Tuberculosis Drug Resistance EPID 816 Winter 2016 Outline

Tuberculosis Drug Resistance EPID 816, Winter 2016

Outline • A brief introduction of anti-TB drug action mechanisms (JC) • Epidemiology of drug resistant TB – The global status – Factors influencing the occurrence, distribution, and spread of drug resistant TB • Biological basis of Mycobacterium tuberculosis drug resistance • Interdisciplinary approach to study epidemiology of drug resistant TB

Anti-tuberculosis Drugs First-Line Drugs Second-Line Drugs • Isoniazid (INH) • Streptomycin (SM) • Rifampin (RIF) • Pyrazinamide (PZA) • Ethambutol (EMB) • Rifabutin* • Rifapentine (RPT) • Cycloserine • p-Aminosalicylic acid • Ethionamide • Amikacin or kanamycin* • Capreomycin • Levofloxacin* • Moxifloxacin* • Gatifloxacin* * Not approved by the U. S. Food and Drug Administration for use in the treatment of TB

MDR, XDR and TDR Multidrug-resistant (MDR) TB is caused by bacteria resistant to isoniazid and rifampicin, the two most effective first-line anti-TB drugs, originally developed and introduced in the 1950 s and 1960 s. Extensively drug-resistant (XDR) TB is resistant to the same drugs as MDR TB (isoniazid and rifampicin), as well as any fluoroquinolone (levofloxacin, moxifloxacin, or ofloxacin) and at least one second-line injectable drug (kanamycin, amikacin, or capreomycin). Totally drug-resistant (TDR) TB is TB for which no effective treatments are available. Facing the Reality of Drug-Resistant Tuberculosis: Challenges and Potential Solutions in India: Summary of a Joint Workshop http: //www. nap. edu/catalog. php? record_id=13243

Types of Drug Resistance Acquired resistance (Resistance among previously treated cases) Drug resistance developed as a result of exposure to a single drug because of poor adherence to treatment, inappropriate prescription, irregular drug supply, and/or poor drug quality suppresses the growth of bacilli susceptible to that drug but permits the multiplication of pre-existing drug-resistant organisms Primary resistance (Resistance among new cases) Drug-resistant disease from the outset due to transmission of resistant tubercle bacilli

Epidemiology of MTB Drug Resistance Status of Global MDR-TB Crisis § An estimated 480 000 new cases of MDR-TB § An additional 100 000 people with rifampicin-resistant TB (RR-TB) who were also newly eligible for MDR-TB treatment. § India, China and the Russian Federation accounted for 45% of the combined total of 580 , 000 cases. § ~ 9. 5% of MDR-TB cases had XDR-TB in 2015. § Worldwide, only 52% of MDR-TB patients and 28% of XDR-TB are currently successfully treated. http: //www. who. int/tb/publications/global_report/gtbr 2016_executive_summ ary. pdf? ua=1

Epidemiology of MTB Drug Resistance Special Report Twenty Years of Global Surveillance of Antituberculosis-Drug Resistance Matteo Zignol, M. D. , Anna S. Dean, Ph. D. , Dennis Falzon, M. D. , Wayne van Gemert, M. P. H. , Abigail Wright, M. P. H. , Armand van Deun, M. D. , Françoise Portaels, Ph. D. , Adalbert Laszlo, Ph. D. , Marcos A. Espinal, M. D. , Ariel Pablos-Méndez, M. D. , Amy Bloom, M. D. , Mohamed A. Aziz, M. D. , Karin Weyer, D. Sc. , Ernesto Jaramillo, M. D. , Paul Nunn, M. D. , Katherine Floyd, Ph. D. , and Mario C. Raviglione, M. D. N Engl J Med Volume 375(11): 1081 -1089 September 15, 2016

Study Overview • The emergence and dissemination of drugresistant Mycobacterium tuberculosis is a global threat to health. • In this report, surveillance of drug-resistant tuberculosis during the past 20 years is described.

