Tropical protozoan infection Amebiasis Protozoa in Greek protofirst
Tropical protozoan infection. Amebiasis.

Protozoa (in Greek proto=first and zoa=animals) are onecelled eukaryotes(that is, unicellular microbes whose cells have membrane-bound nuclei) that commonly show characteristics usually associated with animals, mobility and heterotrophy. Genes • • Flagellates (or Mastigophora), Amoebae (or Sarcodina), Sporozoans (or Sporozoa), Ciliates (or Ciliophora)

General characteristics Most protozoa are too small to be seen with the naked eye, but can easily be found under a microscope Protozoa are ubiquitous throughout aqueous environments and the soil, and play an important role in their ecology. Protozoa occupy a range of trophic levels. As predators upon unicellular or filamentous algae, bacteria, and microfungi, protozoa play a role both as herbivores and as consumers in the decomposer link of the food chain. Protozoa also play a vital role in controlling bacteria population and biomass. • Some protozoa have the ability to form a cyst to protect it from harsh conditions, allowing it to survive exposure to extreme temperatures or harmful chemicals or without food, water, or oxygen for a period of time.
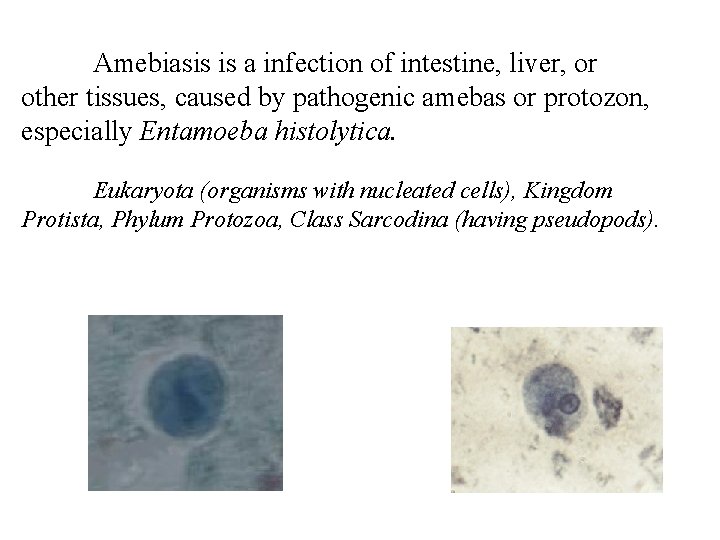
Amebiasis is a infection of intestine, liver, or other tissues, caused by pathogenic amebas or protozon, especially Entamoeba histolytica. Eukaryota (organisms with nucleated cells), Kingdom Protista, Phylum Protozoa, Class Sarcodina (having pseudopods).
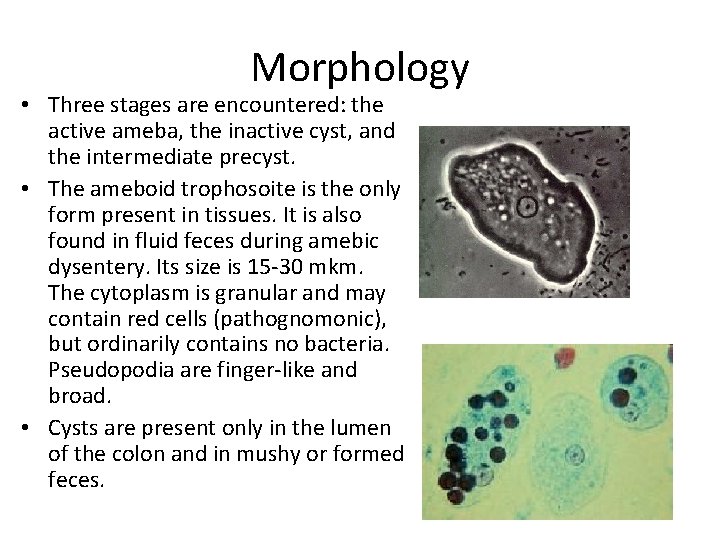
Morphology • Three stages are encountered: the active ameba, the inactive cyst, and the intermediate precyst. • The ameboid trophosoite is the only form present in tissues. It is also found in fluid feces during amebic dysentery. Its size is 15 -30 mkm. The cytoplasm is granular and may contain red cells (pathognomonic), but ordinarily contains no bacteria. Pseudopodia are finger-like and broad. • Cysts are present only in the lumen of the colon and in mushy or formed feces.
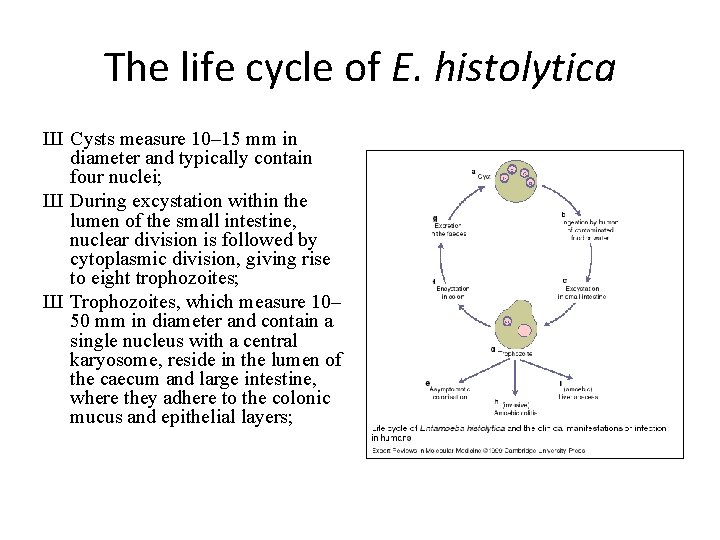
The life cycle of E. histolytica Ш Cysts measure 10– 15 mm in diameter and typically contain four nuclei; Ш During excystation within the lumen of the small intestine, nuclear division is followed by cytoplasmic division, giving rise to eight trophozoites; Ш Trophozoites, which measure 10– 50 mm in diameter and contain a single nucleus with a central karyosome, reside in the lumen of the caecum and large intestine, where they adhere to the colonic mucus and epithelial layers;
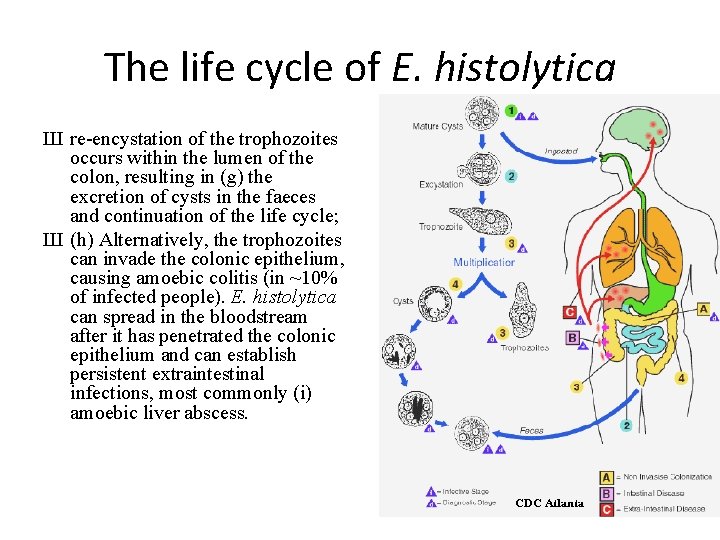
The life cycle of E. histolytica Ш re-encystation of the trophozoites occurs within the lumen of the colon, resulting in (g) the excretion of cysts in the faeces and continuation of the life cycle; Ш (h) Alternatively, the trophozoites can invade the colonic epithelium, causing amoebic colitis (in ~10% of infected people). E. histolytica can spread in the bloodstream after it has penetrated the colonic epithelium and can establish persistent extraintestinal infections, most commonly (i) amoebic liver abscess.

