Tufts Lithiumion Thin Film Rechargeable Battery ee e

Tufts Lithium-ion Thin Film Rechargeable Battery
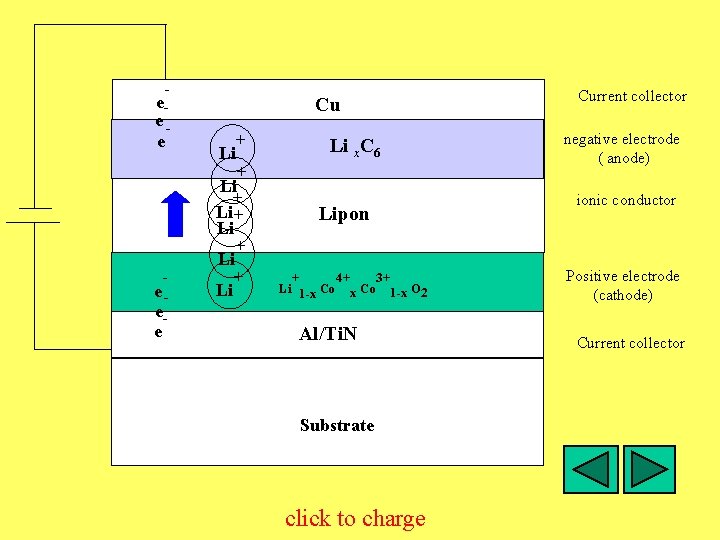
ee- e Cu Li + + Li ee- e Li Li Li C x C 6 Lipon Current collector negative electrode ( anode) ionic conductor + + + 4+ Co 3+ Li O 2 O Li 1 -x Co 1 -x 2 Al/Ti. N Substrate click to charge Positive electrode (cathode) Current collector
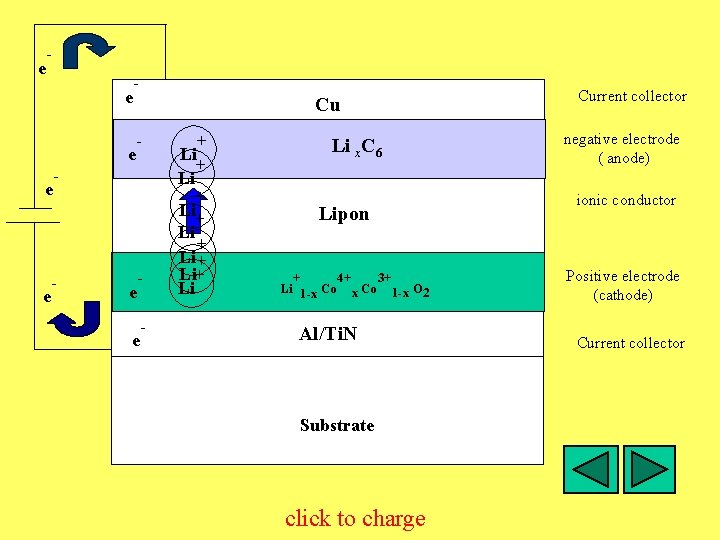
e - e - + - Li + Li - Li+ Li+ Li e e Cu + e - Li C x C 6 Lipon + 4+ Co 3+ Li O 2 O Li 1 -x Co 1 -x 2 Al/Ti. N Substrate click to charge Current collector negative electrode ( anode) ionic conductor Positive electrode (cathode) Current collector
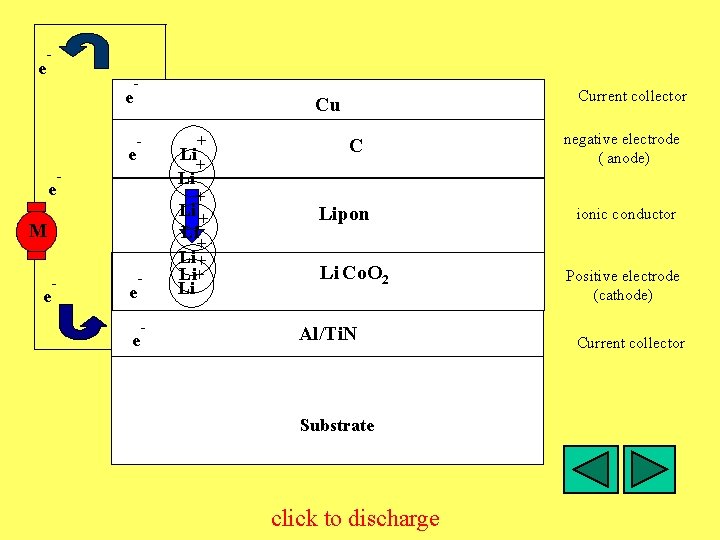