Key Achievements of Global Anti-TB Drug Resistance Surveillance § Wide geographic coverage of data on anti-TB drug resistance § Measurement of levels of MDR TB in 153 countries § A time-trend analysis based on data from 100 countries § Evidence of an association between MDR TB and other factors § Reliable estimates of the global burden of MDR TB (cases and deaths) § XDR TB reported from 105 countries § Increase in patients with MDR TB who were receiving second-line therapy Zignol M et al. N Engl J Med 2016; 375: 1081 -1089
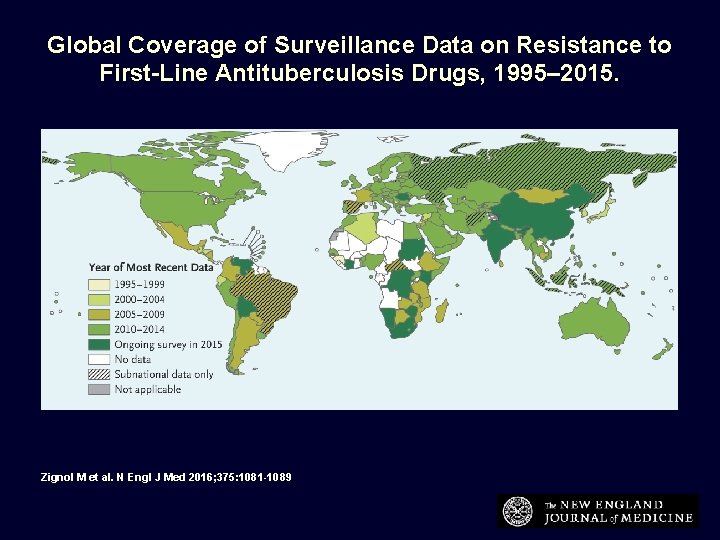
Global Coverage of Surveillance Data on Resistance to First-Line Antituberculosis Drugs, 1995– 2015. Zignol M et al. N Engl J Med 2016; 375: 1081 -1089
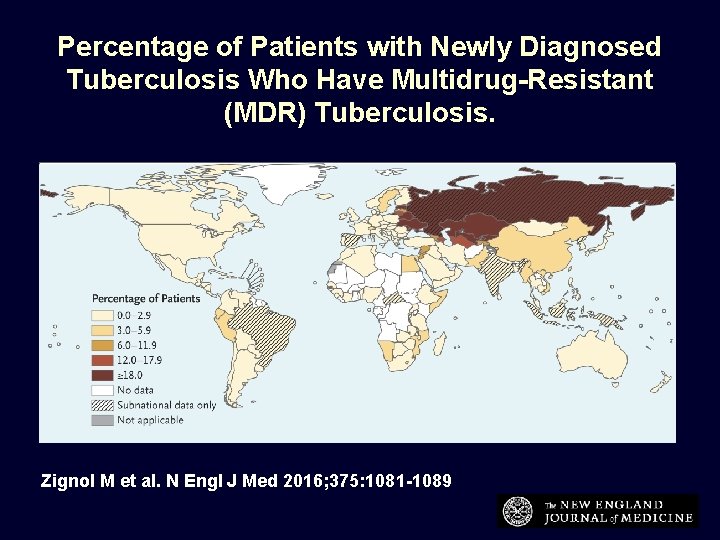
Percentage of Patients with Newly Diagnosed Tuberculosis Who Have Multidrug-Resistant (MDR) Tuberculosis. Zignol M et al. N Engl J Med 2016; 375: 1081 -1089
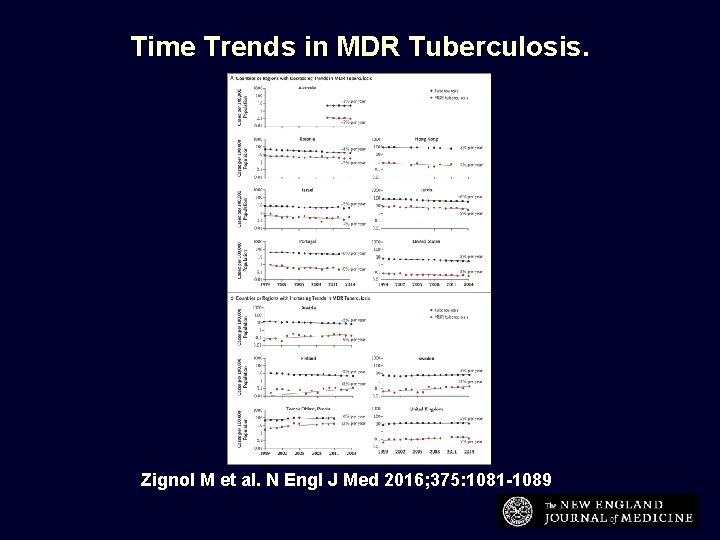
Time Trends in MDR Tuberculosis. Zignol M et al. N Engl J Med 2016; 375: 1081 -1089
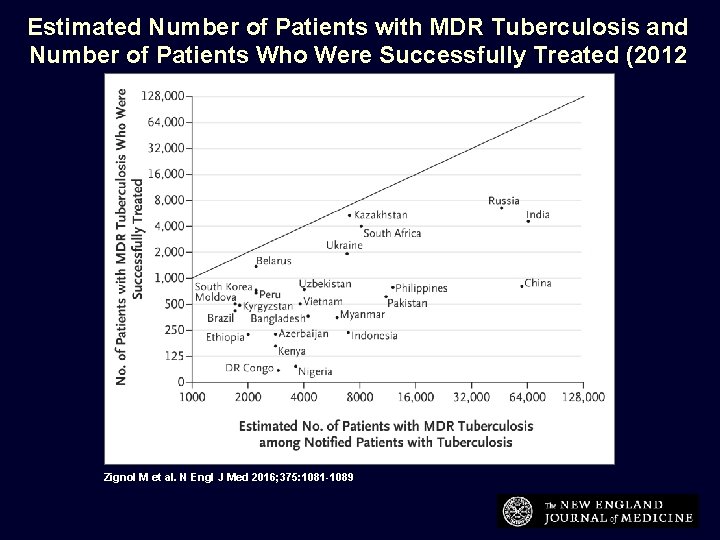
Estimated Number of Patients with MDR Tuberculosis and Number of Patients Who Were Successfully Treated (2012 Cohort). Zignol M et al. N Engl J Med 2016; 375: 1081 -1089

