Tri Guard Device for Cerebral Embolic Protection During

Tri. Guard Device for Cerebral Embolic Protection During Transcatheter Aortic Valve Replacement: A Multicenter Real-World Experience Masieh Abawi, Ermela Yzeiraj; Adriaan Kraaijeveld, ; Michiel Voskuil, ; Pieter A. Doevendans; Joachim Schofer; Pieter R. Stella Consecutive patients with aortic stenosis scheduled for TAVR and suitable anatomy for Tri. Guard, were included in this prospective 2 center study Successful device performance was defined as I) Accessing the aortic arch with delivery catheter; II) Deployment of the Tri. Guard filter unit from the delivery catheter into the aortic arch; III) Positioning the Tri. Guard to cover the cerebral vessels (as verified by angiography) without obstruction of the blood flow and interference with TAVR procedure; IV) Retrieving and removing the Tri. Guard system intact
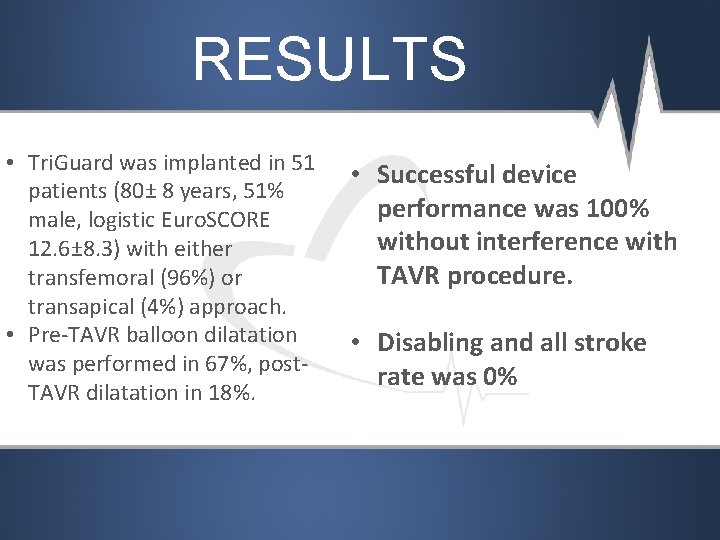
RESULTS • Tri. Guard was implanted in 51 patients (80± 8 years, 51% male, logistic Euro. SCORE 12. 6± 8. 3) with either transfemoral (96%) or transapical (4%) approach. • Pre-TAVR balloon dilatation was performed in 67%, post. TAVR dilatation in 18%. • Successful device performance was 100% without interference with TAVR procedure. • Disabling and all stroke rate was 0%

DW-MRI Results in Patients With and Without Tri. Guard Cerebral Embolic Protection During TAVI Ermela Yzeiraj, Klaudija Bijuklic, Timo Haselbach, Julian Witt, Korff Krause, Lorenz Hansen, Friedrich-Christian Riess, Joachim Schofer • 10 patients received TAVI under cerebral embolic protection with the Tri. Guard device. • Control patients from the same site where 150 patients who underwent TAVI without cerebral embolic protection. • All patients were treated with a balloon expandable valve and underwent DW-MRI 3 -5 days after the procedure. • Cerebral DW-MRI were analysed by a blinded physician to assess incidence, number and volume of new CI.
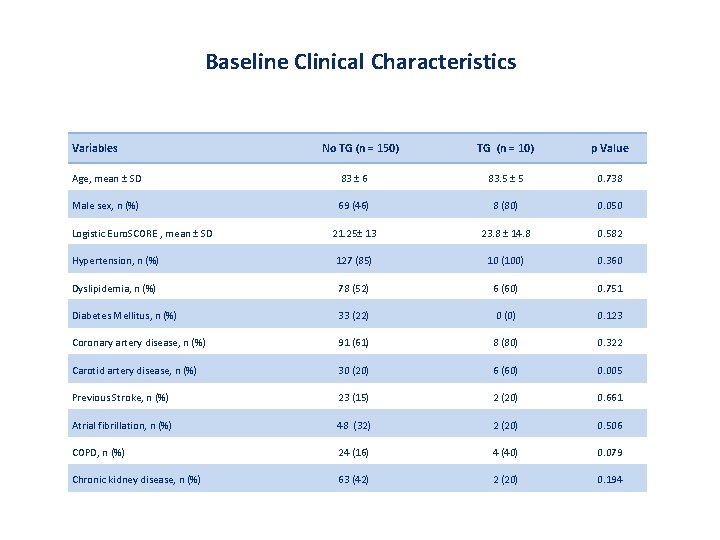
Baseline Clinical Characteristics Variables No TG (n = 150) TG (n = 10) p Value Age, mean ± SD 83 ± 6 83. 5 ± 5 0. 738 Male sex, n (%) 69 (46) 8 (80) 0. 050 21. 25± 13 23. 8 ± 14. 8 0. 582 Hypertension, n (%) 127 (85) 10 (100) 0. 360 Dyslipidemia, n (%) 78 (52) 6 (60) 0. 751 Diabetes Mellitus, n (%) 33 (22) 0 (0) 0. 123 Coronary artery disease, n (%) 91 (61) 8 (80) 0. 322 Carotid artery disease, n (%) 30 (20) 6 (60) 0. 005 Previous Stroke, n (%) 23 (15) 2 (20) 0. 661 Atrial fibrillation, n (%) 48 (32) 2 (20) 0. 506 COPD, n (%) 24 (16) 4 (40) 0. 079 Chronic kidney disease, n (%) 63 (42) 2 (20) 0. 194 Logistic Euro. SCORE , mean ± SD
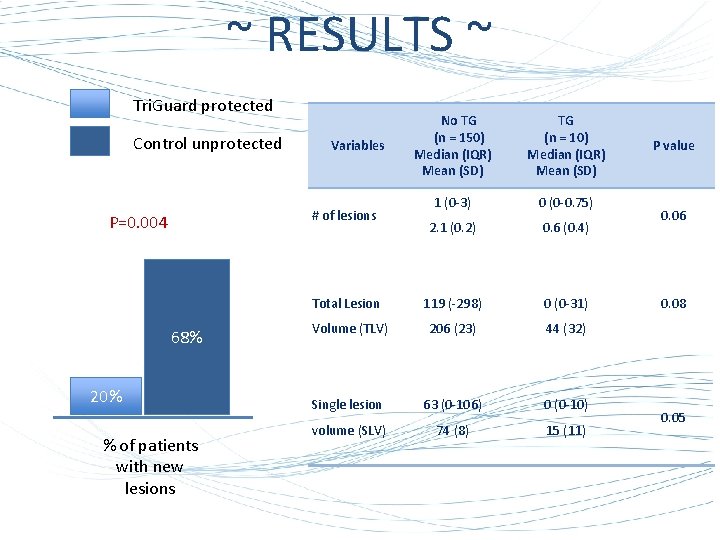
~ RESULTS ~ Tri. Guard protected Control unprotected No TG (n = 150) Median (IQR) Mean (SD) TG (n = 10) Median (IQR) Mean (SD) 1 (0 -3) 0 (0 -0. 75) 2. 1 (0. 2) 0. 6 (0. 4) Total Lesion 119 (-298) 0 (0 -31) Volume (TLV) 206 (23) 44 (32) Single lesion 63 (0 -106) 0 (0 -10) volume (SLV) 74 (8) 15 (11) Variables # of lesions P=0. 004 68% 20% % of patients with new lesions P value 0. 06 0. 08 0. 05
Tri. Guard device is safe and effective with a high performance success without interference with TAVR procedure and with 0% stroke rate
- Slides: 6