Trends the Periodic Table Trends see properties change

Trends & the Periodic Table

Trends • see properties change in predictable ways based location of elements on PT • some properties can be predicted: density melting point/boiling point * atomic radius * ionization energy TABLE S • electronegativity anyone know where we can find these numbers
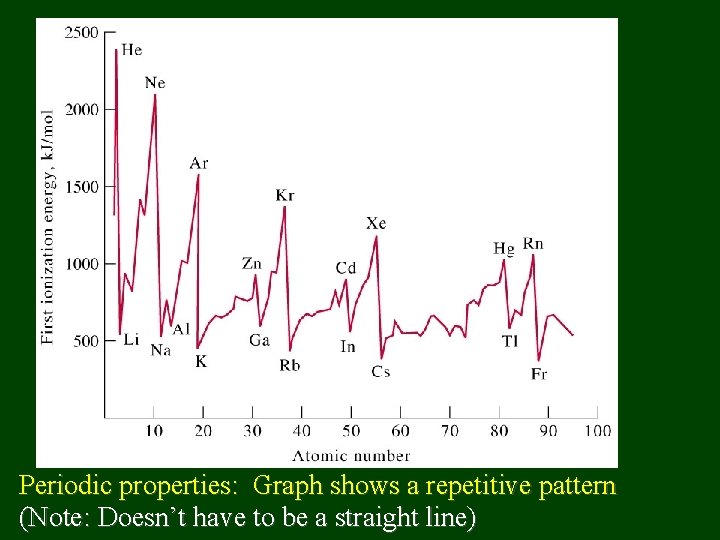
Periodic properties: Graph shows a repetitive pattern (Note: Doesn’t have to be a straight line)
When you’re done it will look like this so leave room for writing!
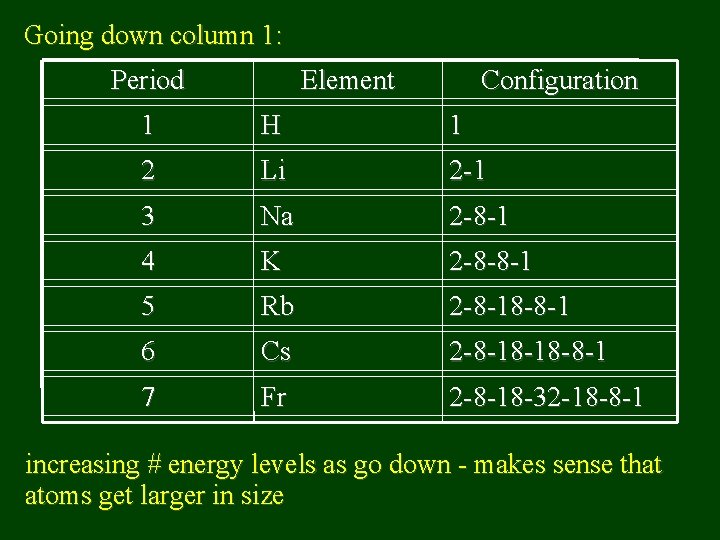