Evidence of an Association between MDR Tuberculosis and Other Factors § Association between MDR tuberculosis and HIV infection at the population level varies from setting to setting § A positive association has been found in Eastern European countries § a moderate association with HIV infection, particularly in patients with newly diagnosed TB showed by a recent systematic review. § on a global level, currently available data do not show a clear, consistent relationship between the levels of HIV infection and MDR TB in a population. Zignol M et al. N Engl J Med 2016; 375: 1081 -1089

Evidence of an Association between MDR Tuberculosis and Other Factors • Analysis of the levels of MDR tuberculosis in different age groups suggested – Children with TB did not have a lower risk of MDR TB than adult patients – Similar percentages of patients with MDR TB in both groups – Caution: any analysis of surveillance data on childhood tuberculosis is limited by the challenges associated with the bacteriologic diagnosis of tuberculosis and the detection of drug resistance in children. Zignol M et al. N Engl J Med 2016; 375: 1081 -1089

National Survey of Drug-Resistant Tuberculosis in China • 3037 new cases of tuberculosis and 892 previously treated cases • Among all cases: ~ 25% was resistant to isoniazid, rifampin, or both and 10% had MDR TB • Among new cases: 5. 7% (95% CI, 4. 5 to 7. 0) had MDR TB • Among previously treated cases : 25. 6% (95% CI, 21. 5 to 29. 8) had MDR TB • Approximately 8% of the patients with MDR TB had XDR TB • In 2007, there were 110, 000 incident cases (95% CI, 97, 000 to 130, 000) of MDR TB and 8200 incident cases (95% CI, 7200 to 9700) of XDR tuberculosis. • Most cases of MDR and XDR TB resulted from primary transmission. • Having multiple previous treatments with last treatment in a TB hospital had the highest risk of MDR TB (adjusted odds ratio, 13. 3; 95% CI, 3. 9 to 46. 0). • Among 226 previously treated patients with MDR TB, 43. 8% had not completed their last treatment; most had been treated in the hospital system. • Among those who had completed treatment, TB developed again in most of the patients after their treatment in the public health system. Zhao Y, et al. , NEJM 366; 23

