TUMOR GROWTH AND METASTASIS UNDERSTANDING THE MOLECULAR BASIS

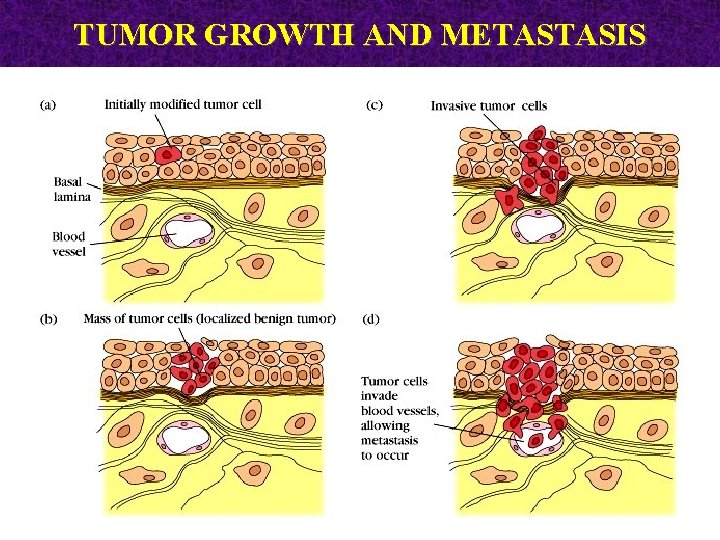
TUMOR GROWTH AND METASTASIS
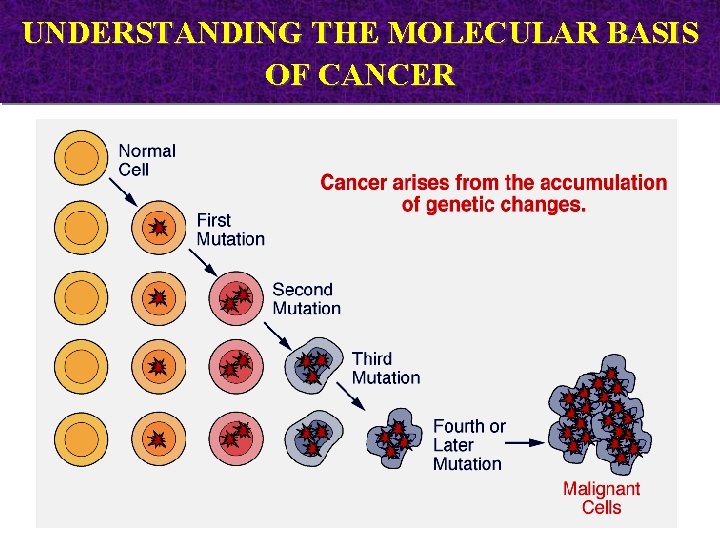
UNDERSTANDING THE MOLECULAR BASIS OF CANCER
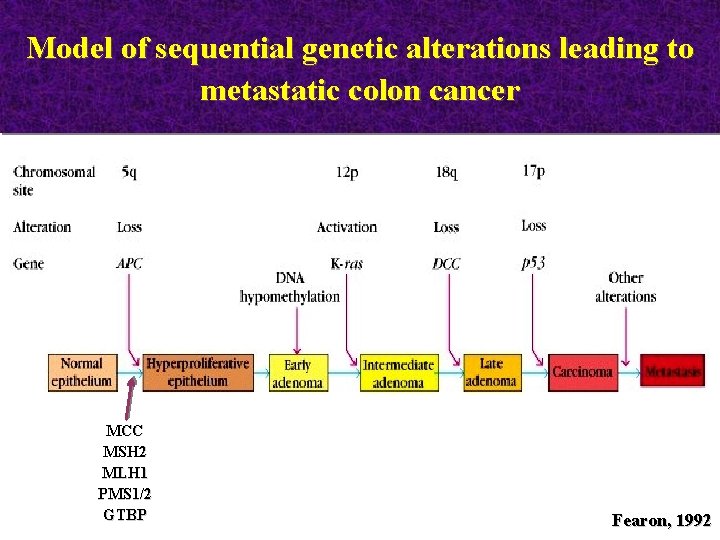
Model of sequential genetic alterations leading to metastatic colon cancer MCC MSH 2 MLH 1 PMS 1/2 GTBP Fearon, 1992

PRINCIPLE LANDMARKS IN CANCER IMMUNOLOGY 1908: positive mechanism eliminating transformed cells 1965: # immunological memory for cancer cells (mice) # immune surveillance-recognition and destruction of non-self tumor cells on their appearance 1970: lymphocytes can kill autologous melanoma cells 1991: cloning of the first tumor antigen (Belgium) 2003: therapeutic & prophylactic vaccines in the market
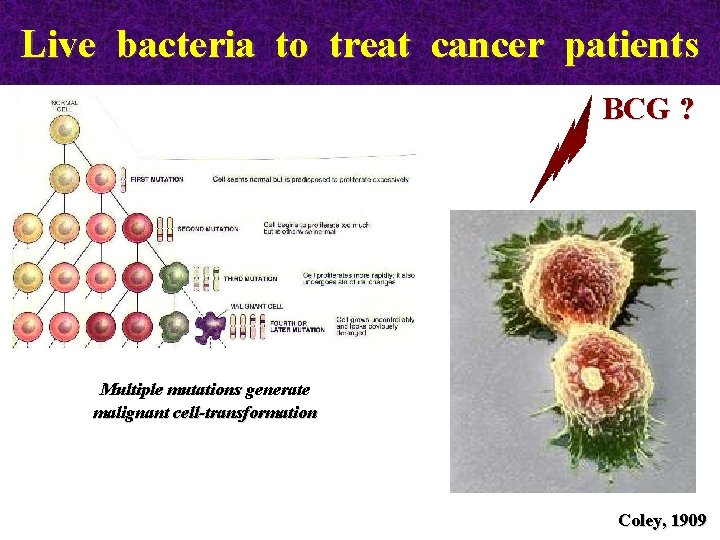
Live bacteria to treat cancer patients BCG ? Multiple mutations generate malignant cell-transformation Coley, 1909

PRINCIPLE LANDMARKS IN CANCER IMMUNOLOGY 1908: positive mechanism eliminating transformed cells 1965: # immunological memory for cancer cells (mice) # immune surveillance-recognition and destruction of non-self tumor cells on their appearance 1970: lymphocytes can kill autologous melanoma cells 1991: cloning of the first tumor antigen (Belgium) 2003: therapeutic & prophylactic vaccines in the market
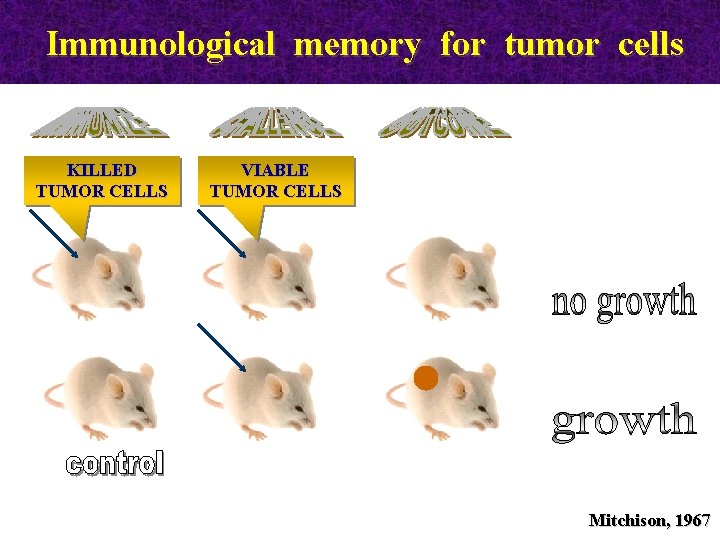
Immunological memory for tumor cells KILLED TUMOR CELLS VIABLE TUMOR CELLS Mitchison, 1967