Going down column 1: Period Element Configuration 1 H 1 2 Li 2 -1 3 Na 2 -8 -1 4 K 2 -8 -8 -1 5 Rb 2 -8 -18 -8 -1 6 Cs 2 -8 -18 -18 -8 -1 7 Fr 2 -8 -18 -32 -18 -8 -1 increasing # energy levels as go down - makes sense that atoms get larger in size
Increasing number of energy levels
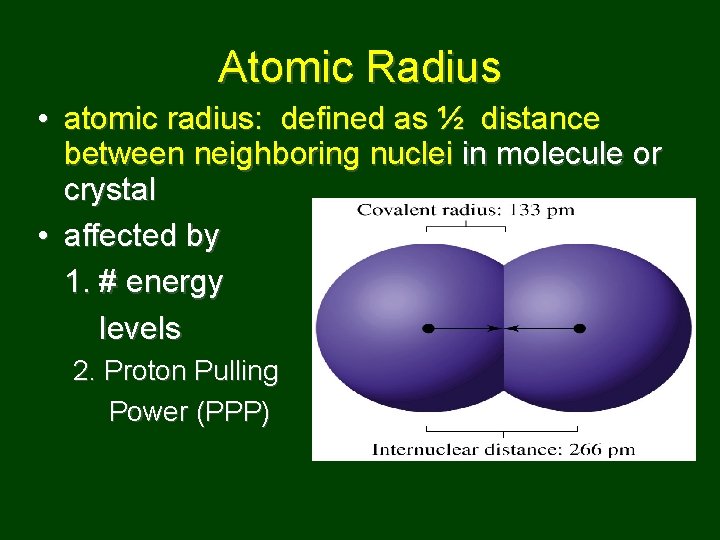
Atomic Radius • atomic radius: defined as ½ distance between neighboring nuclei in molecule or crystal • affected by 1. # energy levels 2. Proton Pulling Power (PPP)
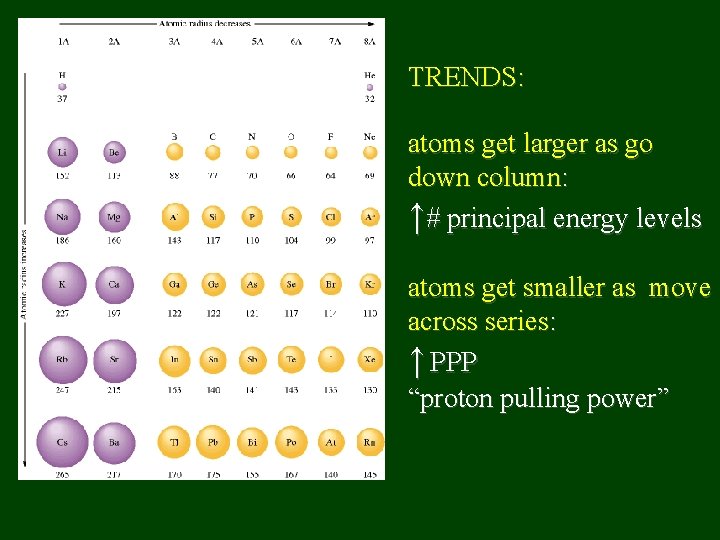
TRENDS: atoms get larger as go down column: ↑# principal energy levels atoms get smaller as move across series: ↑ PPP “proton pulling power”