Epidemiology • The cyst, the infective form of the organism, resists environmental changes and may spread either directly from person to person or indirectly via food or water. • Direct spread occurs when personal hygiene is compromised (eg, among sexual partners, particularly male homosexuals and institutionalized, mentally retarded individuals). • Indirect spread occurs more frequently in areas where sanitation is poor. Fruits and vegetables may be contaminated when grown in soil fertilized by human feces, washed in polluted water, or prepared by an asymptomatic cyst passer. Water-borne outbreaks associated with faulty plumbing have been described; more commonly, amebiasis is sporadic.

Risk factors for amebiasis include • • • malnutrition, old age, pregnancy, use of steroids, malignancy (cancer), and alcoholism. Recent travel to a tropical region.
Pathogenesis • Excystation of ingested cysts occurs in the small intestine. Released trophozoites travel to the colon to grow and multiply in the bowel lumen. Changes in the virulence of these organisms or the host's resistance may lead to extensive tissue damage and disease. • The trophozoites most frequently penetrate the mucous membrane in regions of fecal stasis: cecum, appendix, ascending colon, sigmoid colon, and rectum.

Pathogenesis • The earliest lesion is a small abscess, usually in the submucosa; later, ulcers form that tend to be ragged and undermined. • The lesions are focal and discrete in mild cases but may spread and become confluent, with hemorrhage, edema, and sloughing of large areas of mucosa. Although amebic penetration is limited by the muscular coat, perforation occasionally results; • Amebas enter the radicles of the portal vein and thus reach the liver. Most amebas are probably destroyed, but one or more large hepatic abscesses develop if the survivors are numerous and multiply. • The disease usually spreads further by direct extension from the liver into the pleura, right lung, and pericardium

Clinical Symptoms • Incubation: The incubation period for amebic dysentery is usually 1 - 4 weeks, but may be shorter or substantially longer. • There are two basic types of amebiasis: intestinal and extraintestinal disease, which may exist simultaneously.

Intestinal amebiasis: • fever, • gradual onset of colicky abdominal pain, • increased number of stools (often intermittent, > 6 stools/day, usually containing mucous and blood), • jaundice, • anorexia, • weight loss, • tenesmus. • • More extraintestinal amebiasis is hepatic amebiasis (abscess). fever, right upper quadrant pain (sometimes radiating to the right shoulder), hepatomegaly and tenderness, nausea and vomiting, anorexia, weight loss, and malaise.
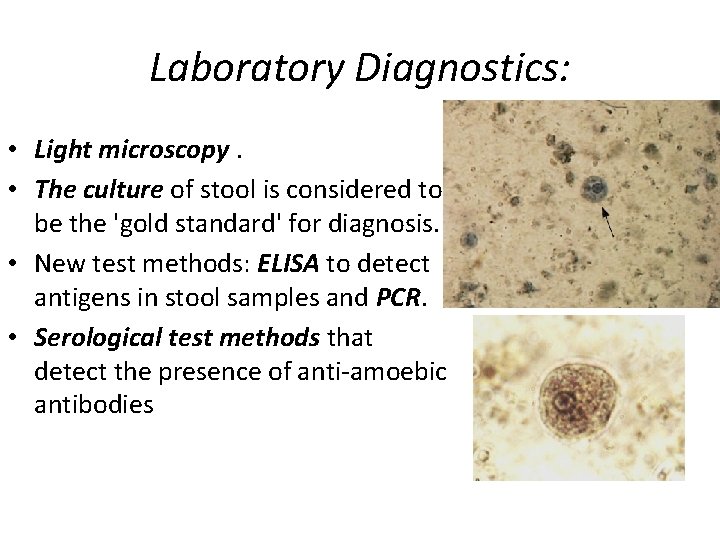
Laboratory Diagnostics: • Light microscopy. • The culture of stool is considered to be the 'gold standard' for diagnosis. • New test methods: ELISA to detect antigens in stool samples and PCR. • Serological test methods that detect the presence of anti-amoebic antibodies

Treatment • Tissue amebicides (dehydroemetine, and metronidazole(Flagyl) ) act on amebas in the bowel wall but not the lumen. • Luminal amebicides (diloxanide furoate, idoquinol, and paromomycin) act on amebas in the bowel lumen. • Tetracycline inhibits bacterial growth in both the bowel wall and lumen. • Metronidazole effects both the bowel wall and lumen but if given alone has a 50% fail rate as it needs a luminal amebicide to augment its action. • Asymptomatic patients are treated with iodoquinol (Yodoxin); or diloxanide furoate (Furamide); or paromomycin (Humatin).