PRINCIPLE LANDMARKS IN CANCER IMMUNOLOGY 1908: positive mechanism eliminating transformed cells 1965: # immunological memory for cancer cells (mice) # immune surveillance-recognition and destruction of non-self tumor cells on their appearance 1970: lymphocytes can kill autologous melanoma cells 1991: cloning of the first tumor antigen (Belgium) 2003: therapeutic & prophylactic vaccines in the market
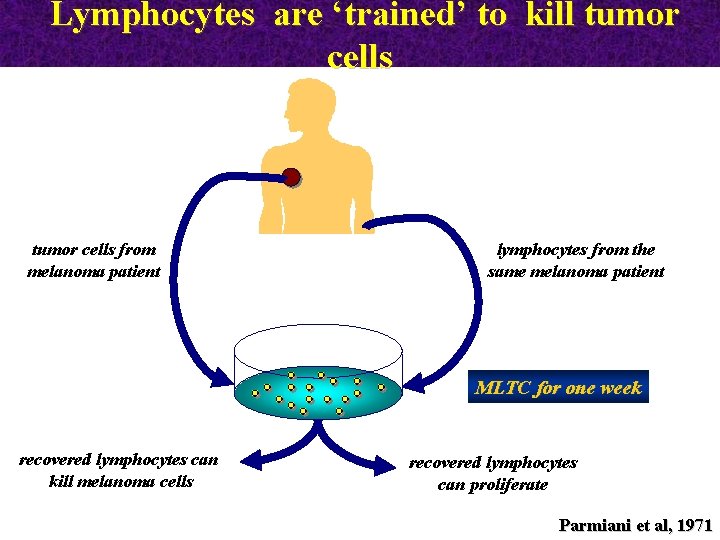
Lymphocytes are ‘trained’ to kill tumor cells from melanoma patient lymphocytes from the same melanoma patient MLTC for one week recovered lymphocytes can kill melanoma cells recovered lymphocytes can proliferate Parmiani et al, 1971

PRINCIPLE LANDMARKS IN CANCER IMMUNOLOGY 1908: positive mechanism eliminating transformed cells 1965: # immunological memory for cancer cells (mice) # immune surveillance-recognition and destruction of non-self tumor cells on their appearance 1970: lymphocytes can kill autologous melanoma cells 1991: cloning of the first tumor antigen (Belgium) 2003: therapeutic & prophylactic vaccines in the market
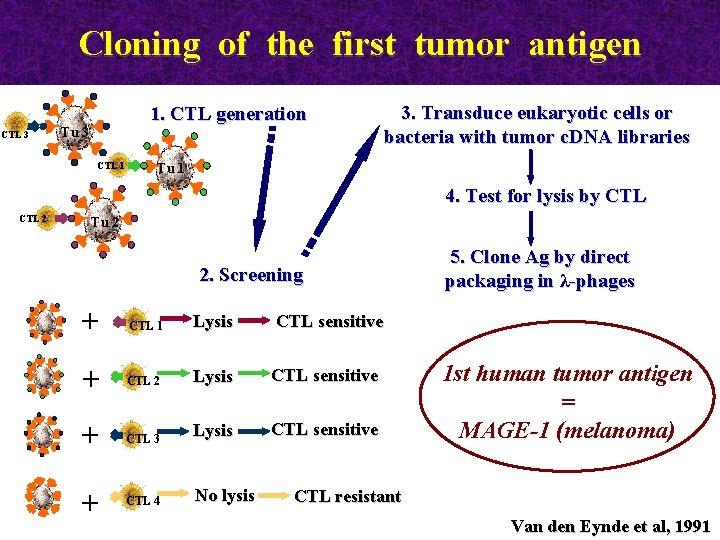
Cloning of the first tumor antigen CTL 3 1. CTL generation Τu 3 CTL 1 3. Transduce eukaryotic cells or bacteria with tumor c. DNA libraries Τu 1 4. Test for lysis by CTL 2 Τu 2 2. Screening + CTL 1 Lysis CTL sensitive + CTL 2 Lysis CTL sensitive + CTL 3 Lysis CTL sensitive + CTL 4 No lysis 5. Clone Ag by direct packaging in λ-phages 1 st human tumor antigen = MAGE-1 (melanoma) CTL resistant Van den Eynde et al, 1991

PRINCIPLE LANDMARKS IN CANCER IMMUNOLOGY 1908: positive mechanism eliminating transformed cells 1965: # immunological memory for cancer cells (mice) # immune surveillance-recognition and destruction of non-self tumor cells on their appearance 1970: lymphocytes can kill autologous melanoma cells 1991: cloning of the first tumor antigen (Belgium) 2003: therapeutic & prophylactic vaccines in the market

Commercially available cancer vaccines Therapeutic Prophylactic Ø Melacine (human melanoma) 10 -20 % OR Ø Canvacine (human melanoma) ? Ø m. Abs (Herceptin, Rituxan, Mylotarg, etc) Ø HPV vaccine (h. cervical cancer) Ø HBV vaccine (liver cancer) ? Ø mucin I (colon cancer) ? Ø breast cancer? pancreatic cancer? Finn OJ, 2003

What do we know Neoplastic transformation Genetic alterations Expression of cell surface antigens Non-self antigens are seen by the immune system cancer cells are antigenic cancer cells are immunogenic they present antigens they can activate lymphocytes Why do we die from cancer?
Where Tumor Immunology stands Basic Immunology Biochemistry and Molecular biology Tumor Immunology Cancer Immunotherapy
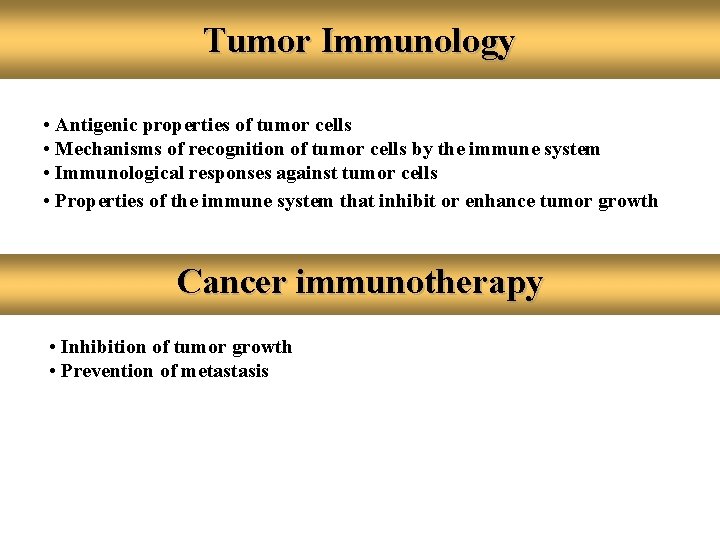
Tumor Immunology • Antigenic properties of tumor cells • Mechanisms of recognition of tumor cells by the immune system • Immunological responses against tumor cells • Properties of the immune system that inhibit or enhance tumor growth Cancer immunotherapy • Inhibition of tumor growth • Prevention of metastasis