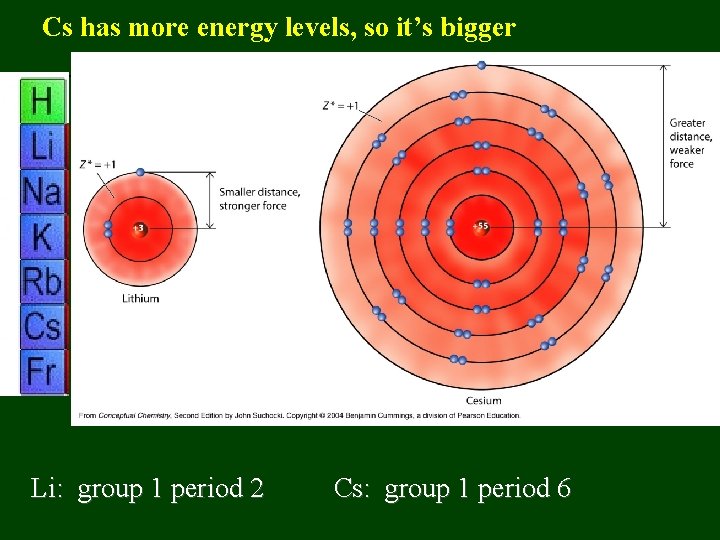
Cs has more energy levels, so it’s bigger Li: group 1 period 2 Cs: group 1 period 6
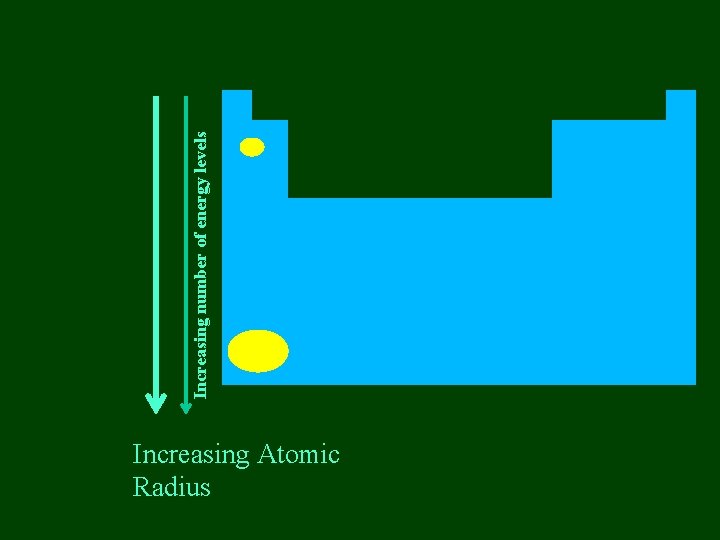
Increasing Atomic Radius Increasing number of energy levels
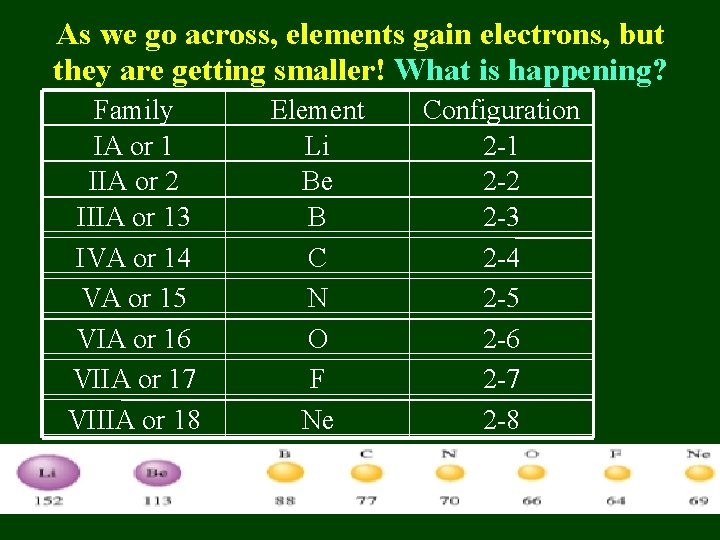
As we go across, elements gain electrons, but they are getting smaller! What is happening? Family IA or 1 IIA or 2 IIIA or 13 IVA or 14 VA or 15 VIA or 16 VIIA or 17 VIIIA or 18 Element Li Be B C N O F Ne Configuration 2 -1 2 -2 2 -3 2 -4 2 -5 2 -6 2 -7 2 -8
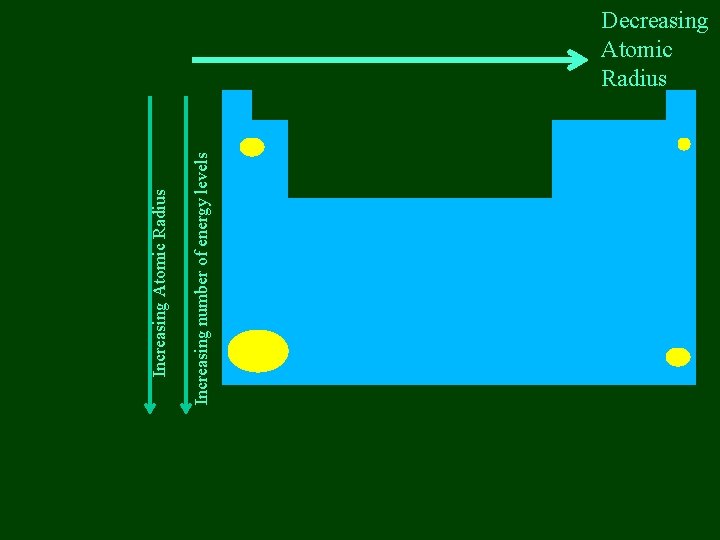
Increasing number of energy levels Increasing Atomic Radius Decreasing Atomic Radius

