Trends the Periodic Table Trends More than 20

Trends & the Periodic Table

Trends • More than 20 properties change in a predictable way based on location of element in P. T. • Include: density, melting point, atomic radius, ionization energy, electronegativity

Atomic Radius • Atomic radius: defined as half the distance between neighboring nuclei in a molecule or crystal. • “size” varies a bit from substance to substance.
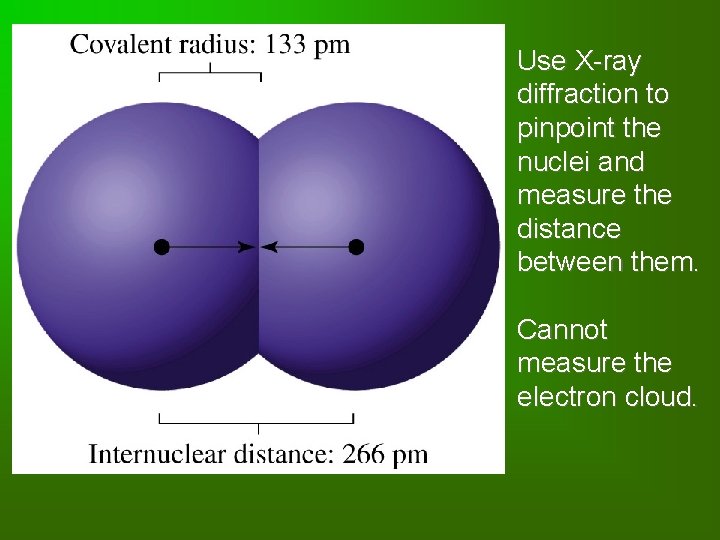
Use X-ray diffraction to pinpoint the nuclei and measure the distance between them. Cannot measure the electron cloud.

Trends: Atoms get larger as you go down a column – more principal energy levels Atoms get smaller as you move across a series – more “proton pulling power. ”
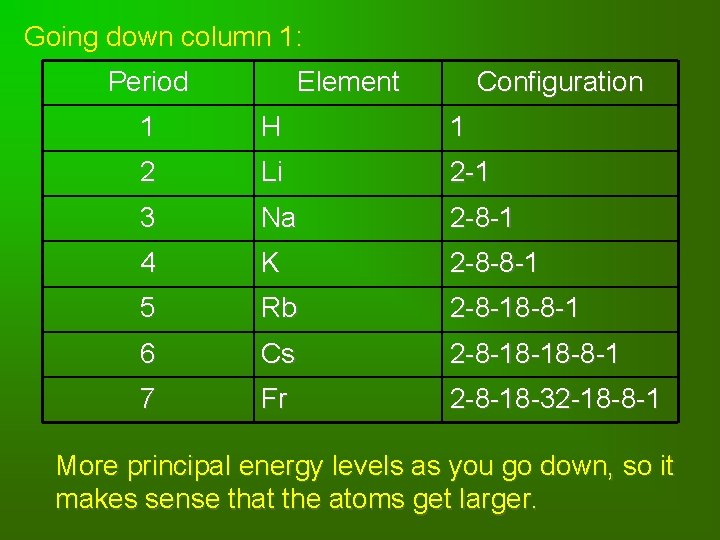
Going down column 1: Period Element Configuration 1 H 1 2 Li 2 -1 3 Na 2 -8 -1 4 K 2 -8 -8 -1 5 Rb 2 -8 -18 -8 -1 6 Cs 2 -8 -18 -18 -8 -1 7 Fr 2 -8 -18 -32 -18 -8 -1 More principal energy levels as you go down, so it makes sense that the atoms get larger.
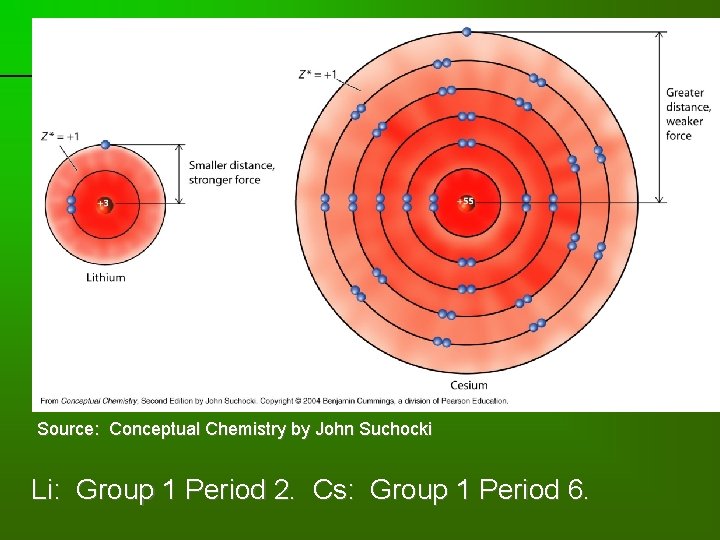
previous | index | next Source: Conceptual Chemistry by John Suchocki Li: Group 1 Period 2. Cs: Group 1 Period 6.