What is the nature of tumor antigens a. Tumor specific antigens (TSA), present only on tumor cells • encoded by genes specifically expressed by tumors (MAGE) • encoded by variant forms of normal genes altered by mutations (β-catenin) b. Tumor associated antigens (TAA), present on tumor cells and some normal cells • normally expressed at certain differentiation stages or lineages (tyrosinase, Melan-A/MART-1) • mutated proteins (k-ras, p 53) • overexpressed proteins (HER-2/neu)
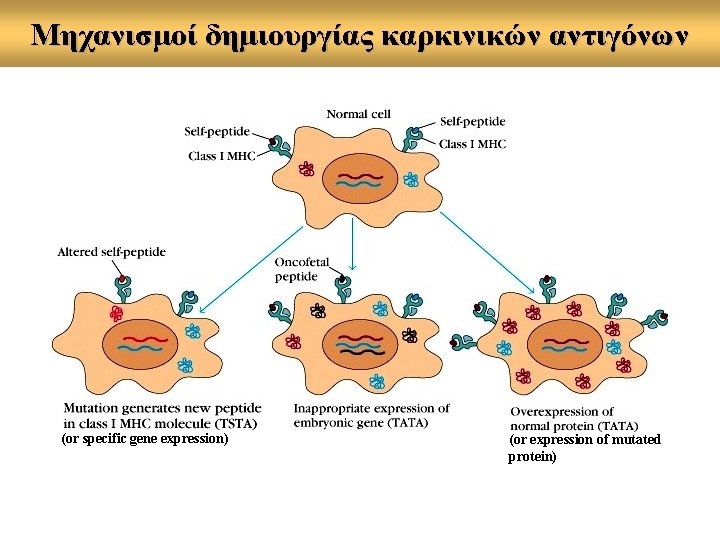
Μηχανισμοί δημιουργίας καρκινικών αντιγόνων (or specific gene expression) (or expression of mutated protein)


Cancer Diagnosis Ideal tumor markers should be: 1. 2. 3. 4. 5. specific for a tumor type released ONLY in response to tumor their levels proportional to tumor mass reflect tumor response quantitatively elevated with low tumor burden Some tumor markers used in clinical practice: α-FP, β 2 -microglobulin, β-h. CG, bombesin, CA 15 -3, CA 19 -9, CA 125, CEA, LDH, PSA, α 1 -antitrypsin, NSE (neuron-specific enolase)
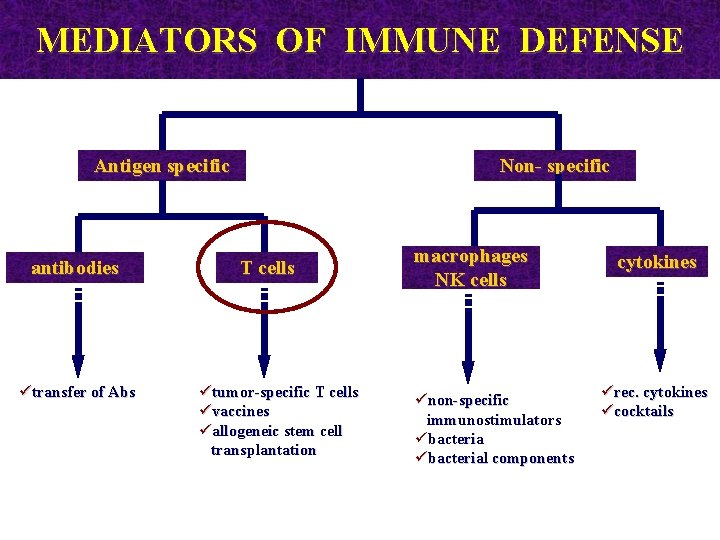
MEDIATORS OF IMMUNE DEFENSE Antigen specific antibodies ütransfer of Abs Non- specific T cells ütumor-specific T cells üvaccines üallogeneic stem cell transplantation macrophages NK cells ünon-specific immunostimulators übacterial components cytokines ürec. cytokines ücocktails
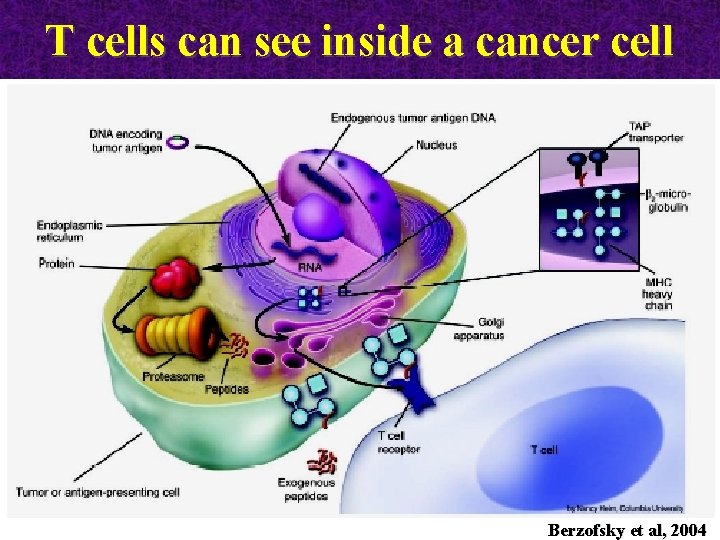
T cells can see inside a cancer cell Berzofsky et al, 2004

Recognition of tumor Ags by T cells I. A tumor cell must: Ø express MHC class I & class II molecules Ø increased levels of antigen expression (40. 000 vs 400 copies) Ø express costimulatory signals & adhesion molecules Ø produce & secrete cytokines (to differentiate and II. Aactivate cancer patient must: Ø possess the relevant T cell clones that recognize lymphocytes to effector cells) cancer antigens & self MHC molecules (directly on tumor cells or
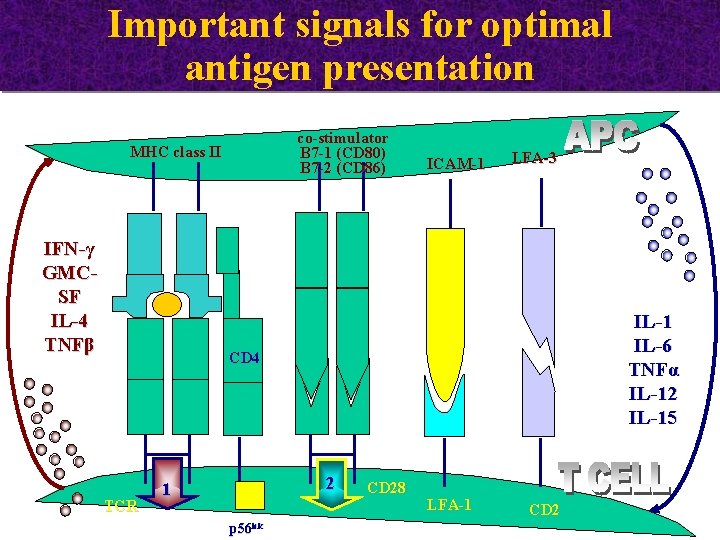
Important signals for optimal antigen presentation co-stimulator B 7 -1 (CD 80) B 7 -2 (CD 86) MHC class II IFN-γ GMCSF IL-4 TNFβ ICAM-1 LFA-3 IL-1 IL-6 TNFα IL-12 IL-15 CD 4 TCR 2 1 p 56 lck CD 28 LFA-1 CD 2