Why does this happen. . • as go from left to right, you gain more protons (atomic number increases) • results in greater “proton pulling power” – remember: nucleus is (+) and electrons are (-) so e- get pulled towards the nucleus • more protons you have, the stronger PPP
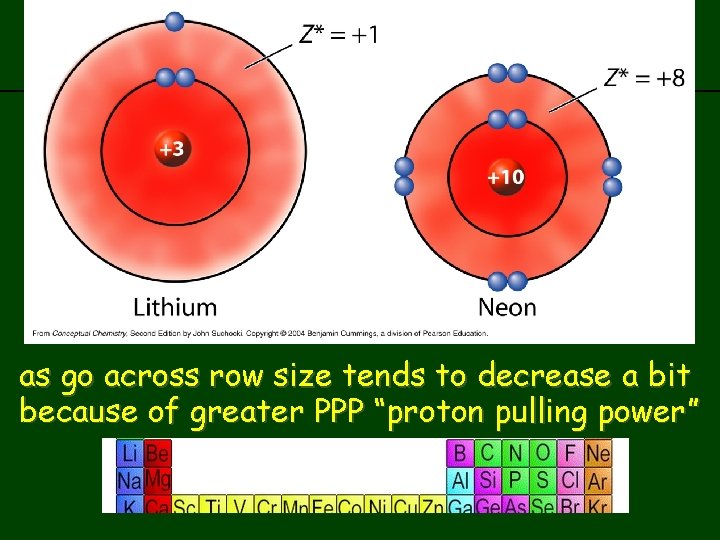
previous | index | next across row size tends to decrease as go a bit because of greater PPP “proton pulling power”