Prevention • Improved sanitation and clean water supply decrease incidence of amebiasis. The amount of chlorine normally used to purify water is inadequate in killing the cysts. Drinking water can be rendered safe by boiling, 0. 22 µm filtration, or iodination with tetraglycine hydroperiodide. Bottled water may be used for drinking when traveling to endemic areas. • Eating only cooked food or self-peeled fruits in endemic areas minimizes risk. Avoid eating raw fruits and salads, which are difficult to sterilize. • Disease transmission can be reduced by early treatment of carriers in nonendemic areas. • Vaccination for amebiasis currently is being investigated. One potential vaccine candidate is the galactose-inhibitable adherence lectin of E histolytica.
MALARIA
Malaria • A protozoan infection characterized by paroxysms of chills, fever, and sweating, and by anemia, splenomegaly, and a chronic relapsing course.
Etiology • Eukaryota (organisms with nucleated cells), Kingdom Protista, Phylum Protozoa, Class Sporozoa. The species • Plasmodium falciparum (is the most dangerous), • P vivax, • P malariae, • P ovale.
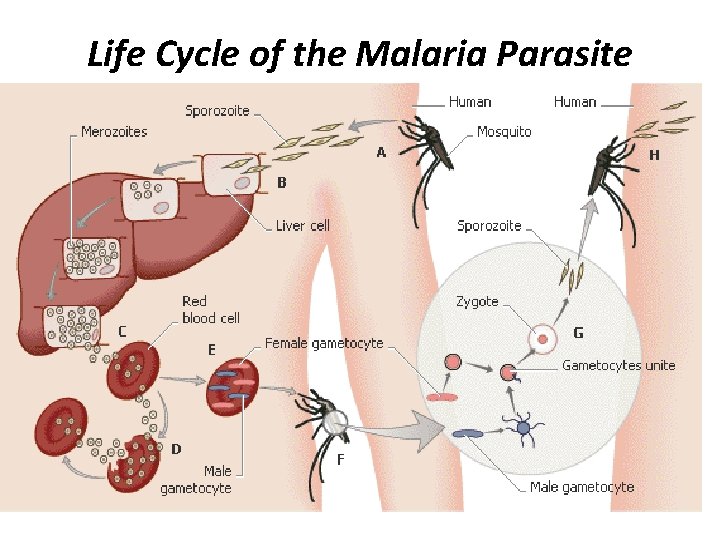
Life Cycle of the Malaria Parasite
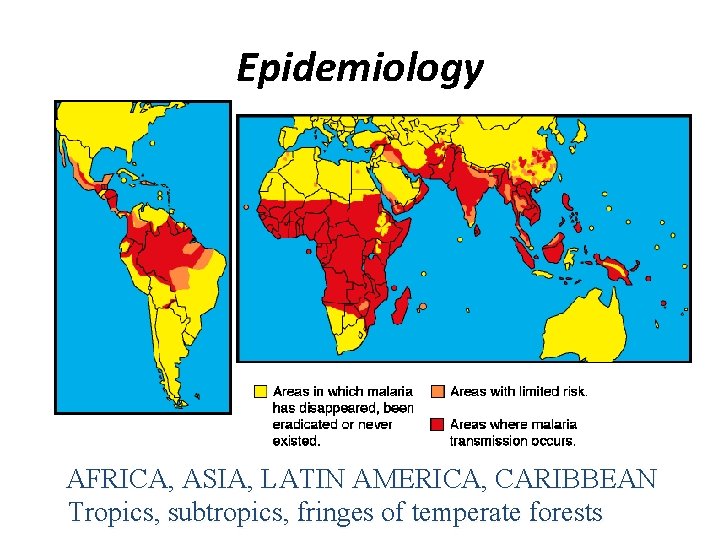
Epidemiology AFRICA, ASIA, LATIN AMERICA, CARIBBEAN Tropics, subtropics, fringes of temperate forests

Transmission • Man is the only important reservoir • Vector is female Anopheles mosquito ® Transmission also possible through: 1. Blood transfusion 2. Contaminated needle 3. Organ transplant 4. Congenital

Pathogenesis • The fever and chills of malaria are associated with the rupture of erythrocyticstage schizonts. • In severe falciparum malaria, parasitized red cells may obstruct capillaries and postcapillary venules, leading to local hypoxia and the release of toxic cellular products. • Obstruction of the microcirculation in the brain (cerebral malaria) and in other vital organs is thought to be responsible for severe complications

The incubation period is usually 10 to 35 days. • Incubation period (the time from the initial malaria infection until symptoms appear) generally ranges from: • 9 to 14 days for P. falciparum • 12 to 18 days for P. vivax and P. ovale • 18 to 40 days for P. malariae

Acute Symptoms • Classical cyclic paroxysm: – Cold stage: chills and shaking – Hot stage: warm, headache, vomiting – Sweating stage: weakness – Feel well for period of time, then cycle repeats itself
• The three stages are often repeated the following day, two days later, or at some later time. In many cases, a person experiences a repetition of the stages again and again during their lifetime. Some people go many years before the symptoms repeat.
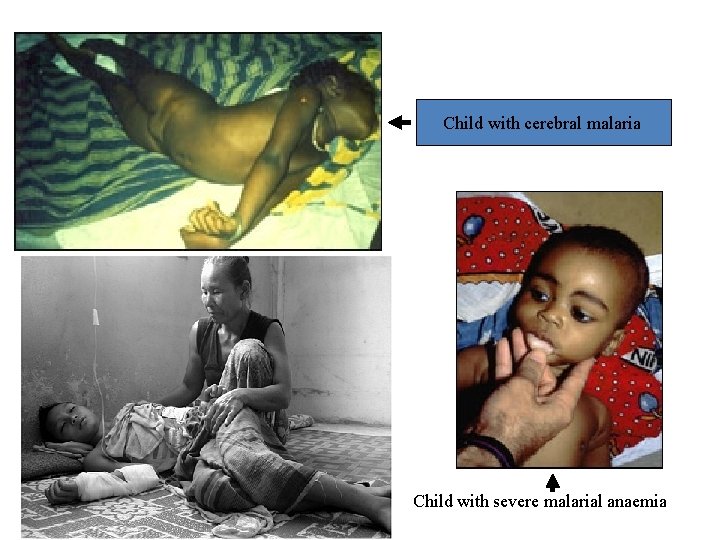
Child with cerebral malaria Child with severe malarial anaemia
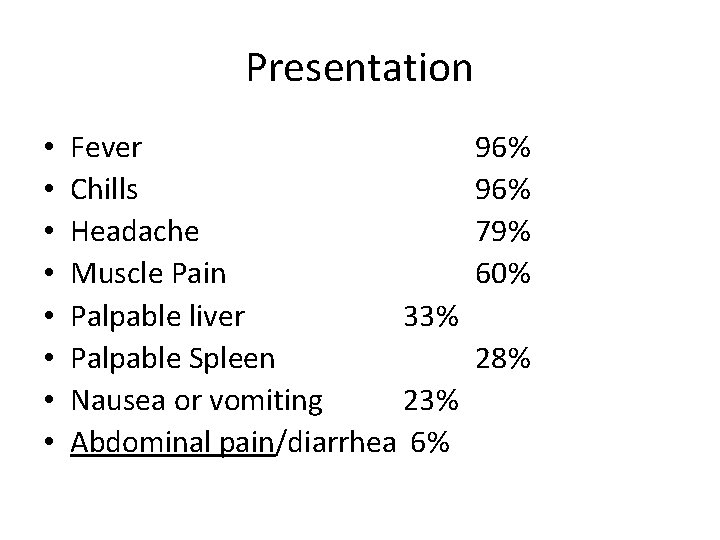
Presentation • • Fever Chills Headache Muscle Pain Palpable liver 33% Palpable Spleen Nausea or vomiting 23% Abdominal pain/diarrhea 6% 96% 79% 60% 28%

