Tuberculosis February 21 2007 Timothy R Sterling M

Tuberculosis February 21, 2007 Timothy R. Sterling, M. D. Vanderbilt University Medical Center Director of TB Research, Nashville-Davidson Metro Health Department

Overview • TB Epidemiology – TB/HIV – Drug Resistance • Diagnosis of TB • TB Treatment – Treatment Regimens – TB/HIV • Diagnosis and Treatment of Latent Infection • Infection control

Global TB Epidemiology 2004 • Estimated # new TB cases: 8. 9 million • Estimated # prevalent cases: 14. 6 million • Estimated # deaths: 1. 7 million • Estimated # infections: 2 billion – 33% of population WHO Report 2006. Global tuberculosis control. WHO/HTM/TB/2006. 362


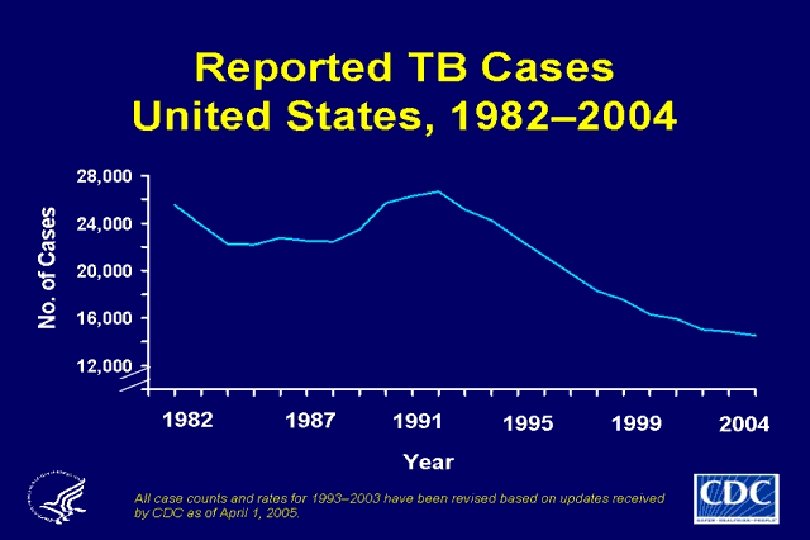
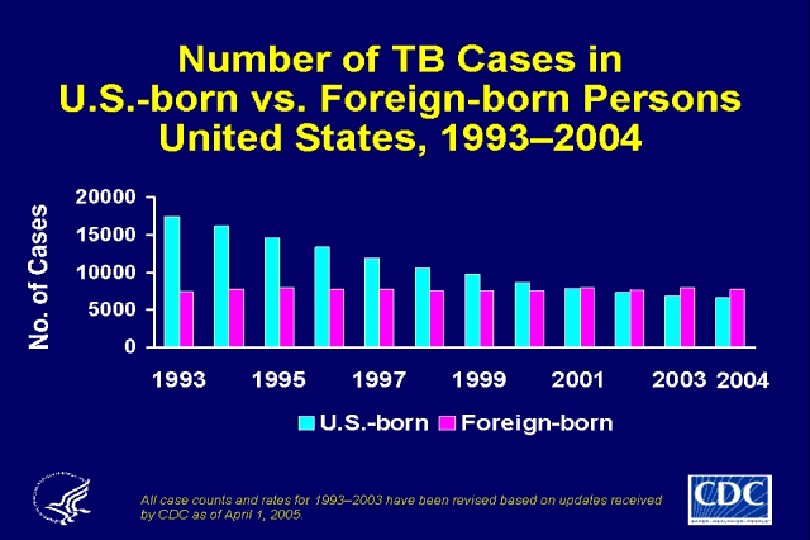

Impact of HIV on TB Epidemiology • HIV responsible for the global TB resurgence – sub-Saharan Africa


Global Incidence of MDR-TB Resistance to INH + rifampin • TB cases in 2004 – Resistance surveys for: • New cases (90 countries) • Previously treated cases (77 countries) – Logistic regression estimated rate among all 184 countries in the world • Global estimates: – 424, 203 cases in 2004 – 4. 3% of all new and previously treated TB cases • China, India, Russian Federation accounted for 62% of estimated global burden of MDR-TB WHO—Zignol M et al. J Infect Dis 2006; 194: 479 -85.