e - e e Cu - - e Li + M - + e - Li + Li+ Li Li. Cx. C 6 Lipon + Li Co. O 4+ 23+ Li 1 -x Co 1 -x O 2 Al/Ti. N Substrate click to discharge Current collector negative electrode ( anode) ionic conductor Positive electrode (cathode) Current collector
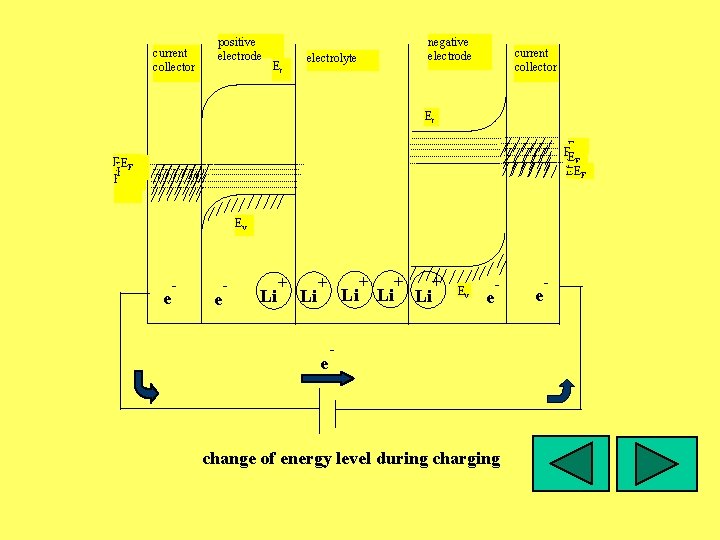
current collector positive electrode Ec negative electrode electrolyte current collector Ec EF EE F EFF EE FF EF EF Ev e - Li + + e Li Li + Ev e - - change of energy level during charging e -
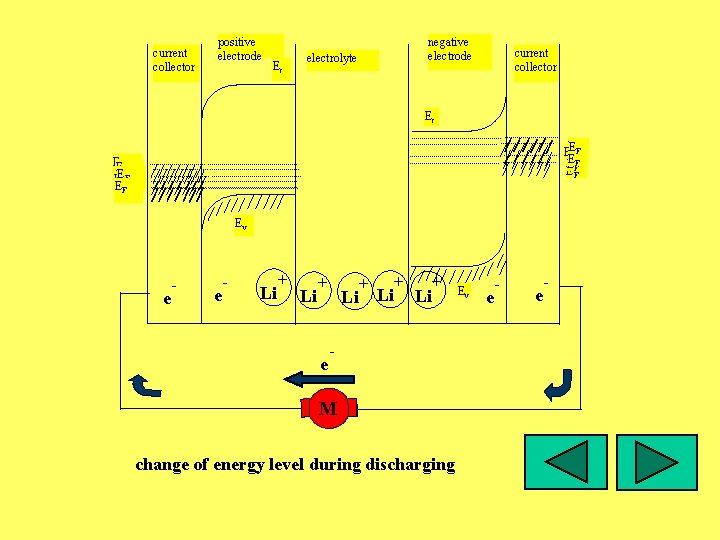
current collector positive electrode Ec negative electrode electrolyte current collector Ec EEFF EF E EFF EEFF EF Ev e - Li + e + Li Li + - M change of energy level during discharging Ev e -
