Tribology Friction and Wear of Silicon Nitride Exposed

Tribology Friction and Wear of Silicon Nitride Exposed to Moisture at High Temperatures

Introduction • What’s the purpose of this study? We know that. . . • Si 3 N 4 + 3 O 2 = 3 Si. O 2 + 2 N 2 • Si. O 2 interacts with water • The goal is to determine the effects of water on Silicon Nitride -For coefficient of friction and wear rate

Purpose • Why is this Relevant? Applications… • Silicon nitride automobile applications exposed to water vapor • Bearing/components of gas turbine engines • Ceramic coating on metallic components

Experimental Procedure • Used sliding ball-on-flat apparatus in different environments containing water vapor at elevated temperature • Silicon nitride flats and isostatically pressed balls • 10, 000 strokes (equivalent to 218 meters sliding distance) • Environments include: Argon, Air, 2% H 20, 8% H 20, 34% H 20
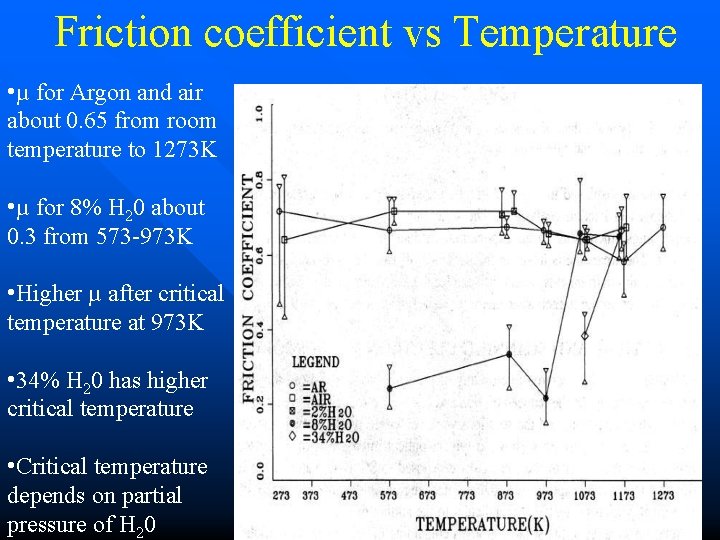
Friction coefficient vs Temperature • µ for Argon and air about 0. 65 from room temperature to 1273 K • µ for 8% H 20 about 0. 3 from 573 -973 K • Higher µ after critical temperature at 973 K • 34% H 20 has higher critical temperature • Critical temperature depends on partial pressure of H 20
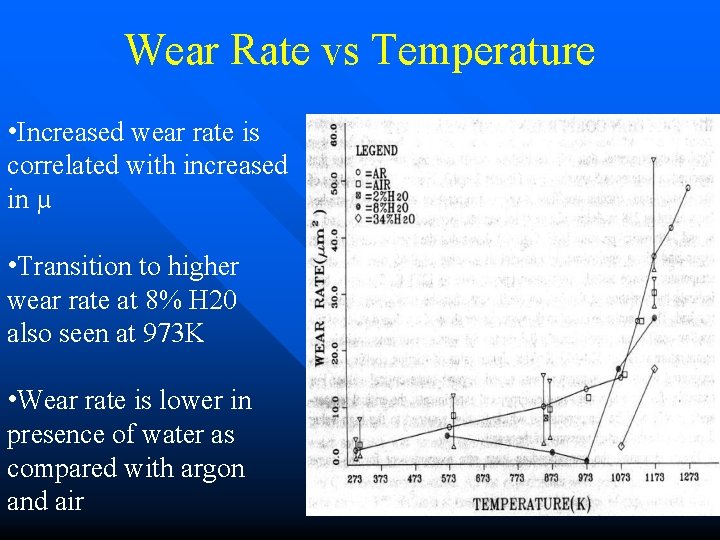
Wear Rate vs Temperature • Increased wear rate is correlated with increased in µ • Transition to higher wear rate at 8% H 20 also seen at 973 K • Wear rate is lower in presence of water as compared with argon and air

Wear Grooves and Rolls • Optical micrograph of wear groove with 8% H 2 O vapor at 973 K • Cylindrical rolls oriented perpendicular to sliding direction • Geometry of rolls dependent on temperature and water vapor content • Rolls provide mechanical support between surfaces and reduce actual surface area contact