DIAGNOSIS 1. microscopic examination of the blood Giemsa-stained thick and thin peripheral blood smears 2. CBC (complete blood count) – anemia 3. Polymerase chain reaction
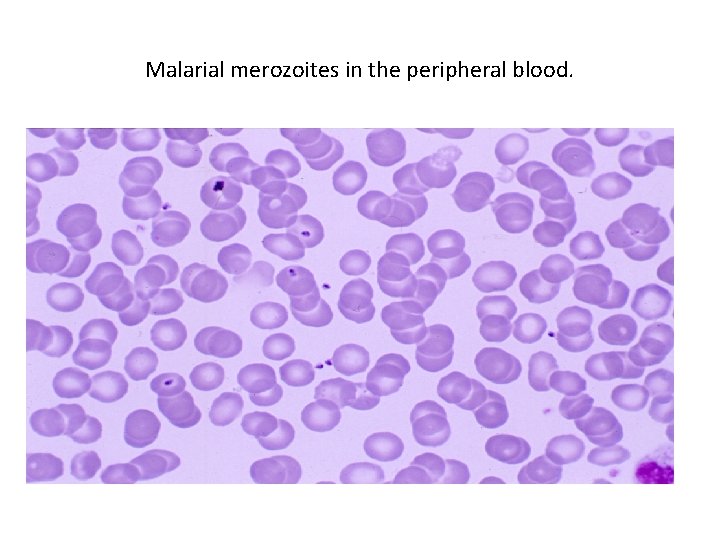
Malarial merozoites in the peripheral blood.
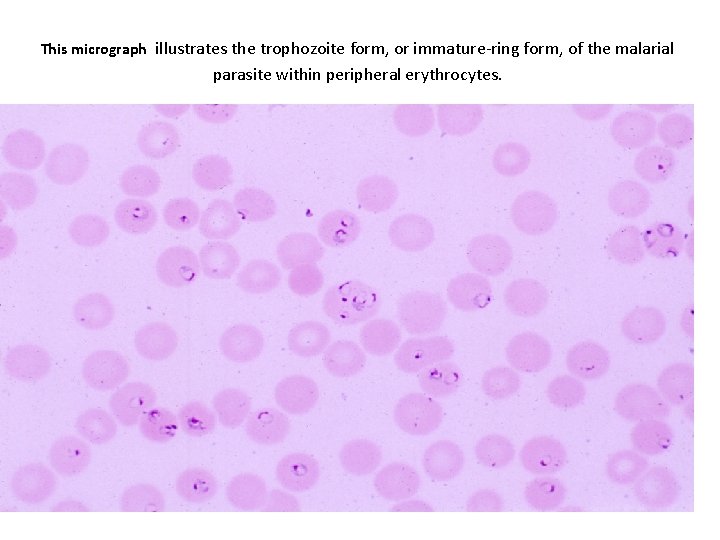
This micrograph illustrates the trophozoite form, or immature-ring form, of the malarial parasite within peripheral erythrocytes.
Treatment • CHLOROQUINE • Quinine and DOXYCYCLINE • MEFLOQUINE • PRIMAQUINE

Points of Attack 1. Attack the parasite in the human host 2. Reduce contact between humans and mosquitoes 3. Decrease mosquito population

Attack The Parasite In The Human Host • Treat malaria infections with effective medications • Use prophylactic drugs to prevent illness and/or infection

Attack The Parasite In The Human Host • Chemoprophylaxis is based on current drug resistance patterns • MEFLOQUINE first line prophylaxis – Mefloquine 250 mg po q week, 1 -2 wks prior to 4 wks after • DOXYCYCLINE as second line drug – Doxy 100 mg po qd, 2 days prior to 4 wks after • PRIMAQUINE – 30 mg* po qd x 14 days terminal prophylaxis *15 mg per FDA and drug product information insert

Reduce Contact Between Humans And Mosquitoes • Personal protective measures – Proper wearing of uniform – PERMETHRIN – Bed nets

Decrease Mosquito Population • Surveillance of mosquito populations • Identify and eliminate breeding sites • Proper insecticide application – Attack larval stages – Attack adult mosquito
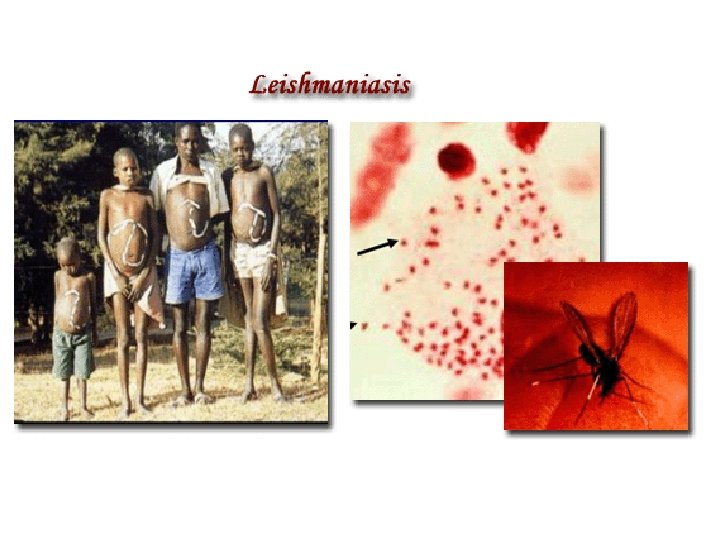