• Genes involved in acquired drug resistance in M. tuberculosis • Gene Encoded protein Protein function Affected drug Drug’s mode of action kat. G Catalase– peroxidase Prodrug activation Isoniazid inh. A Enoyl ACP Drug target reductase ndh NADH dehydrogenase II Inhibiting mycolic acid biosynthesi s and other metabolic processes ahp. C Alkyl Oxidative hydroperoxi stress -dase resistance Modulation of NADH/NAD ratio Nguyen, L. Arch Toxicol (2016) 90: 1585. doi: 10. 1007/s 00204 -016 -17276

• Genes involved in acquired drug resistance in M. tuberculosis • rpo. B β-Subunit of RNA Drug target polymerase Rifampicin Inhibiting transcription pnc. A Pyrazinamidase Prodrug activation Pyrazinamide rsp. A S 1 ribosomal protein Drug target Inhibiting translation emb. CAB Arabinosyltransferases Drug target Ethambutol Inhibiting arabinogalact an synthesis emb. R emb. CAB transcri ption regulator Regulation of emb. CAB expression Nguyen, L. Arch Toxicol (2016) 90: 1585. doi: 10. 1007/s 00204 -016 -1727 -6

• Genes involved in acquired drug resistance in M. tuberculosis • rps. L S 12 ribosomal protein Drug target rrs 16 S r. RNA Drug target gid. B 16 S r. RNA methyltransferase Modification of drug target whi. B 7 MDR Regulation transcription of drug regulator resistance genes Streptomycin Inhibiting protein synthesis Nguyen, L. Arch Toxicol (2016) 90: 1585. doi: 10. 1007/s 00204 -016 -1727 -6

• Genes involved in acquired drug resistance in M. tuberculosis rrs 16 S ribosomal RNA Drug target eis Aminoglycoside acetyltransferase Drug inactivation whi. B 7 MDR transcription regulator Amikacin/k anamycin Inhibiting protein synthesis Regulation of eis expression • Nguyen, L. Arch Toxicol (2016) 90: 1585. doi: 10. 1007/s 00204 -016 -1727 -6

• Genes involved in acquired drug resistance in M. tuberculosis eth. A Flavin monooxygenase Prodrug activation eth. R eth. A transcription repressor Regulation of eth. Aexpre ssion inh. A Enoyl ACP reductase Drug target ndh NADH dehydrogenase Modulation II of NADH/NAD ratio • msh. A Glycosyltransferase Ethionamide Inhibiting mycolate biosynthesis Prodrug activation Nguyen, L. Arch Toxicol (2016) 90: 1585. doi: 10. 1007/s 00204 -016 -1727 -6

• Genes involved in acquired drug resistance in M. tuberculosis gyr. A DNA gyrase Drug target subunit A gyr. B DNA gyrase Drug subunit B binding alr. A D-Alanine Drug target racemase cyc. A D-Alanine-D Drug target -alanine ligase ddl Fluoroquinolones Inhibiting DNA gyrase D-Cycloserine Inhibiting peptidogly can synthesis Amino acid Drug uptake transporter • Nguyen, L. Arch Toxicol (2016) 90: 1585. doi: 10. 1007/s 00204 -016 -1727 -6

• Genes involved in acquired drug resistance in M. tuberculosis • thy. A Thymidylate d. TTP synthase A synthesis dfr. A Dihydrofolat e reductase Drug target fol. C Dihydrofolat e synthase Prodrug activation rib. D Dihydrofolat e reductase analog Replacemen t of drug target activity p. Inhibiting Aminosalicyl folate ic acid (PAS) biosynthesis Nguyen, L. Arch Toxicol (2016) 90: 1585. doi: 10. 1007/s 00204 -016 -1727 -6

• Genes involved in acquired drug resistance in M. tuberculosis tly. A r. RNA methyltransf erase Ribosome protection rrs 16 S ribosomal RNA Drug target Capreomycin Inhibiting protein synthesis • Nguyen, L. Arch Toxicol (2016) 90: 1585. doi: 10. 1007/s 00204 -016 -1727 -6