Recognition of tumor Ags by T cells I. A tumor cell must: Ø express MHC class I + class II molecules Ø increased levels of antigen expression (40. 000 vs 400 copies) Ø express costimulatory signals & adhesion molecules Ø produce & secrete cytokines (to differentiate and II. Aactivate cancer patient must: Ø possess the relevant T cell clones that recognize lymphocytes to effector cells) cancer antigens & self MHC molecules (directly on tumor cells or
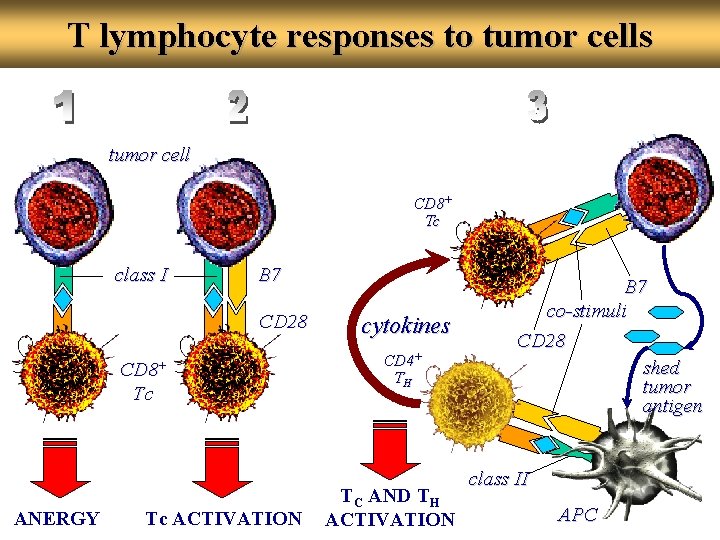
T lymphocyte responses to tumor cells tumor cell CD 8+ Tc class I B 7 CD 28 CD 8+ Tc ANERGY Tc ACTIVATION cytokines CD 4+ TH TC AND TH ACTIVATION B 7 co-stimuli CD 28 shed tumor antigen class II APC







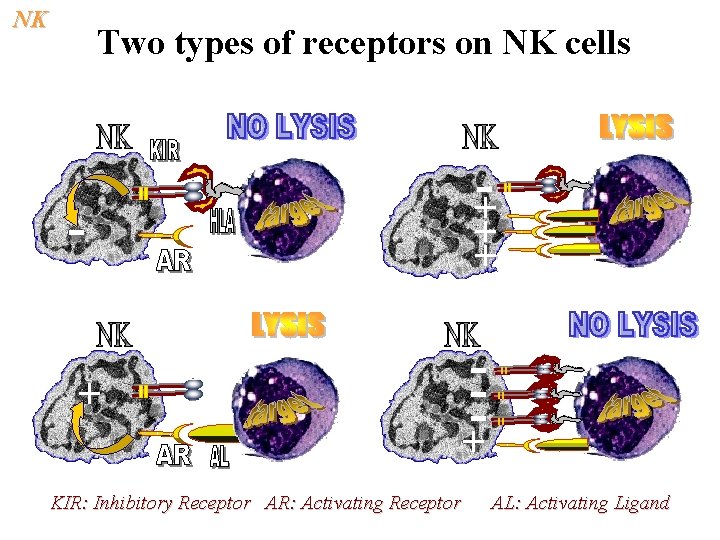
NK Two types of receptors on NK cells + KIR: Inhibitory Receptor AR: Activating Receptor -+ + + --+ AL: Activating Ligand


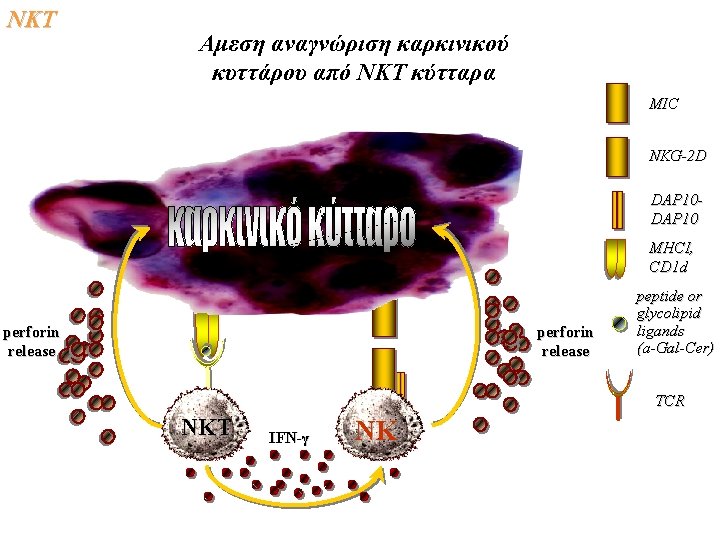
NKΤ Αμεση αναγνώριση καρκινικού κυττάρου από ΝΚΤ κύτταρα MIC NKG-2 D DAP 10 MHCI, CD 1 d perforin release peptide or glycolipid ligands (a-Gal-Cer) TCR ΝΚΤ IFN-γ ΝΚ




Potential mechanisms of tumor cell escape from host immune recognition • • • Peptide induced T cell tolerance Exhaustion of antitumor CTL Fas/Fas. L induced apoptosis Immune suppression Deficient signal transduction Failure of tumor cells to function as antigen-presenting cells: üTAA loss or down-regulation üTAP loss or down-regulation üHLA class I antigen loss or down regulation
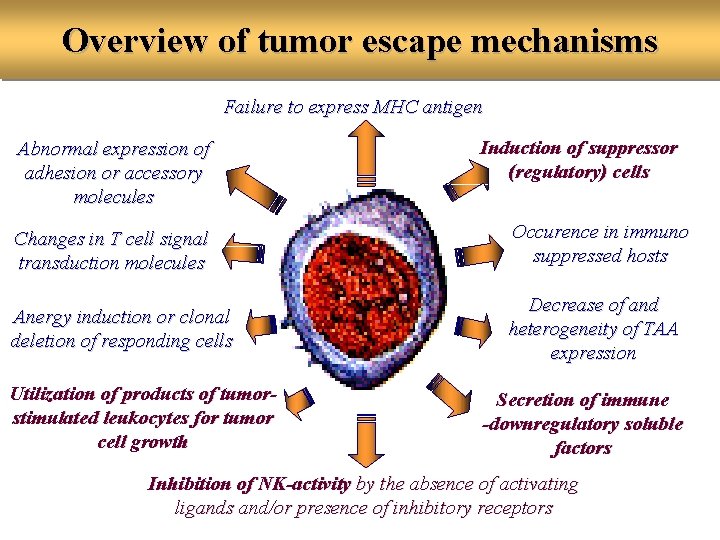
Overview of tumor escape mechanisms Failure to express MHC antigen Abnormal expression of adhesion or accessory molecules Induction of suppressor (regulatory) cells Changes in T cell signal transduction molecules Occurence in immuno suppressed hosts Anergy induction or clonal deletion of responding cells Decrease of and heterogeneity of TAA expression Utilization of products of tumorstimulated leukocytes for tumor cell growth Secretion of immune -downregulatory soluble factors Inhibition of NK-activity by the absence of activating ligands and/or presence of inhibitory receptors