Leishmaniasis is a parasitic disease caused by the protozoa belonging to the genus, Leishmania. Human leishmaniasis is not a disease, but a group of diseases. While several ways to classify leishmaniasis (eg, by geography or taxonomy) are available, clinically, it can present itself in various ways, and is more easily classified as cutaneous, mucocutaneous, and visceral leishmaniasis.
Classification • • Eukaryota (organisms with nucleated cells), Kingdom Protista, Phylum Protozoa, Class Flagellates, Genus Leishmania.
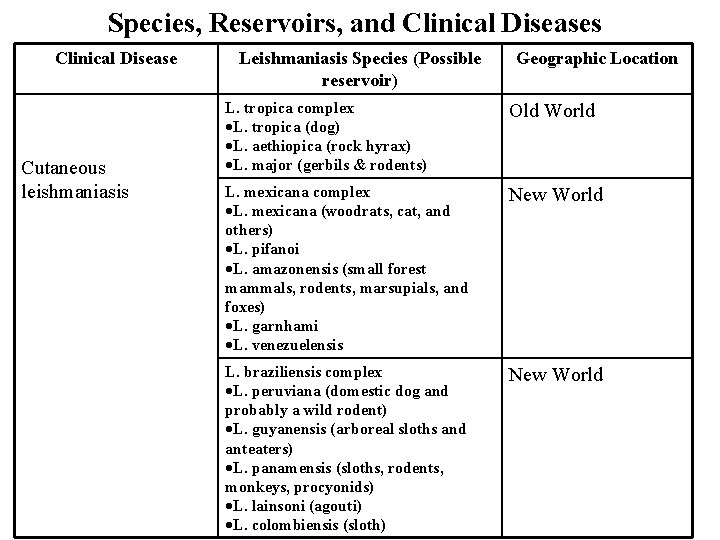
Species, Reservoirs, and Clinical Diseases Clinical Disease Cutaneous leishmaniasis Leishmaniasis Species (Possible reservoir) Geographic Location L. tropica complex L. tropica (dog) L. aethiopica (rock hyrax) L. major (gerbils & rodents) Old World L. mexicana complex L. mexicana (woodrats, cat, and others) L. pifanoi L. amazonensis (small forest mammals, rodents, marsupials, and foxes) L. garnhami L. venezuelensis New World L. braziliensis complex L. peruviana (domestic dog and probably a wild rodent) L. guyanensis (arboreal sloths and anteaters) L. panamensis (sloths, rodents, monkeys, procyonids) L. lainsoni (agouti) L. colombiensis (sloth) New World

Species, Reservoirs, and Clinical Diseases Cutaneous leishmaniasis Mucocutaneous leishmaniasis L. infantum Old World L. chagasi New World L. braziliensis complex L. braziliensis L. guyanensis L. panamensis New World L. mexicana New World L. tropica Old World L. major Old World