Targeting Persisters for Tuberculosis Control Ying Zhang, a Wing Wai Yew, b and Michael R. Barerc Antimicrob Agents Chemosphere. 2012 May; 56(5): 2223– 2230 Mycobacterial persisters, the survivors from antibiotic exposure, necessitate the lengthy treatment of tuberculosis (TB) and pose a significant challenge for our control of the disease. We suggest that persisters in TB are heterogeneous in nature and comprise various proportions of the population depending on the circumstances; the mechanisms of their formation are complex and may be related to those required for persistence in chronic infection. Results from recent studies implicate multiple pathways for persister formation, including energy production, the stringent response, global regulators, the translation pathway, proteasomal protein degradation, toxin-antitoxin modules, and transporter or efflux mechanisms. A combination of specifically persistertargeted approaches, such as catching them when active and susceptible either by stimulating them to “wake up” or by intermittent drug dosing, the development of new drugs, the use of appropriate drug combinations, and combined chemotherapy and immunotherapy, may be needed for more effective elimination of persisters and better treatment of TB. Variations in levels of persister formation and in host genetics can play a role in the outcome of clinical treatment, and thus, these may entail personalized treatment regimens.

Yin and yang of persisters and replicating bacteria and their interconversions. Ying Zhang , et al. , Antimicrob Agents Chemosphere. 2012 May; 56(5): 2223– 2230

Yin and yang of latent infection and overt disease and their interconversions. Ying Zhang , et al. , Antimicrob Agents Chemosphere. 2012 May; 56(5): 2223– 2230

Drug-Susceptibility Testing § Conduct drug-susceptibility testing on initial M. tb isolate § Promptly forward results to the health department § Repeat for patients who § Do not respond to therapy or § Have positive cultures despite 3 months of therapy CDC. Core Curriculum on Tuberculosis: What the Clinician Should Know. Sixth Edition 2013

Drug-Susceptibility Testing CDC. Core Curriculum on Tuberculosis: What the Clinician Should Know. Sixth Edition 2013

Second-line Drug-Susceptibility Testing § Limit to persons at increased risk for drug resistance: § Have history of treatment with TB drugs § Had contact with a person with drug-resistant TB § Demonstrated resistance to first-line drugs § Has positive smears or cultures despite 3 months of TB treatment CDC. Core Curriculum on Tuberculosis: What the Clinician Should Know. Sixth Edition 2013

Molecular Detection of Drug Resistance § Drug resistance is caused by mutations in specific M. tb genes. § Several molecular assays and tests can detect mutations § Molecular detection should be used for patients with high risk for rifampin resistance (MDR TB). § Conventional drug susceptibility testing should be done in conjunction with molecular tests. CDC. Core Curriculum on Tuberculosis: What the Clinician Should Know. Sixth Edition 2013

Whole genome sequencing of Mycobacterium tuberculosis for detection of drug resistance: a systematic review. § § Papaventsis D et al. , Clin Microbiol Infect. 2016 Sep 22 A systematic review of 20 publications to determine the diagnostic accuracy of whole genome sequencing (WGS) of MTB for the detection of resistance to first- and second-line anti-TB drugs. Included all first-line drugs, a variety of reserve drugs, as well as new drugs. Polymorphisms in a total of 53 genes were tested for associations with drug resistance. Pooled sensitivity and specificity values for detection of resistance to selected first-line drugs were 0. 98 (95% CI 0. 93 -0. 98) and 0. 98 (95% CI 0. 98 -1. 00) for rifampicin and 0. 97 (95% CI 0. 94 -0. 99) and 0. 93 (95% CI 0. 91 -0. 96) for isoniazid, respectively. Due to high heterogeneity in study designs, lack of data, knowledge of resistance mechanisms and clarity on exclusion of phylogenetic markers, there was a significant variation in analytical performance of WGS for the remaining first-line, reserved drugs and new drugs.

Whole genome sequencing of Mycobacterium tuberculosis for detection of drug resistance: a systematic review. Papaventsis D et al. , Clin Microbiol Infect. 2016 Sep 22 Conclusions: § Whole genome sequencing could be considered a promising alternative to existing phenotypic and molecular drugsusceptibility testing methods for rifampicin and isoniazid pending standardization of analytical pipelines. § To ensure clinical relevance of WGS for detection of M. tuberculosis complex drug resistance, future studies should include information on clinical outcomes. ion:

Discussion 1. What research questions need to be addressed to develop more effective strategies for the control of drug resistant TB epidemics? 2. What approaches would you use to address the identified questions?
- Slides: 34