SEM of “Rolls” • SEM of “rolls” with 34% H 2 O vapor at 873 K • Rolls develop perpendicular to the sliding direction • Rolls are formed from smaller wear particles that adhere and form the cylinders (ie Playdoh)

SEM of “Rolls” • SEM of “rolls” with 34% H 2 O vapor at 873 K • Surface shows delamination and resulting debris particles • Debris particles are flattened and curled into a roll • Many layers of debris can be seen on rolls
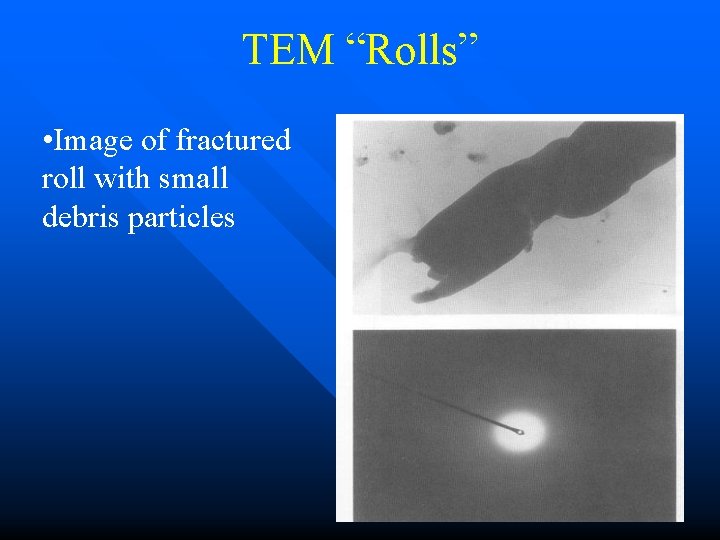
TEM “Rolls” • Image of fractured roll with small debris particles
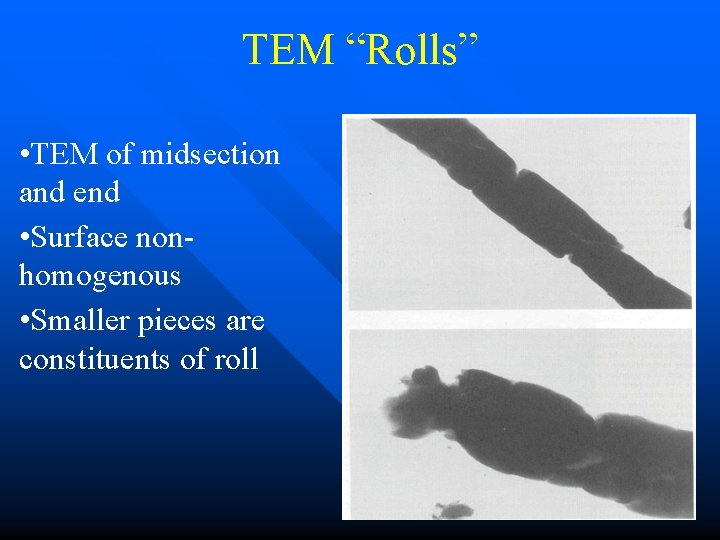
TEM “Rolls” • TEM of midsection and end • Surface nonhomogenous • Smaller pieces are constituents of roll

Friction and Wear vs Temperature • 2 transition temperatures for friction and wear • At the lower transition temperature, for H 2 O trials, µ reduces to about 1/2 the coefficient of friction at room temperature.

Friction and Wear vs Temperature • At the higher transition temperature, for H 2 O trials, the µ increases to level of air and argon • This higher transition temperature is dependent on the partial pressure of water.

Lower Transition Temperature What going on at the lower transition temperature? • Formation of Oxide Si 3 N 4 + 3 O 2 = 3 Si. O 2 + 2 N 2 • The increase in temperature allows: • significant oxide formation to reduce µ and wear • H 20 vapor to modify Si. O 2 and lower it’s viscosity to form rolls • No rolls if Si. O 2 is too hard and brittle

Higher Transition Temperature What going on at the higher transition temperature? • Rolls begin to break down • Bigger and thicker rolls last longer • Produced by higher H 2 O vapor pressure • Si. O 2 layer breaks down • Becomes too soft • Displaced and squeezed out of contact surface • Therefore wear increases

Conclusion §Formation of rolls is a big factor in reducing µ and wear §Formation of rolls are dependent on H 20 vapor pressure and temperature §Therefore µ and wear rates of silicon nitride are dependent on temperature and humidity
- Slides: 16