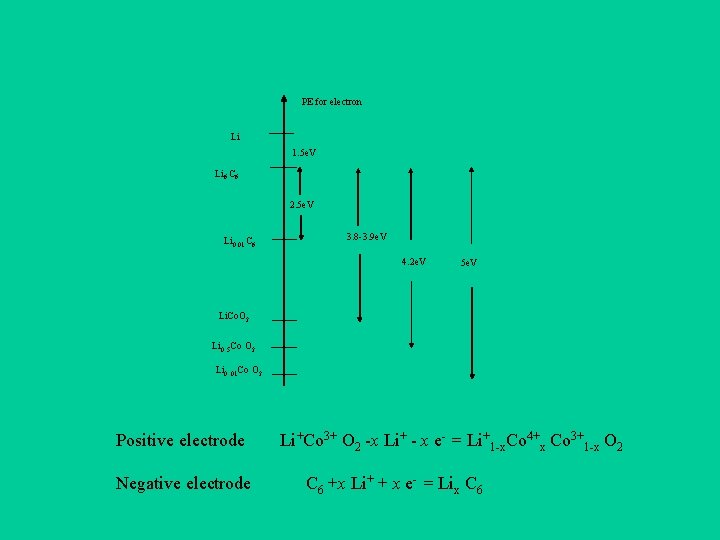
PE for electron Li 1. 5 e. V Li 6 C 6 2. 5 e. V Li 0. 01 C 6 3. 8 -3. 9 e. V 4. 2 e. V 5 e. V Li. Co. O 2 Li 0. . 5 Co O 2 Li 0. . 01 Co O 2 Positive electrode Negative electrode Li+Co 3+ O 2 -x Li+ - x e- = Li+1 -x. Co 4+x Co 3+1 -x O 2 C 6 +x Li+ + x e- = Lix C 6
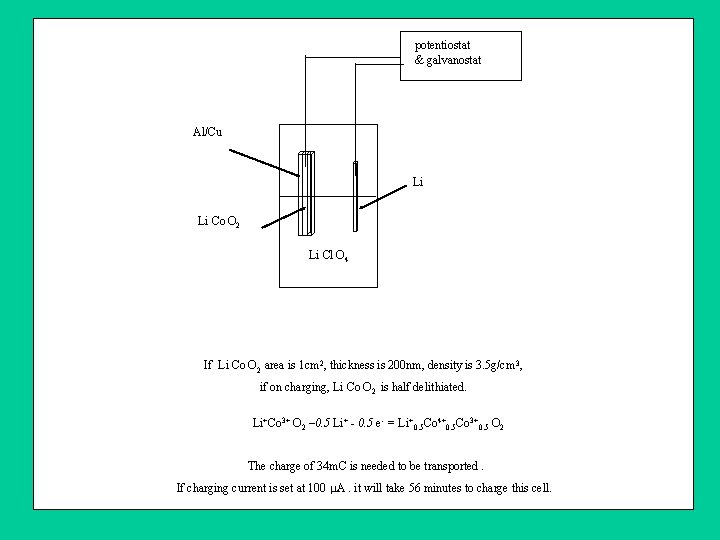
potentiostat & galvanostat Al/Cu Li Li Co O 2 Li Cl O 4 If Li Co O 2 area is 1 cm 2, thickness is 200 nm, density is 3. 5 g/cm 3, if on charging, Li Co O 2 is half delithiated. Li+Co 3+ O 2 – 0. 5 Li+ - 0. 5 e- = Li+0. 5 Co 4+0. 5 Co 3+0. 5 O 2 The charge of 34 m. C is needed to be transported. If charging current is set at 100 A. it will take 56 minutes to charge this cell.
