TURQUOISEIII Study ombitasvirparitaprevirritonavir dasabuvir for genotype 1 b
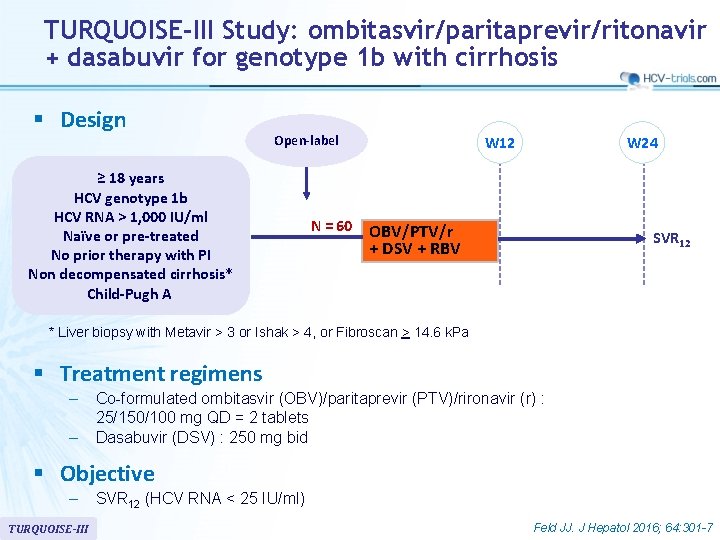
TURQUOISE-III Study: ombitasvir/paritaprevir/ritonavir + dasabuvir for genotype 1 b with cirrhosis § Design Open-label ≥ 18 years HCV genotype 1 b HCV RNA > 1, 000 IU/ml Naïve or pre-treated No prior therapy with PI Non decompensated cirrhosis* Child-Pugh A N = 60 W 12 W 24 OBV/PTV/r + DSV + RBV SVR 12 * Liver biopsy with Metavir > 3 or Ishak > 4, or Fibroscan > 14. 6 k. Pa § Treatment regimens – – Co-formulated ombitasvir (OBV)/paritaprevir (PTV)/rironavir (r) : 25/150/100 mg QD = 2 tablets Dasabuvir (DSV) : 250 mg bid § Objective – TURQUOISE-III SVR 12 (HCV RNA < 25 IU/ml) Feld JJ. J Hepatol 2016; 64: 301 -7

TURQUOISE-III Study: ombitasvir/paritaprevir/ritonavir + dasabuvir for genotype 1 b with cirrhosis Baseline characteristics and outcome N = 60 Mean age, years Female 38% Race : black 12% Body mass index, mean 27. 8 IL 28 B non-CC genotype 83% HCV RNA log 10 IU/ml, mean (SD) 6. 6 ± 0. 6 Fibroscan, k. PA, median (range) 19 (12. 5 -67. 8) Prior treatment with PEG-IFN + RBV, N (%) SVR 12 TURQUOISE-III 60 33 (55%) 100% Feld JJ. J Hepatol 2016; 64: 301 -7
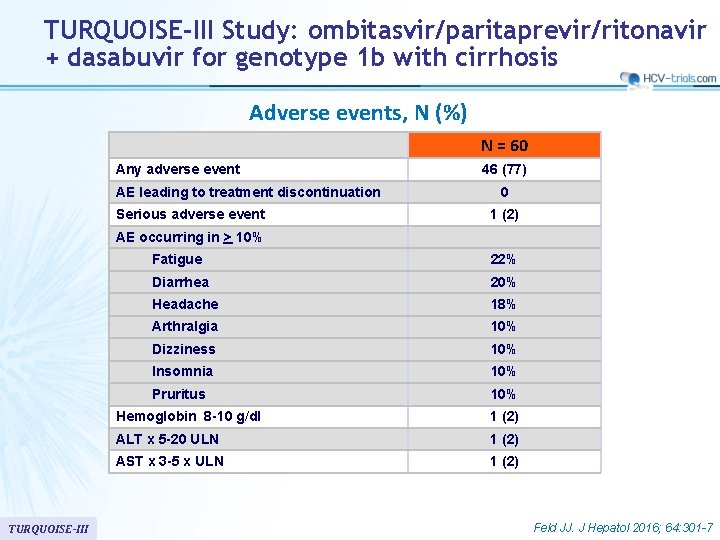
TURQUOISE-III Study: ombitasvir/paritaprevir/ritonavir + dasabuvir for genotype 1 b with cirrhosis Adverse events, N (%) N = 60 Any adverse event AE leading to treatment discontinuation Serious adverse event 46 (77) 0 1 (2) AE occurring in > 10% TURQUOISE-III Fatigue 22% Diarrhea 20% Headache 18% Arthralgia 10% Dizziness 10% Insomnia 10% Pruritus 10% Hemoglobin 8 -10 g/dl 1 (2) ALT x 5 -20 ULN 1 (2) AST x 3 -5 x ULN 1 (2) Feld JJ. J Hepatol 2016; 64: 301 -7

TURQUOISE-III Study: ombitasvir/paritaprevir/ritonavir + dasabuvir for genotype 1 b with cirrhosis § Summary – OBV/PTV/r FDC + DSV, without the use of RBV, given for 12 weeks achieved SVR 12 of 100% in genotype 1 b infected patients with compensated cirrhosis, including treatment-experienced patients – Treatment was very well tolerated, with a low rate of serious adverse events, no premature discontinuations, and infrequent laboratory abnormalities that were not clinically relevant TURQUOISE-III Feld JJ. J Hepatol 2016; 64: 301 -7
- Slides: 4