Multidrug-Resistant Tuberculosis • Global “hotspots”: Russia, Latvia, India, Dominican Republic • Risk factors: – prior TB treatment – noncompliance – adding one drug to failing regimen – HIV? ; outbreaks among AIDS patients • Treatment: 18 -24 months – complex regimens

XDR TB Extensively Drug Resistant Revised WHO Case Definition (Oct 11, 2006): Resistance to at least isoniazid and rifampin (MDR) plus resistance to: • Fluoroquinolones + • 1 of the second-line injectable drugs – amikacin, kanamycin, or capreomycin

Diagnosis of Tuberculosis • • Clinical signs/symptoms Chest radiograph Microscopy Culture – DNA probe after growth in culture • Nucleic acid amplification

Sputum • Sputum expectoration – 3 samples • Every 8 hours, with 1 specimen in early AM – For hospitalized patients; more convenient though few data • Sputum induction – In persons from whom expectorated sputum cannot be obtained or is smear-negative – Repeated sputum induction (3 -4) increases yield – Cost-effective, even in resource-poor countries • Bronchoscopy – Unclear whether yield is greater than with repeated sputum induction – Consider when sputum induction negative but CXR suggests TB

Microscopy • Techniques: – Acid-Fast (Ziehl-Neelsen; Kinyoun) – Fluorochrome (auramine-rhodamine) • Faster, easier to interpret • Sputum – + smear requires 5, 000 -10, 000 bacilli/ml – 50% (34 -80%) sensitive; nonspecific • Extrapulmonary specimens – Lower sensitivity

Diagnosis of TB Culture • 80% sensitive; 98% specific • Solid media: – 7 H-11, Lowenstein-Jensen – 21 -42 days required for growth • Liquid media: – 7 H-12 Bactec (radiometric); MGIT (fluorescent) – 5 -10 days for growth • DNA probe once growth in culture: – M. tuberculosis complex, MAI, M. kansasii, M. gordonae

Diagnosis of TB Nucleic Acid Amplification • FDA-approved for respiratory specimens from untreated patients AFB smear + AFB smear neg Sensitivity 95% 48% Specificity 98% 95% • Greater sensitivity if TB suspected clinically

Diagnosis of TB Nucleic Acid Amplification • Does not replace need to obtain AFB smear and culture • If high index of suspicion but NAA negative, TB has not been ruled out • Expensive • Extrapulmonary specimens: limited data, but less sensitive

Treatment of TB • Goal of therapy: – kill M. tuberculosis, prevent resistance + relapse • Induction phase: – Isoniazid kills 95% of organisms (growing rapidly) during first 2 days of treatment – RIF, PZA then supplant INH in cidal role during the 2 month induction phase (slowly metabolizing bacilli) • Continuation phase – Rifampin primarily effective against persistent bacilli, though INH also cidal Mitchison DA. IJTLD 2000; 4: 796 -806

Treatment of TB • Culture-positive pulmonary tuberculosis caused by drug-susceptible organisms – American Thoracic Society – Centers for Disease Control & Prevention – Infectious Diseases Society of America Am J Respir Crit Care Med 2003; 167: 603 -62

Initial Treatment Regimens Drug Combinations and Dosing Intervals Initial 2 weeks HRZE daily + 6 weeks HRZE 2 x/wk Continuation 18 weeks HR 2 x/week H/RPT 1 x/week* Rating HIV- HIV+ A (II) B (I) E (I) * no cavity on initial CXR, and smear-negative after 2 months. Extend Rx if cx+ at 2 mos H: isoniazid R: rifampin Z: pyrazinamide E: ethambutol RPT: rifapentine