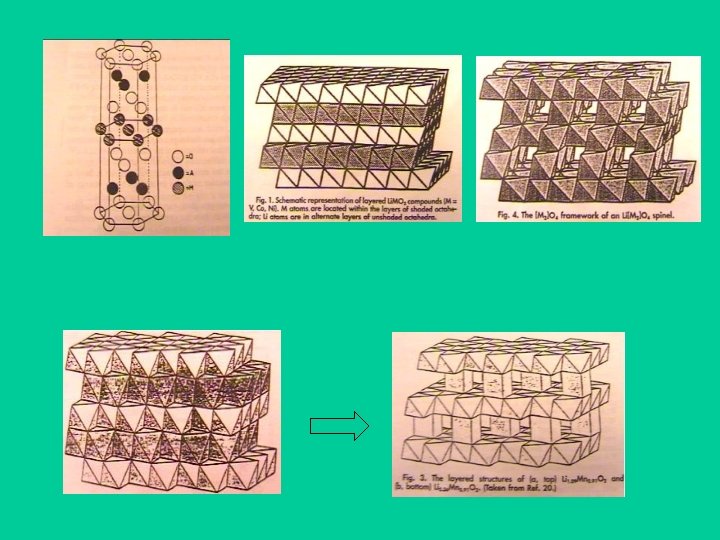
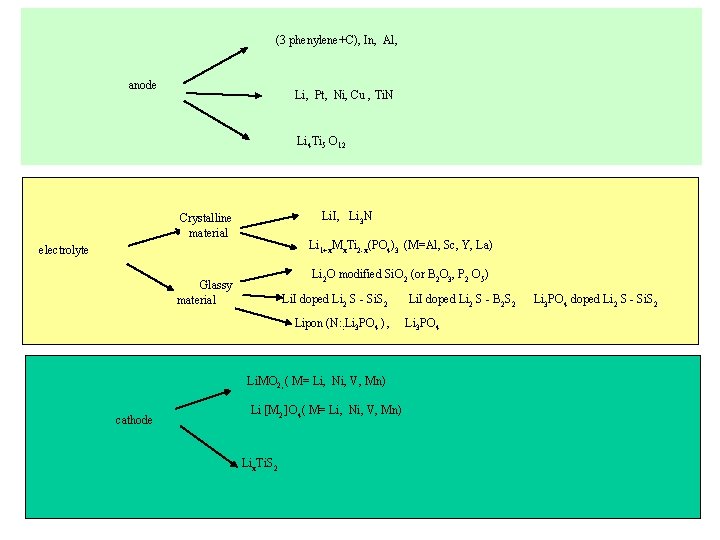
(3 phenylene+C), In, Al, anode Li, Pt, Ni, Cu , Ti. N Li 4 Ti 5 O 12 Li. I, Li 3 N Crystalline material Li 1+x. Mx. Ti 2 -x(PO 4)3 (M=Al, Sc, Y, La) electrolyte Li 2 O modified Si. O 2 (or B 2 O 3, P 2 O 5) Glassy material Li. I doped Li 2 S - Si. S 2 Lipon (N: : Li 3 PO 4 ) , Li. MO 2, ( M= Li, Ni, V, Mn) cathode Li [M 2 ]O 4( M= Li, Ni, V, Mn) Lix. Ti. S 2 Li. I doped Li 2 S - B 2 S 2 Li 3 PO 4 doped Li 2 S - Si. S 2
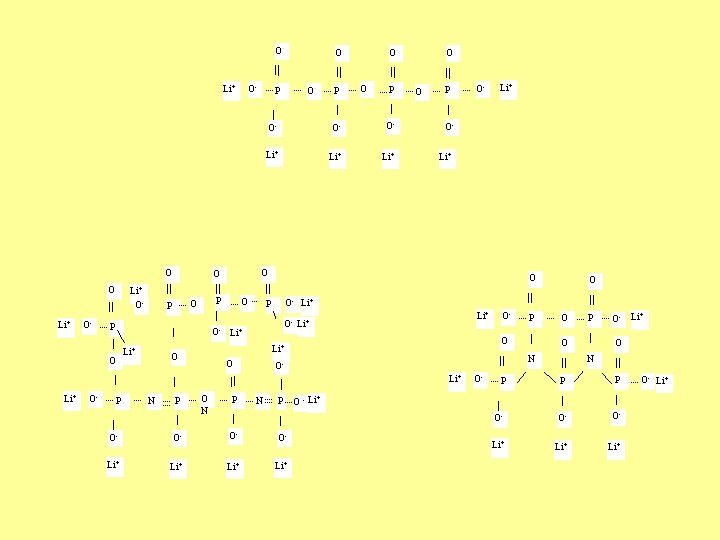
O Li+ O- P O- O P O- O- Li+ Li+ O- Li+ O O- P N P O- O- O- Li+ Li+ O- P OLi+ P O O O- P O N ON O O- Li+ P Li+ O- O O P Li+ O- O OO N P P O- O- O- Li+ Li+ P Li+ + O - Li O- Li+
- Slides: 11