Species, Reservoirs, and Clinical Diseases Visceral leishmaniasis L. donovani complex L. donovani (no reservoir in Indian or Kenyan area, various rodents in Sudan , dogs in China ) L. infantum (human is accidental host, natural infection in dogs, other Canidae, and porcupines) Old World L. chagasi (domestic dogs and cats, foxes) New World L. tropica Old World L. amazonensis New World Old World
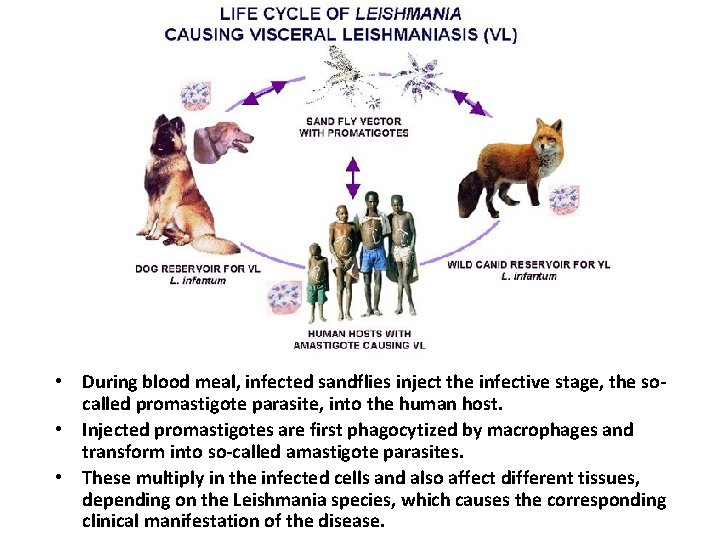
• During blood meal, infected sandflies inject the infective stage, the socalled promastigote parasite, into the human host. • Injected promastigotes are first phagocytized by macrophages and transform into so-called amastigote parasites. • These multiply in the infected cells and also affect different tissues, depending on the Leishmania species, which causes the corresponding clinical manifestation of the disease.
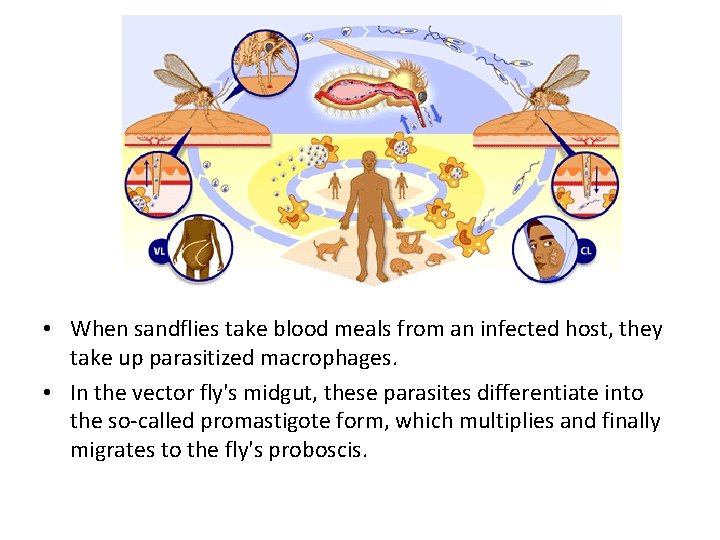
• When sandflies take blood meals from an infected host, they take up parasitized macrophages. • In the vector fly's midgut, these parasites differentiate into the so-called promastigote form, which multiplies and finally migrates to the fly's proboscis.
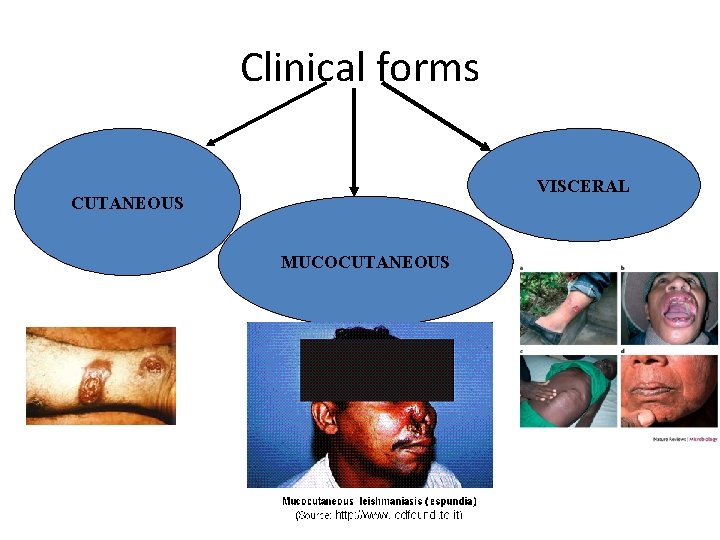
Clinical forms VISCERAL CUTANEOUS MUCOCUTANEOUS
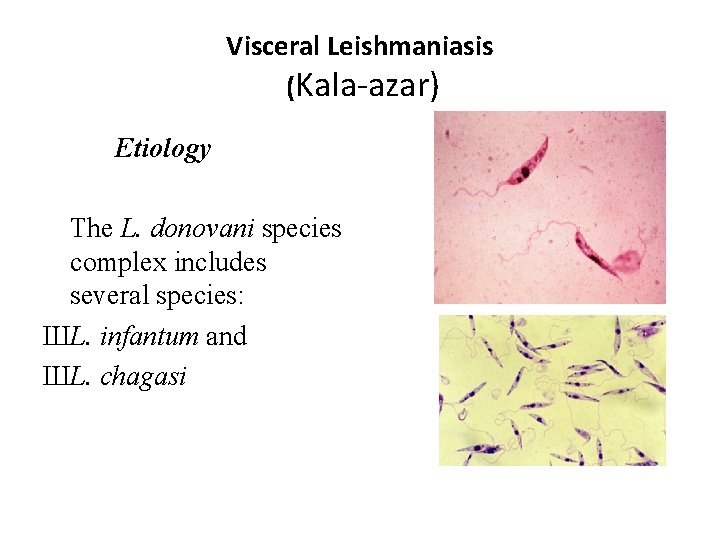
Visceral Leishmaniasis (Kala-azar) Etiology The L. donovani species complex includes several species: ШL. infantum and ШL. chagasi

Epidemiology • The species of visceral leishmaniasis are endemic in areas of India, China, Central and South America, East and West Africa, and the countries surrounding the Mediterranean. • In India, no extrahuman reservoirs are known, but in other regions, infection may involve several mammalian species, including dogs, foxes, and wild rodents. • Sandflies of the genus Phlebotomus are the insect vectors that spread L. donovani.

Pathogenesis • The flagellated promastigotes of L. donovani are introduced by an insect bite. • After entering macrophages of the reticuloendothelial system, these forms change into amastigotes, which multiply in phagocytic cells. • Released amastigotes disseminate hematogenously and invade reticuloendothelial cells in the spleen, liver, lymph nodes, bone marrow, and skin.
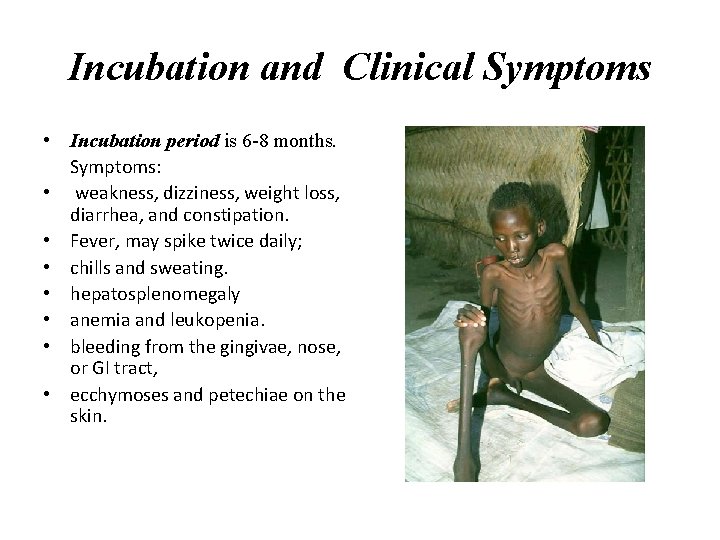
Incubation and Clinical Symptoms • Incubation period is 6 -8 months. Symptoms: • weakness, dizziness, weight loss, diarrhea, and constipation. • Fever, may spike twice daily; • chills and sweating. • hepatosplenomegaly • anemia and leukopenia. • bleeding from the gingivae, nose, or GI tract, • ecchymoses and petechiae on the skin.
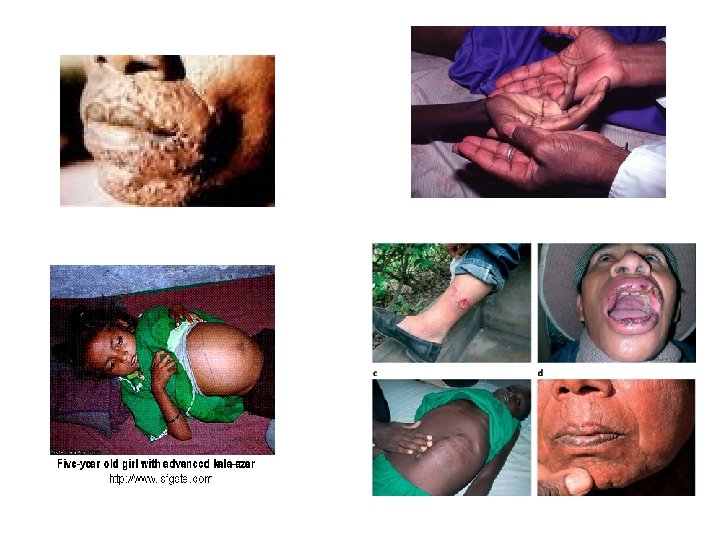