Additional Points • Ethambutol can be discontinued as soon as susceptibility to isoniazid + rifampin demonstrated • Obtain monthly sputum cultures until 2 consecutive negative cultures – Culture after 2 months of treatment VERY important • HIV testing recommended in all patients

Optimal Duration of TB Treatment Regardless of HIV status Site of Disease Pulmonary Bone/joint CNS/meningeal Other extrpulmonary Duration 6 months* 6 -9 months 9 -12 months 6 months *Extend to 9 months if cavitary and culture + at 2 months American Thoracic Society, Centers for Disease Control, IDSA. Treatment of Tuberculosis. AJRCCM 2003; 167: 603 -62

Renal Failure • Drugs excreted by kidneys: – ethambutol – pyrazinamide (metabolites) • Maintain dose, but increase interval: – Thrice-weekly dosing • Administer drugs after hemodialysis (DOT) – Pyrazinamide, cycloserine dialyzed out

Hepatic Disease Treatment when underlying liver disease • Treat with standard therapy • Other options: – RZE x 6 months – HRE x 9 months – RE (+/- FQ) x 12 months (no data) • Check baseline transaminases, bili, alk – Follow closely, along with symptoms H: isoniazid R: rifampin Z: pyrazinamide E: ethambutol FQ: fluoroquinolone

Hepatic Disease Treatment when hepatitis develops on therapy • Stop treatment: – isoniazid, rifampin, pyrazinamide • Check hepatitis A, B, C serology • Assess for other hepatotoxins – alcohol – acetominophen • Could continue treatment with ethambutol, aminoglycoside, fluoroquinolone • Sequential drug rechallenge of 1 st-line agents when AST < 2 x upper limit of normal

Pregnancy • No aminoglycosides – Congenital deafness • Pyrazinamide: – U. S. : – WHO, IUATLD: probably OK OK • Breastfeeding fine with 1 st-line agents

Children • Treatment same as in adult, except: – No ethambutol if visual acuity cannot be monitored, unless: • High risk of drug resistance (INH) • Upper lobe infiltrate/cavity (higher organism burden) • Cultures often difficult to obtain • Rely on susceptibilities of presumed source case

Recommendations for Treatment of TB in HIV-Infected Patients • TB/HIV patients with CD 4 < 100 should not receive once- or twice- weekly therapy – Daily therapy during induction – Daily or thrice-weekly therapy during continuation MMWR 2002; 51: 214 -5

Treatment of TB/HIV Drug Interactions • Possible combinations: – rifampin + efavirenz – rifabutin + ritonavir-boosted protease inhibitors • Avoid these combinations: – rifampin + : saquinavir, indinavir, nelfinavir, amprenavir (fos), atazanavir, or delavridine – rifabutin +: delavridine, saquinavir • Nucleoside/tide reverse transcriptase inhibitors and enfuvirtide not affected rifamycins, so can be given

Immune Reconstitution Inflammatory Syndrome • Clinical Manifestations – Constitutional: fever, weight loss, – Pulmonary: cough, increased infiltrates – Extrapulmonary: • Lymphatic: increased cervical, intra-thoracic, intra-abdominal adenopathy • Serositis: pleural, pericardial effusions • CNS: expanding tuberculomas • Other: soft tissue, bone abscesses, skin, +PPD

Risk Factors for IRIS In (roughly) decreasing order of importance • HAART initiation within 2 months of starting anti -tuberculosis treatment • Disseminated/extrapulmonary TB • Low baseline CD 4 (< 100/mm 3)--trend, but consistent • Increase in CD 4% on HAART • HIV-1 RNA decline on HAART • Antiretroviral therapy-naïve Narita 1998, Wendel 2001, Navas 2002, Breen 2004, Breton 2004, Burman 2004, Shelburne 2005