But why do the atoms get smaller as you go across the periodic table? • You are still adding electrons!
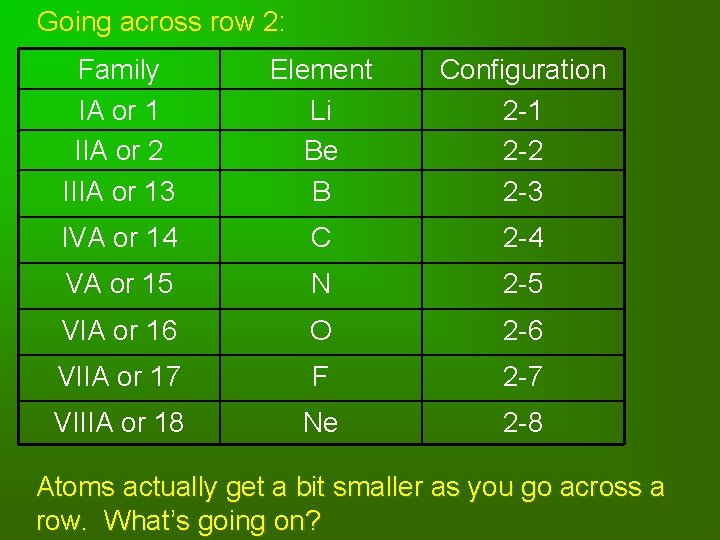
Going across row 2: Family IA or 1 IIA or 2 IIIA or 13 Element Li Be B Configuration 2 -1 2 -2 2 -3 IVA or 14 C 2 -4 VA or 15 N 2 -5 VIA or 16 O 2 -6 VIIA or 17 F 2 -7 VIIIA or 18 Ne 2 -8 Atoms actually get a bit smaller as you go across a row. What’s going on?

What do you remember about charge? • Opposites attract and like charges repel. • The valence electrons are pulled into the atom by the positive charge on the nucleus. • The greater the positive charge, the more pulling power, right?

Well, almost. . . • Except for H and He, the valence electrons don’t feel the full effect of all the protons in the nucleus. • For most atoms, the inner shell electrons “shield” the valence electrons from the nucleus.

Effective nuclear charge • = Atomic Number - # of inner shell electrons.

Effective nuclear charge • Charge actually felt by valence electrons. • = Atomic Number - # of inner shell electrons. • Not usually the same as the nuclear charge or number of protons in the nucleus. • Charge felt by valence electrons is attenuated or shielded by inner shell electrons.
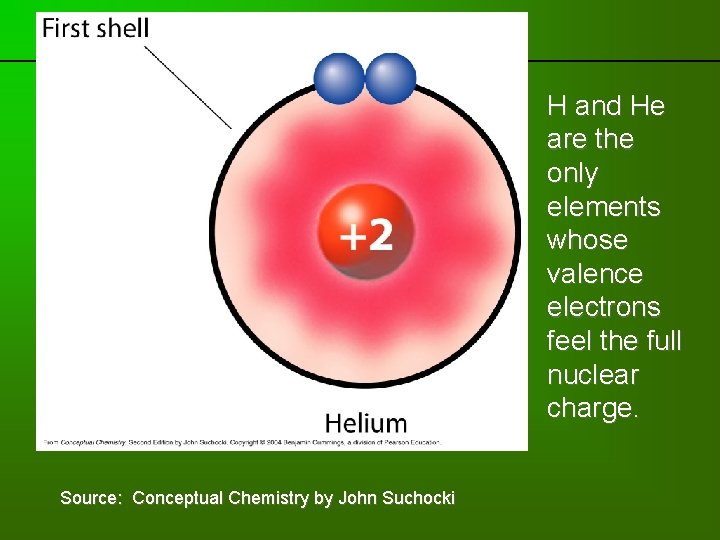
previous | index | next Source: Conceptual Chemistry by John Suchocki H and He are the only elements whose valence electrons feel the full nuclear charge.
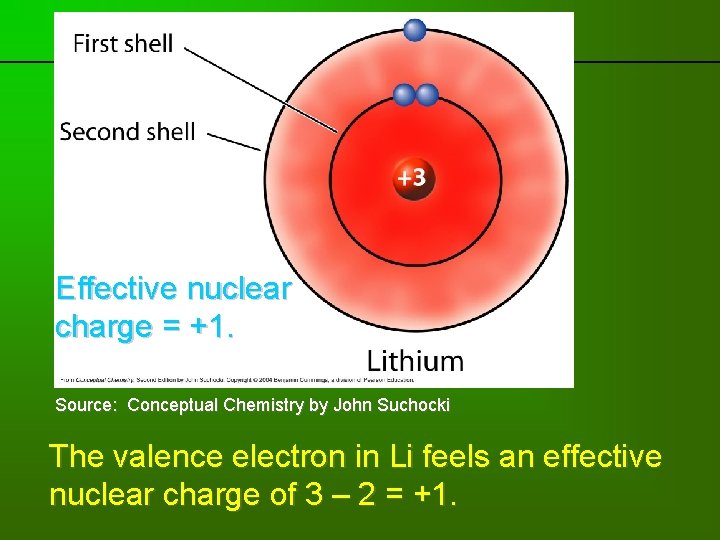
previous | index | next Effective nuclear charge = +1. Source: Conceptual Chemistry by John Suchocki The valence electron in Li feels an effective nuclear charge of 3 – 2 = +1.