Cancer Immunotherapy 3 basic concepts 1. Active immunotherapy (vaccination) 2. Adoptive or passive immunotherapy (repeated infusions of ex vivo activated cells or cytokines) 3. Gene therapy (arm the cell with impaired molecules or functions)
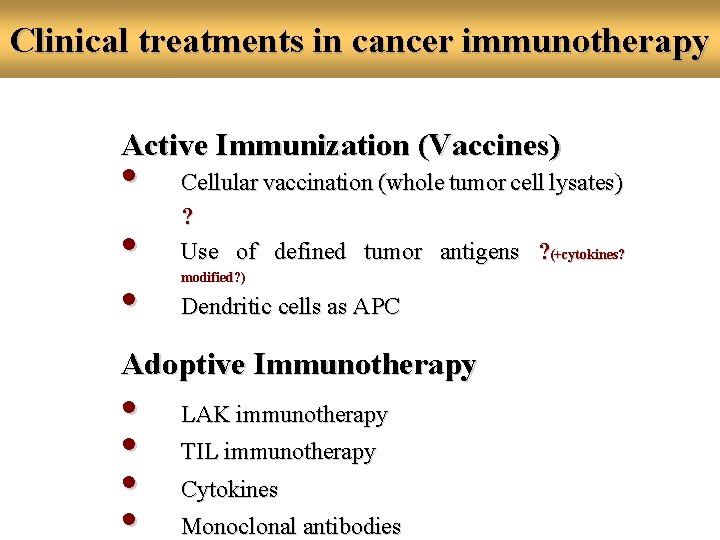
Clinical treatments in cancer immunotherapy Active Immunization (Vaccines) • • • Cellular vaccination (whole tumor cell lysates) ? Use of defined tumor antigens ? (+cytokines? modified? ) Dendritic cells as APC Adoptive Immunotherapy • • LAK immunotherapy TIL immunotherapy Cytokines Monoclonal antibodies

Cancer Vaccines Rationale: Tumor associated antigens (TAA) are immunogenic enough to induce immune responses in vivo against tumor cells expressing the same TAA


Anti-tumor vaccination strategies

Whole tumor cells as vaccines Rational: some patients have circulating T cells directed against their tumor cells. The aim is to increase their frequency. • • Advantages Autologous tumor cells comprise all relevant tumor antigens Disadvantages Tumor cells are not fully phenotypically characterized and therefore it is difficult to understand their therapeutic effect

Tumor-associated antigens as Vaccines Rational: T cells will be directed only against the patient’s tumor cells. Immunotherapy will be specific. • • Advantages Allow the systemic analysis of vaccine-induced immunity in relation to clinical responses Disadvantages An HLA-restricted CTL response can be raised to only one antigen

How to induce tumor antigen-specific T cell responses in vivo INJECTION OF PEPTIDE ANTIGENS • • • alone combined with cytokines (IL-2, IL-12, GMCSF) combined with adjuvants (CFA, IFA, BCG) Routes of injection: id, sc, it
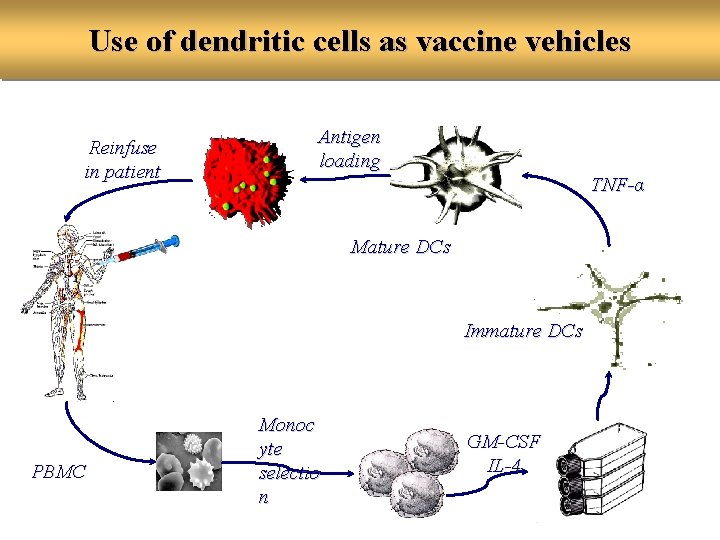
Use of dendritic cells as vaccine vehicles Reinfuse in patient Antigen loading TNF-α Mature DCs Immature DCs PBMC Monoc yte selectio n GM-CSF IL-4

Can we improve peptide-based vaccines? • • • By defining more tumor antigens, naturally processed and expressed on primary tumor cells and tumor cell lines By increasing the affinity of binding to a MHC molecule and/or the affinity of the peptide-MHC complex for the TCR By stimulating CD 8 - & CD 4 -specific anti-tumor responses By using peptides active on preclinical studies By optimizing dose, route of immunization Coadministration of appropriate adjuvants and/or cytokines
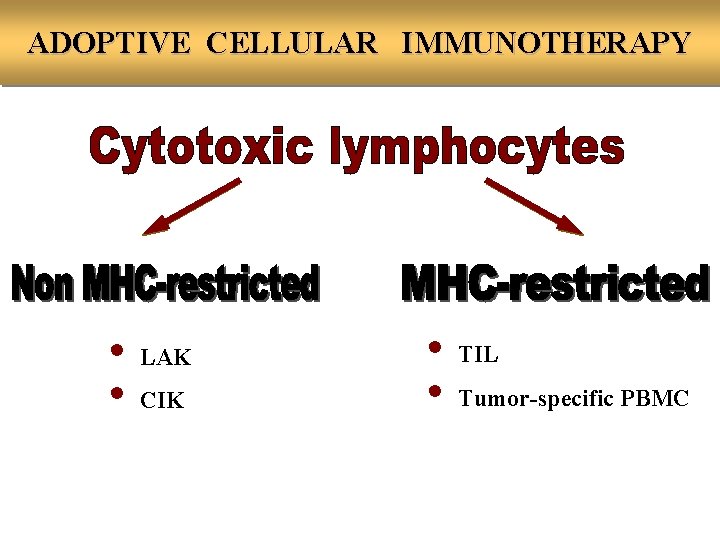
ADOPTIVE CELLULAR IMMUNOTHERAPY • LAK • CIK • TIL • Tumor-specific PBMC

TUMOR INFILTRATING LYMPHOCYTES (TIL) : LYMPHOCYTES WITH ANTI-TUMOR MHCRESTRICTED CYTOTOXICITY A. CHARACTERISTICS • TIL comprise of both CD 4 + and CD 8+ αβTCR+ CD 3+ (Τ) lymphocytes • In some types of Ca (e. g. lung Ca) γδTCR CD 3 + + ΤIL have been also reported • A small percentage of TIL (5 -10 %) express the highaffinity IL-2 receptor • Large scale expansion of TIL can be achieved in cultures with IL-2

TUMOR INFILTRATING LYMPHOCYTES (TIL) B. FUNCTIONAL CHARACTERISTICS • Freshly isolated TIL do not respond to mitogens and do not lyse tumor cells • TIL can be activated with IL-2 to produce cytokines and express cytotoxicity on response to autologous tumor cells (ATC) • Specific lysis of TIL can be inhibited by antibodies to CD 3 or to MHC class I molecules • Growth of TIL in IL-2 plus other cytokines results in increased in vitro lytic specificity for ATC (IL-2+IL-4: melanoma, IL-2+IL-1β or +IFN-γ or +TNF-a: ovarian,