We can “measure” the PPP by determining the effective nuclear charge • this is charge actually felt by valence electrons • equation to calculate effective nuclear charge: nuclear charge - # inner shell electrons (doesn’t include valance e-)
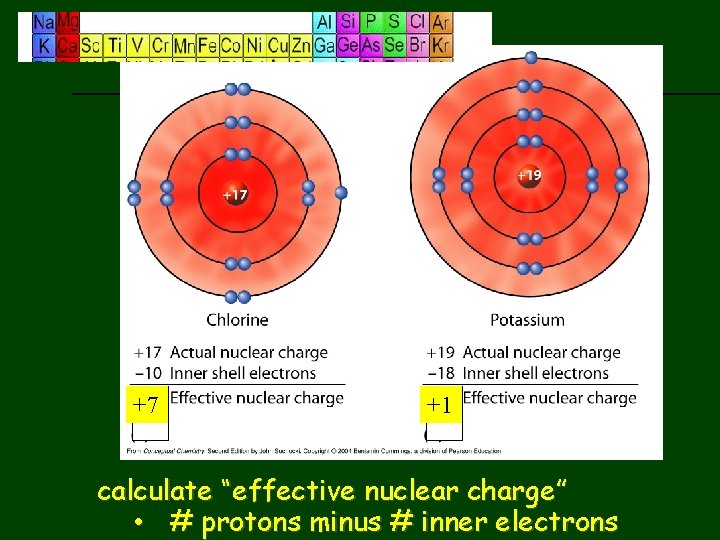
previous | index | next +7 +1 calculate “effective nuclear charge” • # protons minus # inner electrons
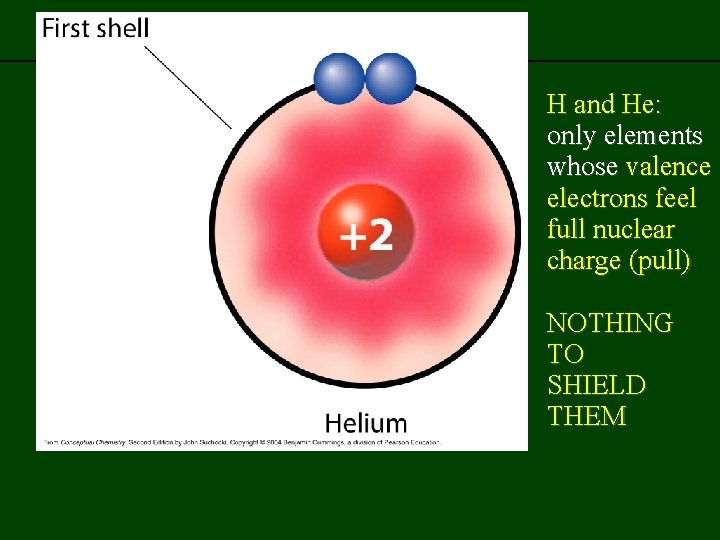
previous | index | next H and He: only elements whose valence electrons feel full nuclear charge (pull) NOTHING TO SHIELD THEM
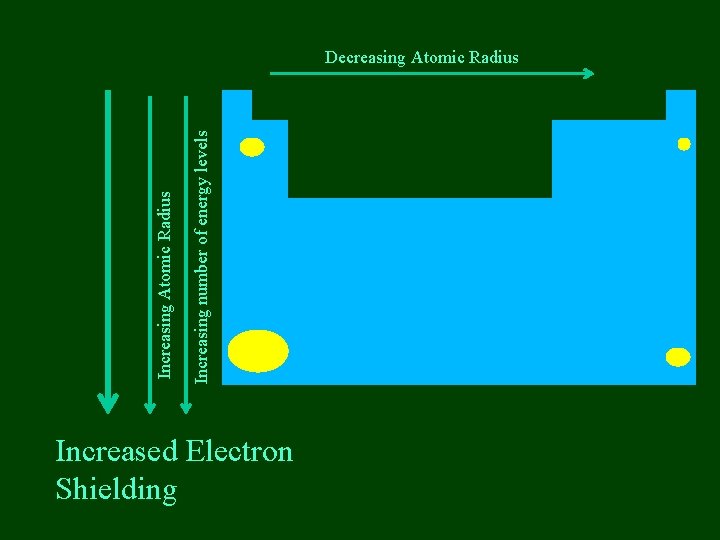
Increasing number of energy levels Increasing Atomic Radius Decreasing Atomic Radius Increased Electron Shielding
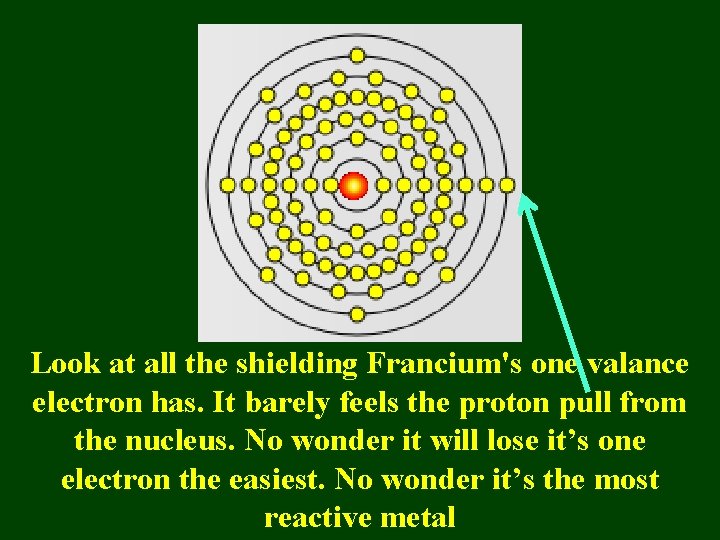
Look at all the shielding Francium's one valance electron has. It barely feels the proton pull from the nucleus. No wonder it will lose it’s one electron the easiest. No wonder it’s the most reactive metal