Cutaneous and Mucocutaneous Leishmaniasis Etiology and Epidemiology Old World cutaneous leishmaniasis is caused by three species of Leishmania that belong to the L. tropica complex: ь L. tropica is present in the Middle East and the Mediterranean littoral; ь L. major is found in the Middle East, Arabia, India, and sub-Saharan Africa; ь L. aethiopica is found principally in Ethiopia and Kenya. Phlebotomus sandflies are the principal vectors. • Infections that are caused by Leishmania can be acquired by travelers, as well as by military and other personnel residing in endemic areas. • Military personnel in the Middle East have acquired cutaneous leishmaniasis with L. major and viscerotropic infections with L. tropica.

Cutaneous and Mucocutaneous Leishmaniasis • New World cutaneous leishmaniasis arises from infection with parasites belonging to the L. mexicana group or the L. braziliensis (Viannia subgenus) group. • The patterns of illness vary with the nature of the infecting leishmanial organisms, which are found in different regions of North, Central, and South America. • Infections with strains of L. viannia, which are endemic in various areas of South America, cause cutaneous leishmaniasis and, in a small percentage of those infected, result in the later development of mucocutaneous leishmaniasis. Such mucocutaneous disease (espundia) involves the nasal or oropharyngeal mucosa, or both, and may prove fatal. • All of these New World leishmanial parasites are transmitted principally by sandfly vectors, although direct human contact may also bring about infection. • Various mammals are naturally infected reservoirs of the organisms.

Pathogenesis • Both Old World and New World forms of leishmaniasis are initiated when the bite of an infected sandfly injects promastigotes into the human host. • The organisms enter tissue macrophages and capillary endothelial cells, become amastigotes, and multiply. • A granulomatous inflammatory response develops at the bite site. • With local ischemia, the lesion ulcerates; a bacterial infection of the necrotic area may extend the ulceration.
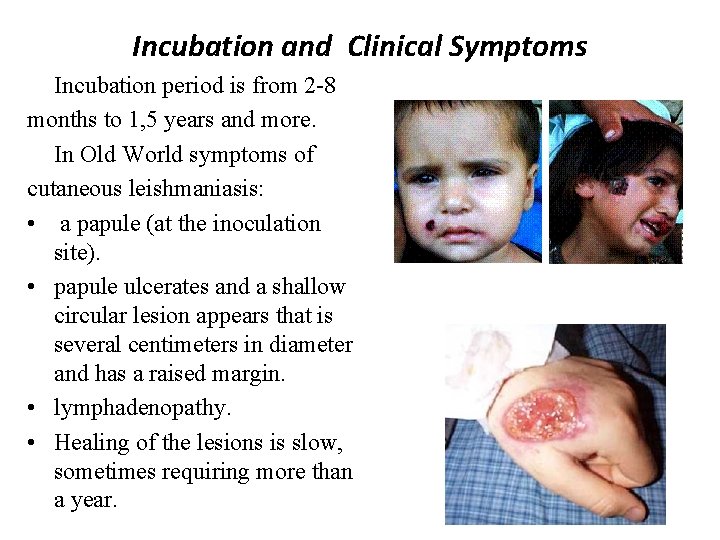
Incubation and Clinical Symptoms Incubation period is from 2 -8 months to 1, 5 years and more. In Old World symptoms of cutaneous leishmaniasis: • a papule (at the inoculation site). • papule ulcerates and a shallow circular lesion appears that is several centimeters in diameter and has a raised margin. • lymphadenopathy. • Healing of the lesions is slow, sometimes requiring more than a year.
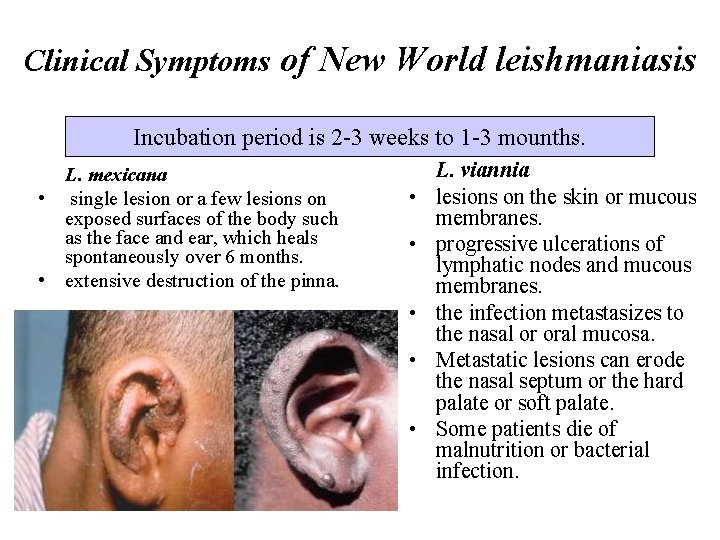
Clinical Symptoms of New World leishmaniasis Incubation period is 2 -3 weeks to 1 -3 mounths. L. mexicana • single lesion or a few lesions on exposed surfaces of the body such as the face and ear, which heals spontaneously over 6 months. • extensive destruction of the pinna. • • • L. viannia lesions on the skin or mucous membranes. progressive ulcerations of lymphatic nodes and mucous membranes. the infection metastasizes to the nasal or oral mucosa. Metastatic lesions can erode the nasal septum or the hard palate or soft palate. Some patients die of malnutrition or bacterial infection.
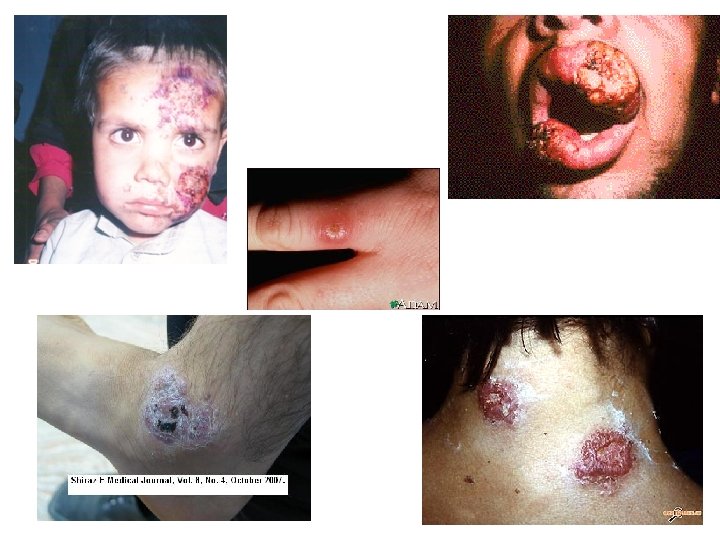
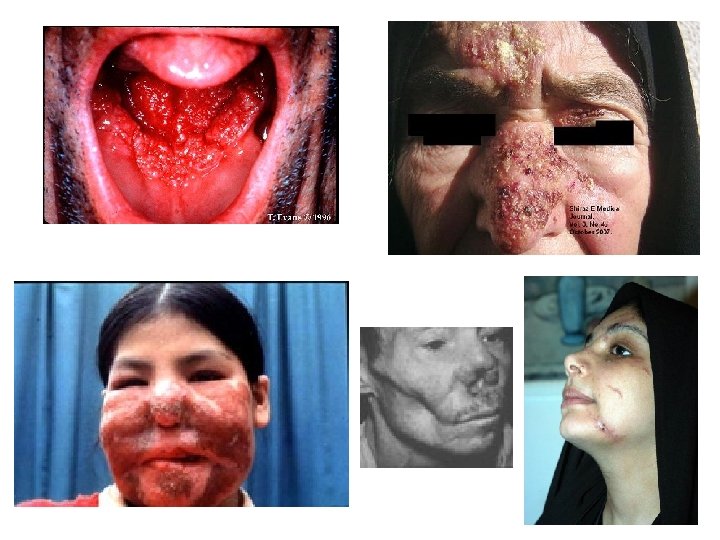