Problems which appeared during treatment of patients with LAK cells and IL-2 • Toxicities associated with large amounts of interleukin 2 infused to patients • Inherently low anti-tumor activity of LAK cells • Lack of specificity for the autologous tumor • Difficulties in generating large numbers of cells

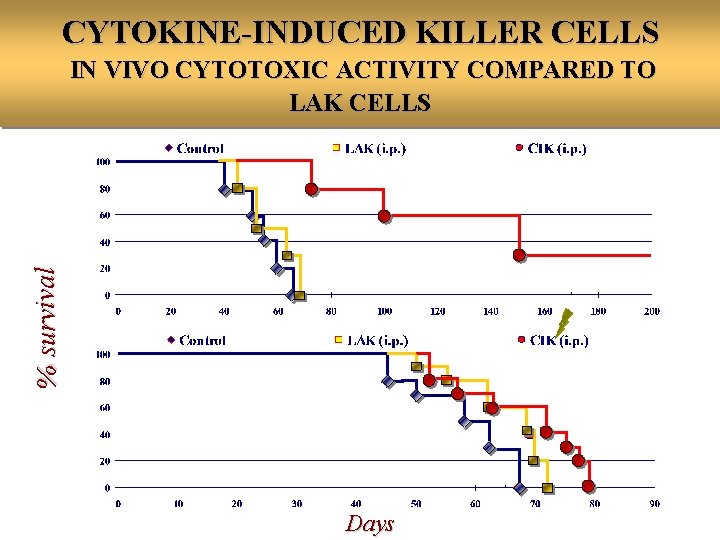
CYTOKINE-INDUCED KILLER CELLS % survival IN VIVO CYTOTOXIC ACTIVITY COMPARED TO LAK CELLS Days
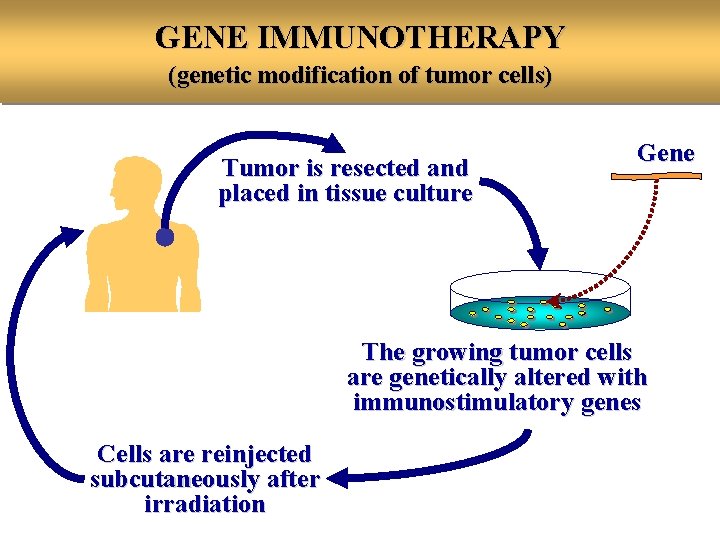
GENE IMMUNOTHERAPY (genetic modification of tumor cells) Tumor is resected and placed in tissue culture Gene The growing tumor cells are genetically altered with immunostimulatory genes Cells are reinjected subcutaneously after irradiation



Immunostimulating agents • Interferon-a: melanoma, renal carcinoma, hairy cell leukemia, CML • Interleukin-2: melanoma, renal cell carcinoma, ALL, CLL, non-Hodkin’s lymphoma • GM-CSF: melanoma • TNF-a, IL-3, IL-4, IL-6, IL-12, IFN-β, IFN-γ • BCG, alum, Cp. G oligos • Thymic peptides: thymic fraction V, thymosin α 1, prothymosin α


ANTIBODY-BASED CANCER IMMUNOTHERAPY • • • A d v a n t a g e s Production of monoclonal antibodies High affinity for antigen Induction of immune effector functions a. complement-dependent cytotoxicity b. cellular-dependent cytotoxicity (ADCC)

LIMITATIONS WITH THE USE OF m. Ab FOR CANCER IMMUNOTHERAPY • Development of human anti-mouse antibodies (generationof humanized or human m. Ab) • • • Low half-life Low tissue penetration Low immunostimulation

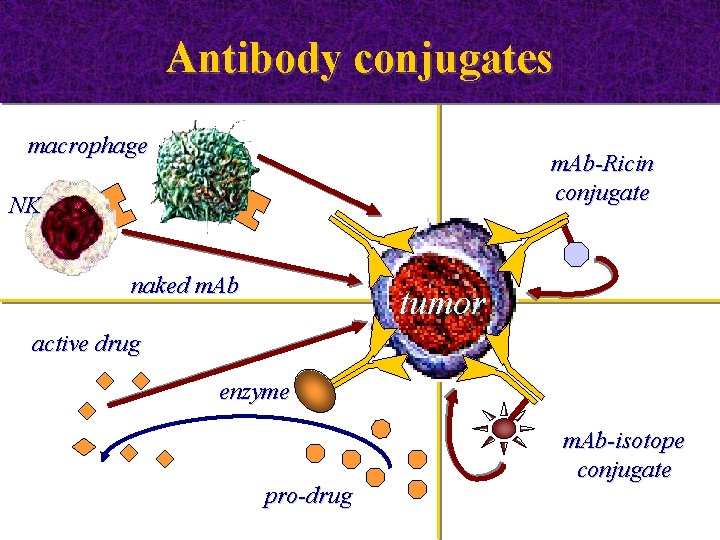
Antibody conjugates macrophage m. Ab-Ricin conjugate NK naked m. Ab tumor active drug enzyme pro-drug m. Ab-isotope conjugate
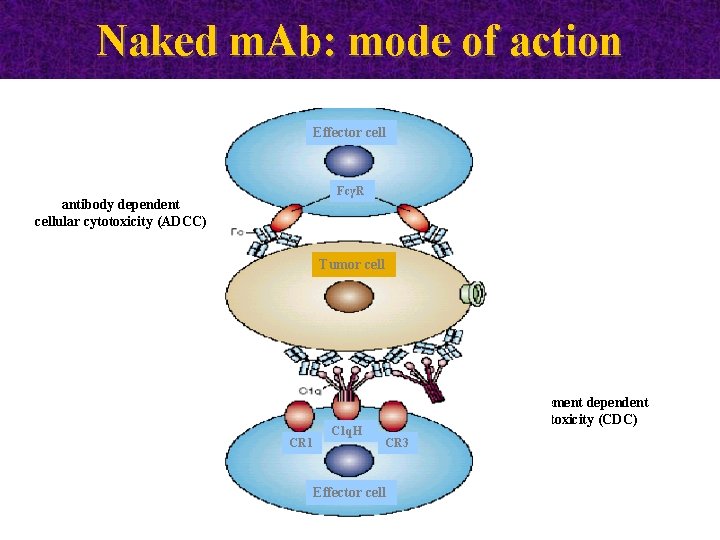
Naked m. Ab: mode of action Effector cell FcγR antibody dependent cellular cytotoxicity (ADCC) Tumor cell CR 1 C 1 q. H complement dependent cytotoxicity (CDC) CR 3 Effector cell