Ionization Energy • definition: amount energy required to remove farthest valence e- from atom • 1 st ionization energy: energy required to remove most loosely held valence electron (valence e- farthest from nucleus)

Trends in Ionization Energy • What do you think happens to the ionization energy as go down column of PT? decreases • As go across row? increases

Electronegativity • ability of atom to attract electrons to itself so can form bonds with other elements (to create cmpds) • noble gases tend not to form bonds, so don’t have electronegativity values • Fluorine: most electronegative element = 4. 0 Paulings • Francium: least electronegative element = 0. 7 Paulings
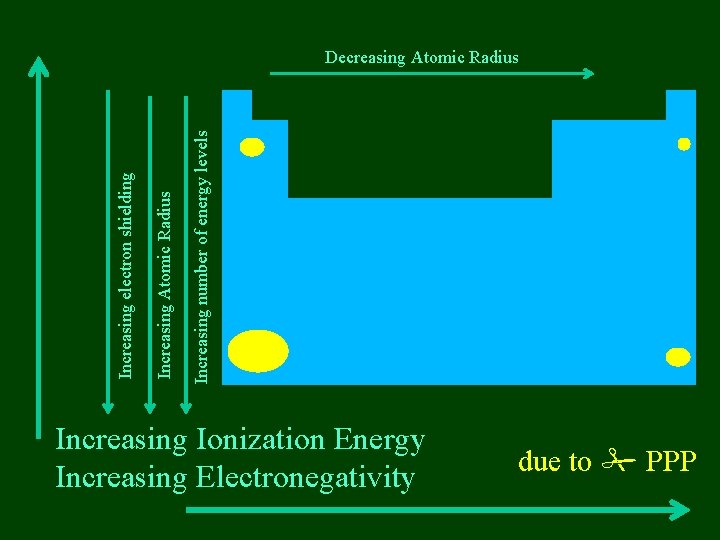
Increasing number of energy levels Increasing Atomic Radius Increasing electron shielding Decreasing Atomic Radius Increasing Ionization Energy Increasing Electronegativity due to PPP

previous | index | next elements in same group: farther away valence electrons are from nucleus the easier to remove them easier for Cs (top of column) to lose electrons than Li (bottom of column) so Cs is a more reactive metal!
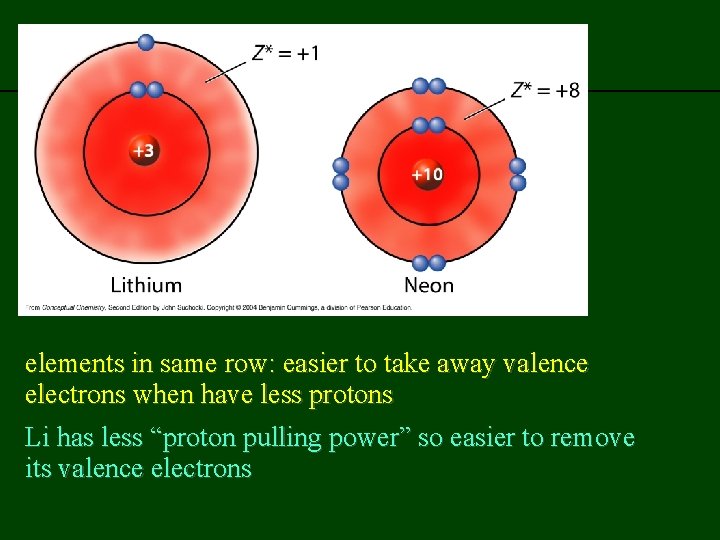
previous | index | next elements in same row: easier to take away valence electrons when have less protons Li has less “proton pulling power” so easier to remove its valence electrons

Reactivity of Metals • metals are losers! • judge reactivity of metals by how easily give up electrons to form (+) ions • most active metals: Fr (then Cs) • for metals, reactivity increases as ionization energy goes down

Trends for Reactivity (Metallic Character) of Metals • increases as go down column – easier to lose electrons! • decreases as go across row – more difficult to lose electrons!