Immunity • In visceral leishmaniasis (Kala-Azar) cellular immunity is responsible for resolving mild disease. High levels of antibodies are found. • In cutaneous and mucocutaneous leishmaniasis host defense relies on cell-mediated immunity; antibody titers are low. The response ranges from a local granuloma with few parasites to a histiocytoma with many parasites.

Laboratory Diagnostics of visceral leishmaniasis • Demonstration of the organism in host tissues cultured on a Novy-Mac. Neal-Nicolle (NNN) or other medium or detection of Leishman-Donovan bodies (amastigotes) in stained tissue samples. • PCR can be performed using genus- or species-specific oligonucleotides. • Established by examining bone marrow aspirates. • Splenic aspirates have the highest yields but may be risky. • Liver biopsy or aspiration of enlarged lymph nodes can also provide diagnostic material.
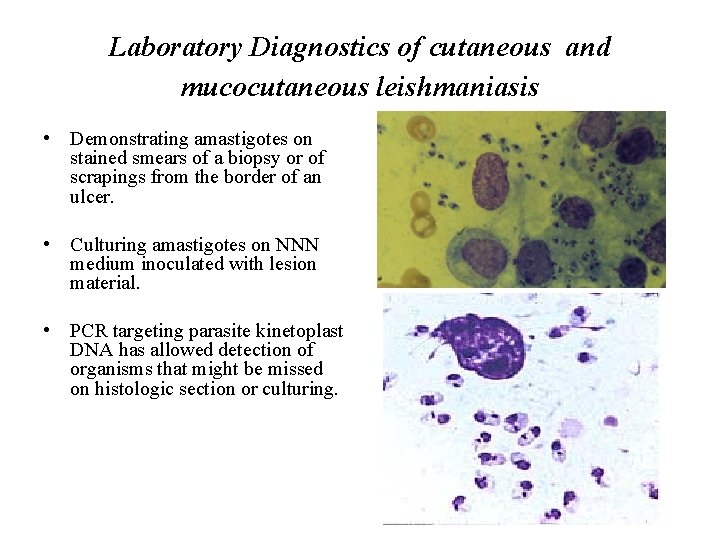
Laboratory Diagnostics of cutaneous and mucocutaneous leishmaniasis • Demonstrating amastigotes on stained smears of a biopsy or of scrapings from the border of an ulcer. • Culturing amastigotes on NNN medium inoculated with lesion material. • PCR targeting parasite kinetoplast DNA has allowed detection of organisms that might be missed on histologic section or culturing.

Laboratory Diagnostics of cutaneous and mucocutaneous leishmaniasis • Except in diffuse cutaneous leishmaniasis, the leishmanin skin test is usually positive.

Treatment • There are two common therapies containing antimony, meglumine antimoniate (Glucantim®) and sodium stibogluconate (Pentostam®). Unfortunately, in many parts of the world, the parasite has become resistant to antimony and for visceral or mucocutaneous leishmaniasis, amphotericin is now the treatment of choice. • Miltefosine (Impavido®), is a new drug for visceral, mucocutaneous and cutaneous leishmaniasis. • Drug-resistant leishmaniasis may respond to immunotherapy (inoculation with parasite antigens plus an adjuvant) which aims to stimulate the body's own immune system to kill the parasite.

Prevention: • Preventing sandfly bites is the most immediate form of protection. Insect repellent, appropriate clothing, screening of windows, and fine mesh netting around the bed (in endemic areas) will reduce exposure. • Public health measures to reduce the sandfly population and animal reservoirs are important. There are no preventive vaccines or drugs for leishmaniasis.
- Slides: 65