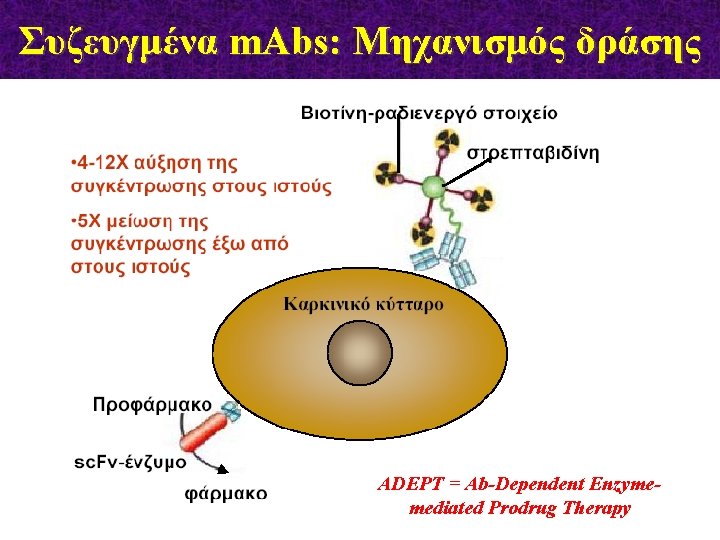
Συζευγμένα m. Abs: Μηχανισμός δράσης ADEPT = Ab-Dependent Enzymemediated Prodrug Therapy

FDA approved m. Abs m. Ab name Trade Name Capromab pendetide (111 In) Prosta. Scint Rituximab Rituxan Used for the treatment of in Prostate cancer 1996 B-cell lymphoma 1997 Trastuzumab Herceptin Breast cancer 1998 Gemtuzumab ozogamicin Mylotarg Acute myelogenous leukemia (AML) 2000 Alemtuzumab Campath Chronic lymphocytic leukemia (CLL) 2001 Ibritumomab tiuxetan (90 Y) Zevalin B-cell lymphoma 2002 Tositumomab (131 I) Bexxar B-cell lymphoma 2003 Cetuximab Erbitux Colorectal cancer 2004 Head and neck 2006 Colorectal cancer 2004 Bevacizumab Avastin
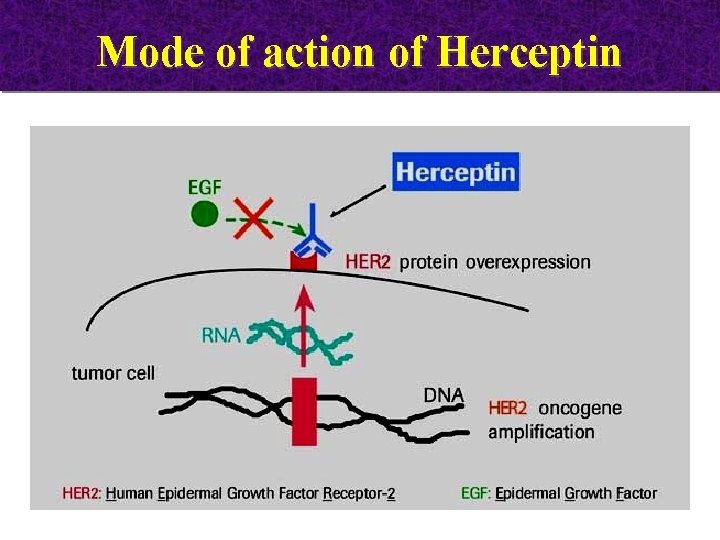
Mode of action of Herceptin

Unconjugated m. Abs in Advanced Clinical Testing m. Ab name Apolizumab Agent/Target Human anti-HLA-DR To be used for treatment of CLL, SLL Neuroblastoma Chimeric 14. 18 Chimeric anti-GD 2 Epratuzumab Humanized anti-CD 22 NHL Galiximab Humanized anti-CD 80 NHL Hu. Max-CD 4 Human anti-CD 4 Lumilliximab Humanized anti-CD 23 MDX-010 Matuzumab Anti-CTLA-4 Humanized antri. EGFR Cutaneous T cell lymphoma CLL Melanoma Colorectal cancer Orgegovomab Murine anti-CA-125 Ovarian cancer Panitumumab Human anti-EGFR NSCLC, colorectal, renal Humanized anti-HER 2 Breast, prostate, ovarian Pertuzumab Rencarex Vitaxin Chimeric anti-G 250 Humanized anti-αvβ 3 integrin Kidney Melanoma, prostate
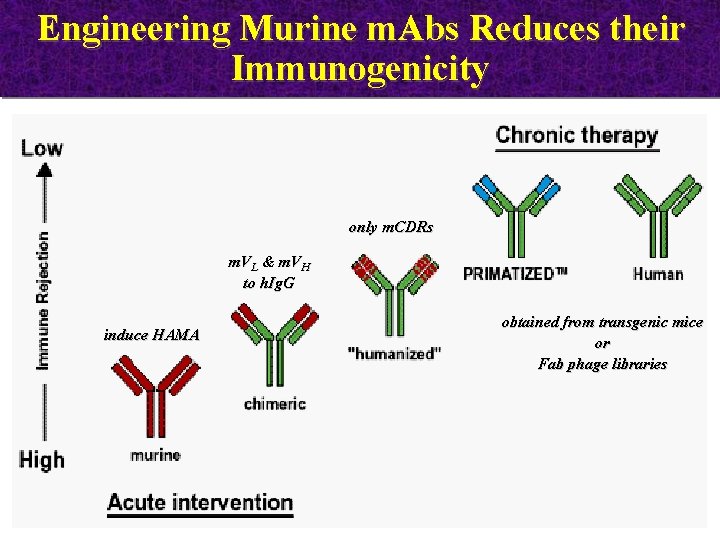
Engineering Murine m. Abs Reduces their Immunogenicity only m. CDRs m. VL & m. VH to h. Ig. G induce HAMA obtained from transgenic mice or Fab phage libraries