Diagnosis of M. tuberculosis infection Tuberculin skin test (TST) • Tuberculin=broth culture filtrate of tubercle bacilli – Purified protein derivative (PPD), a standardized form of tuberculin, was introduced in 1934 • Contains ~ 200 antigens, including those shared by M. bovis BCG and non-tuberculous mycobacteria • T-cells sensitized by M. tuberculosis infection respond to M. tuberculosis antigens in PPD and release IFN- – Cutaneous induration due to delayed-type hypersenstivity to intradermal injection of PPD • Positive test determined by determining mm of induration – Inter- and intra-reader variability – Possible human error

Tuberculin Skin Test • Sensitivity: – Presumably high in latently infected persons with normal immune response • Decreased in immunocompromised patients: – HIV (CD 4 < 100), corticosteroids, other immunosuppressants, young children (< 1 year) • False negative until 8 -12 weeks after infection • Specificity: – False positives due to environmental mycobacteria, BCG • Decreased specificity and positive predictive value in populations at low risk for TB infection • Improved specificity if greater mm induration for + test

Interferon- Release Assays • Detection of IFN- or IFN- -producing T-cells after stimulation of sensitized T-cells by M. tuberculosis antigens – Assesses cell-mediated immunity • Early-generation tests used PPD as the stimulus – As non-specific as the TST • More recent assays have used synthetic peptides of 2 proteins present in M. tuberculosis, but not BCG or most nontuberculous mycobacteria (NTM): – Early secretory antigen target 6 (ESAT-6) – Culture filtrate protein-10 (CFP-10)

CDC Guidelines Quanti. FERON-TB Gold • Can be used in all circumstances in which TST used: – Contact investigations, recent immigrants (BCG), serial testing (e. g. , health-care workers) • TB risk among persons with + test unknown • Limited data: – – – Immunocompromised (e. g. , HIV) Close contacts Persons at risk of progressing to TB Children (no data in persons < 17 years old) Persons who undergo periodic screening (HCW) MMWR 2005; 54(RR-15): 49 -55.

Indications for Treatment of Latent Infection • Perform skin testing only on persons at high risk for progression to active TB • If persons in these high-risk groups are latently infected (i. e. PPD+), treat regardless of age Am J Resp Crit Care Med 2000; 161: S 221

Treatment of Latent TB Infection 2000 ATS/CDC Regimen INH q. D x 9 months INH biw x 9 months INH q. D x 6 months INH biw x 6 months HIVAII BI BII HIV+ AII BII CI CII A: preferred I: Randomized Clinical Trial B: acceptable alternative II: non. RCT C: offer when cannot give A, B III: expert opinion

Treatment of Latent TB Infection 2000 ATS/CDC Regimen HIV- HIV+ RIF q. D x 4 months BIII A: preferred B: acceptable alternative C: offer when cannot give A, B I: Randomized Clinical Trial II: non. RCT III: expert opinion

Initiating Respiratory Isolation • Policies vary at different hospitals • Cough > 2 weeks and abnormal CXR – Additional symptoms c/w TB • Epidemiology suggestive of TB: – Recent TB exposure – Incarceration – HIV (10% may have negative CXR)

Discontinuing Respiratory Isolation TB suspects • CDC guidelines: – 3 negative AFB smears – Collected at least 8 (not 24) hours apart – At least one collection during early AM • Based on expert opinion, not evidence • To get patients out of isolation in < 48 hours – Another diagnosis assigned – Infectious TB unlikely MMWR 2005; 54(RR-17): 1 -121.

When to Refer a TB Suspect to the Health Department • EARLY • As soon as they start anti-TB therapy • Allows for: – TB Clinic staff to visit patient in hospital • Establish rapport • Review medications, drug-drug interactions Nashville Metro Health Department TB clinic telephone #: 340 -5650

When Can a TB Suspect be Discharged? • On appropriate anti-TB treatment • Health department notified and follow-up, treatment plan in place • Stable residence at verifiable address

When Can a TB Suspect be Discharged? • They can be discharged before they have 3 negative smears unless “home” includes: – Young children (< 5 y. o. ) – Immunocompromised (e. g. , HIV) – Prison – Nursing home – Homeless shelter
- Slides: 44