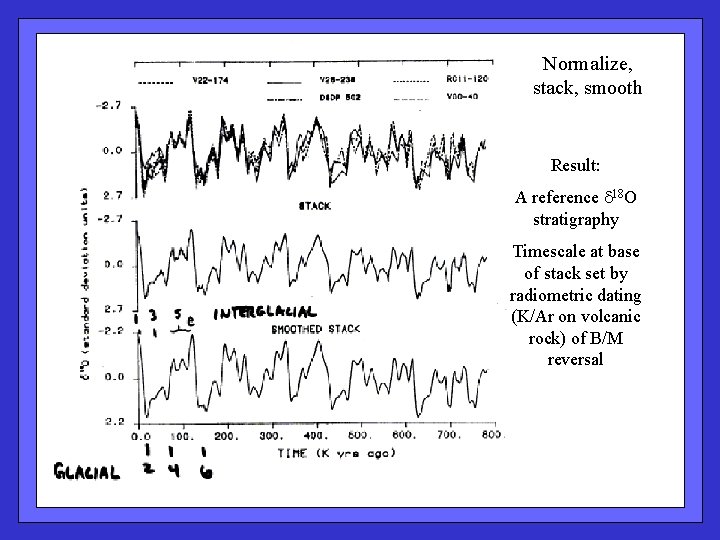
Normalize, stack, smooth Result: A reference 18 O stratigraphy Timescale at base of stack set by radiometric dating (K/Ar on volcanic rock) of B/M reversal
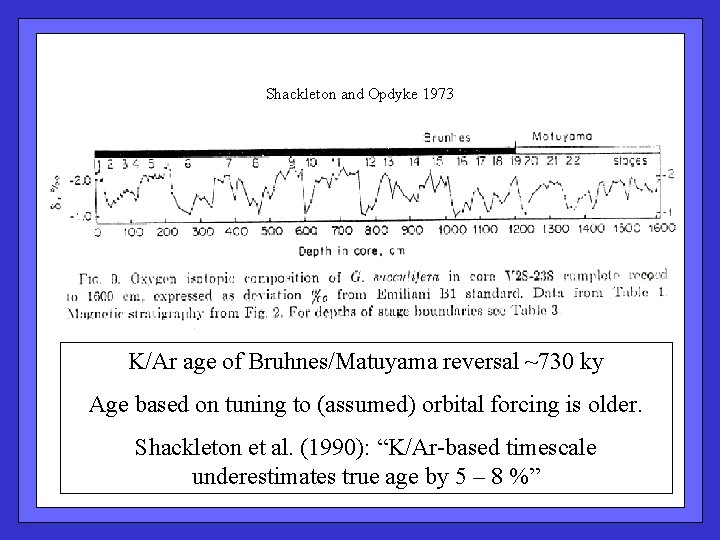
Shackleton and Opdyke 1973 K/Ar age of Bruhnes/Matuyama reversal ~730 ky Age based on tuning to (assumed) orbital forcing is older. Shackleton et al. (1990): “K/Ar-based timescale underestimates true age by 5 – 8 %”
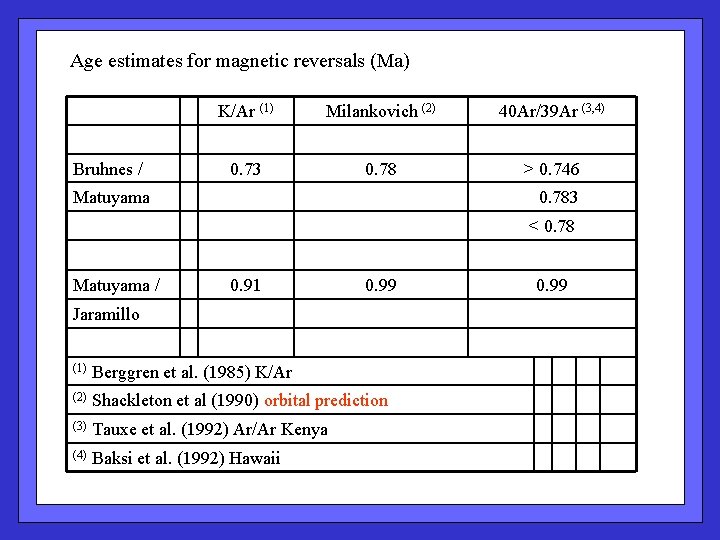
Age estimates for magnetic reversals (Ma) Bruhnes / K/Ar (1) Milankovich (2) 40 Ar/39 Ar (3, 4) 0. 73 0. 78 > 0. 746 Matuyama 0. 783 < 0. 78 Matuyama / 0. 91 0. 99 Jaramillo (1) Berggren et al. (1985) K/Ar (2) Shackleton et al (1990) orbital prediction (3) Tauxe et al. (1992) Ar/Ar Kenya (4) Baksi et al. (1992) Hawaii 0. 99
- Slides: 40