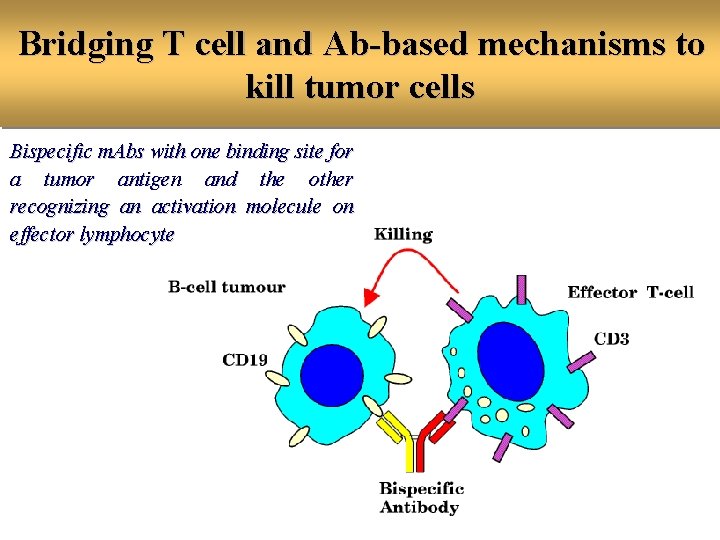
Bridging T cell and Ab-based mechanisms to kill tumor cells Bispecific m. Abs with one binding site for a tumor antigen and the other recognizing an activation molecule on effector lymphocyte
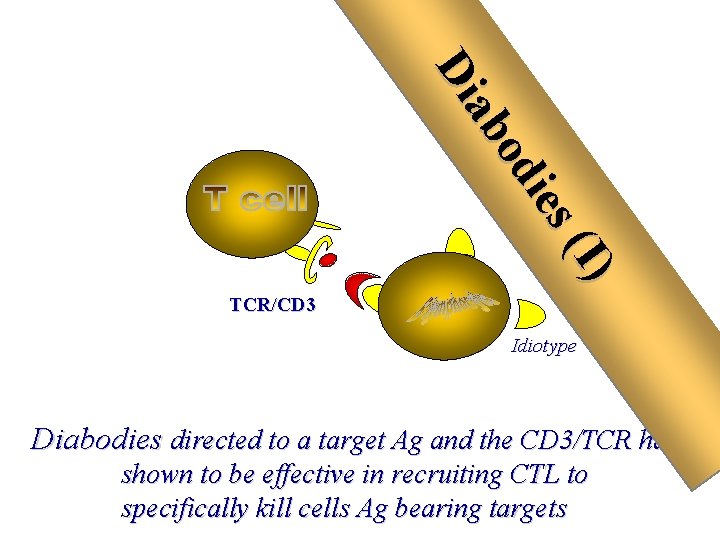
(I ) ies od ab Di TCR/CD 3 Idiotype Diabodies directed to a target Ag and the CD 3/TCR have shown to be effective in recruiting CTL to specifically kill cells Ag bearing targets
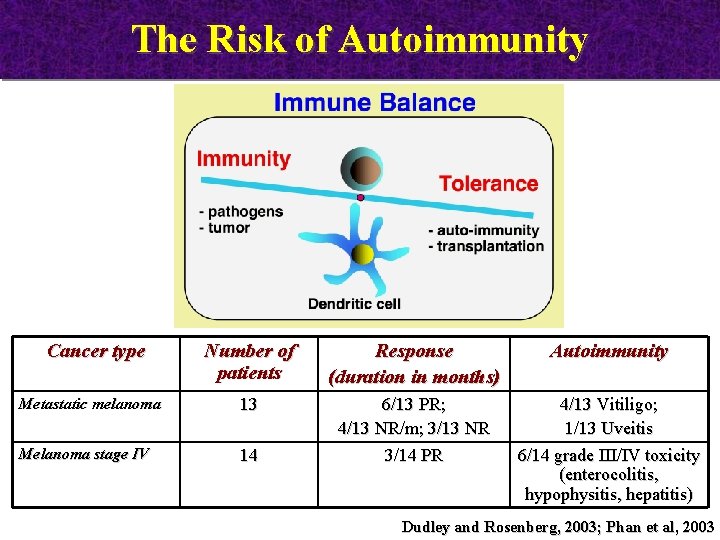
The Risk of Autoimmunity Cancer type Number of patients Response (duration in months) Autoimmunity Metastatic melanoma 13 Melanoma stage IV 14 6/13 PR; 4/13 NR/m; 3/13 NR 3/14 PR 4/13 Vitiligo; 1/13 Uveitis 6/14 grade III/IV toxicity (enterocolitis, hypophysitis, hepatitis) Dudley and Rosenberg, 2003; Phan et al, 2003
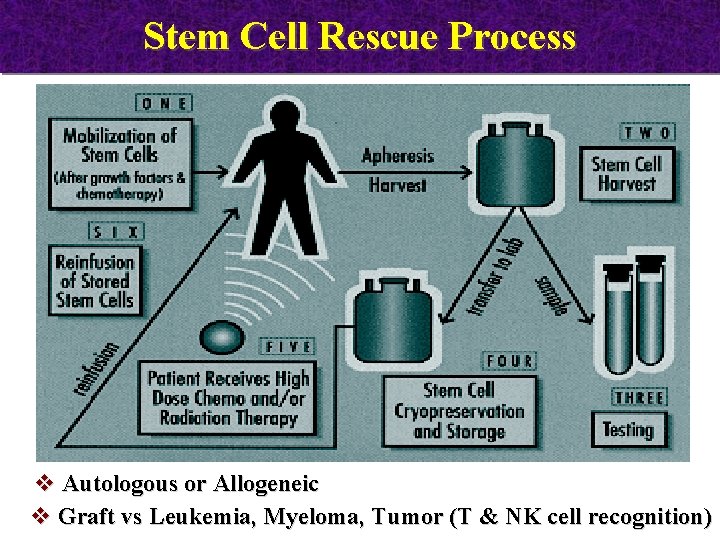
Stem Cell Rescue Process v Autologous or Allogeneic v Graft vs Leukemia, Myeloma, Tumor (T & NK cell recognition)

Malignancies treatable with Stem Cell Transplantation Autologous Allogeneic Neuroblastoma Acute Lymphocytic Leukemia Non-Hodgkin’s Lymphoma Acute Myeloid Leukemia Hodgkin’s Lymphoma Chronic Myelogenous Leukemia Wilm’s Tumor Juvenile Chronic Myelocytic leukemia Ewing’s Sarcoma Myelodysplastic Syndrome + Brain Tumors
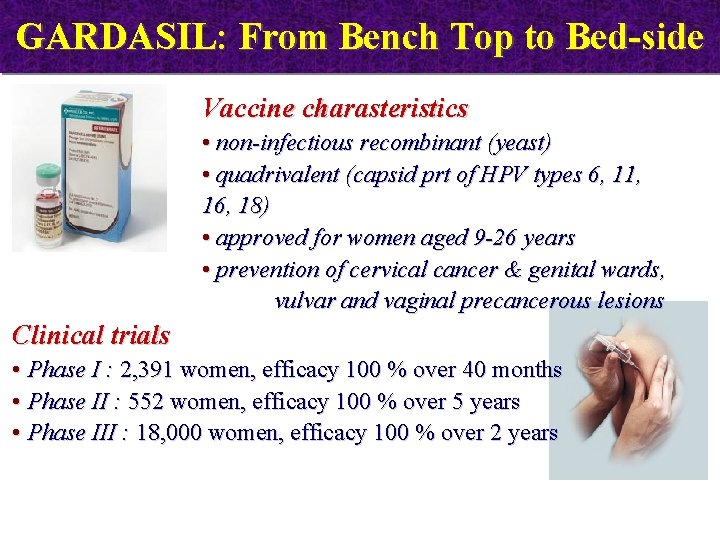
GARDASIL: From Bench Top to Bed-side Vaccine charasteristics • non-infectious recombinant (yeast) • quadrivalent (capsid prt of HPV types 6, 11, 16, 18) • approved for women aged 9 -26 years • prevention of cervical cancer & genital wards, vulvar and vaginal precancerous lesions Clinical trials • Phase I : 2, 391 women, efficacy 100 % over 40 months • Phase II : 552 women, efficacy 100 % over 5 years • Phase III : 18, 000 women, efficacy 100 % over 2 years


Mechanisms we should concentrate on ØIncrease immune response Enhance cell-mediated immunity ØIncrease tumor-specific immune responses Direct effect on tumor cells ØRestore immunocompetence ØInduce prolonged tumor immunity ØRemove inhibitory factors (eg. tumor antigen immune complexes)

- Slides: 88