Reactivity of Non-metals • non-metals are winners! • judge reactivity of non-metals by how easily gain electrons • F: most active non-metal • for non-metals: – reactivity ↑ as electronegativity ↑

Trend for Reactivity of Non-metals: depends on PPP • increases as go across row • decreases as go down column – (shielded by more inner-shell electrons)

How do you know if an atom gains or loses electrons? • • • think back to the Lewis structures of ions atoms form ions to get a valence # of 8 (or 2 for H) metals tend to have 1, 2, or 3 valence electrons – it’s easier to lose these than gain extra needed • non-metals tend to have 5, 6, or 7 valence electrons – it’s easier to add extra needed than to lose these • noble gases already have 8 so they don’t form ions very easily
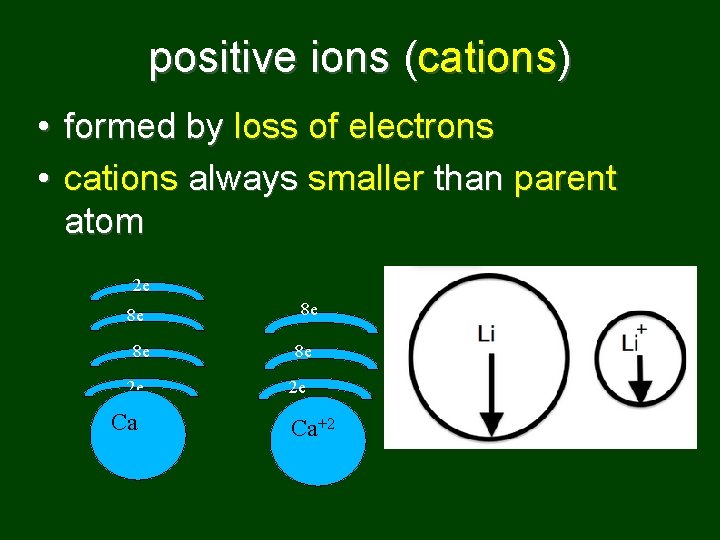
positive ions (cations) • formed by loss of electrons • cations always smaller than parent atom 2 e 8 e 8 e 2 e Ca Ca 8 e 8 e 2 e Ca+2

negative ions or (anions) • formed by gain of electrons • anions always larger than parent atom

Allotropes • different structural forms of element in same phase – different structures and properties – examples: C and O

Graphite and Diamond: both carbon in solid form
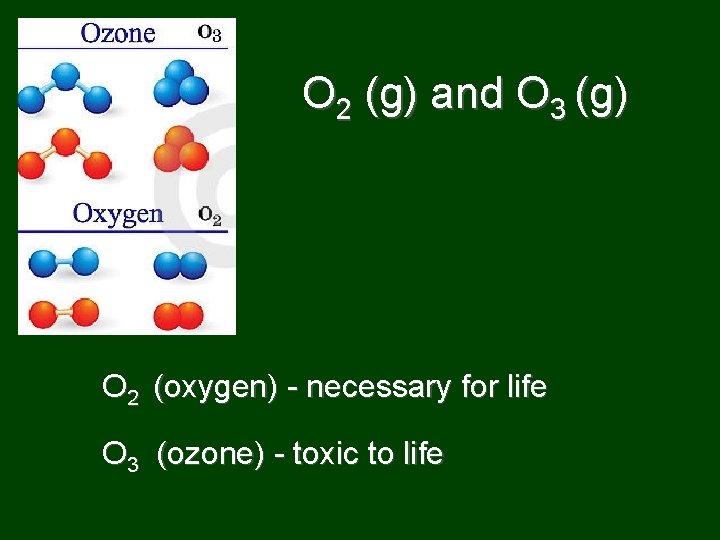
O 2 (g) and O 3 (g) O 2 (oxygen) - necessary for life O 3 (ozone) - toxic to life
- Slides: 35