previous | index | next Source: Conceptual Chemistry by John Suchocki Calculating “effective nuclear charge. ”
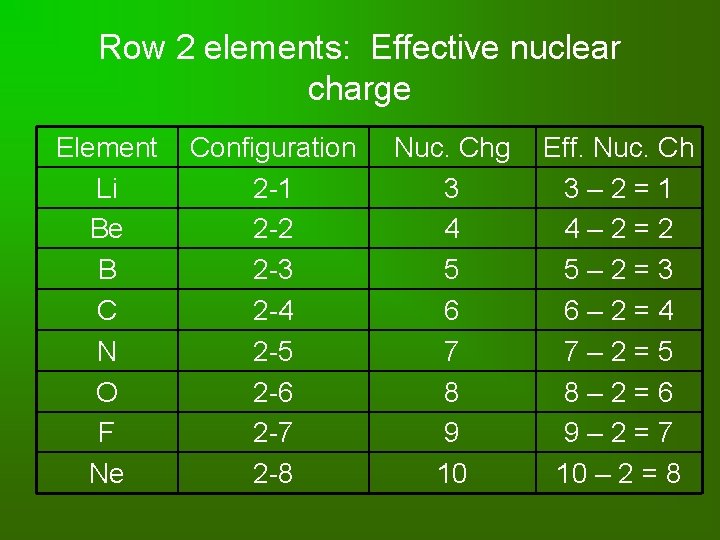
Row 2 elements: Effective nuclear charge Element Li Be B C N O F Ne Configuration 2 -1 2 -2 2 -3 2 -4 2 -5 2 -6 2 -7 2 -8 Nuc. Chg 3 4 5 6 7 8 9 10 Eff. Nuc. Ch 3 – 2 = 1 4 – 2 = 2 5 – 2 = 3 6 – 2 = 4 7 – 2 = 5 8 – 2 = 6 9 – 2 = 7 10 – 2 = 8
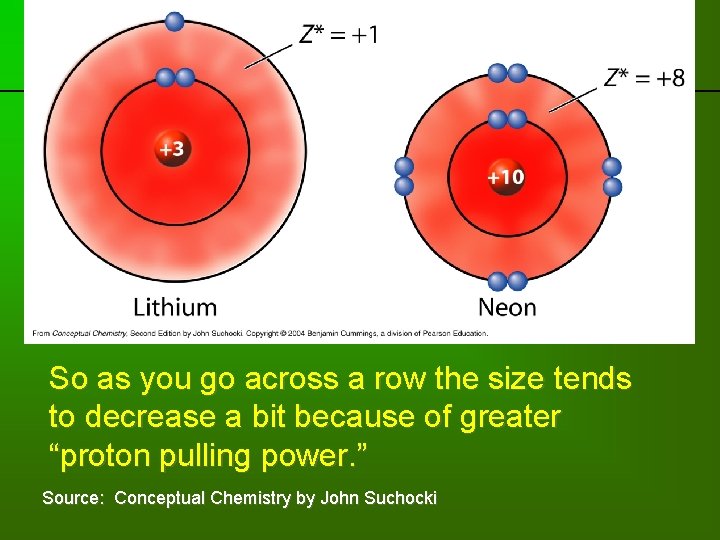
previous | index | next So as you go across a row the size tends to decrease a bit because of greater “proton pulling power. ” Source: Conceptual Chemistry by John Suchocki
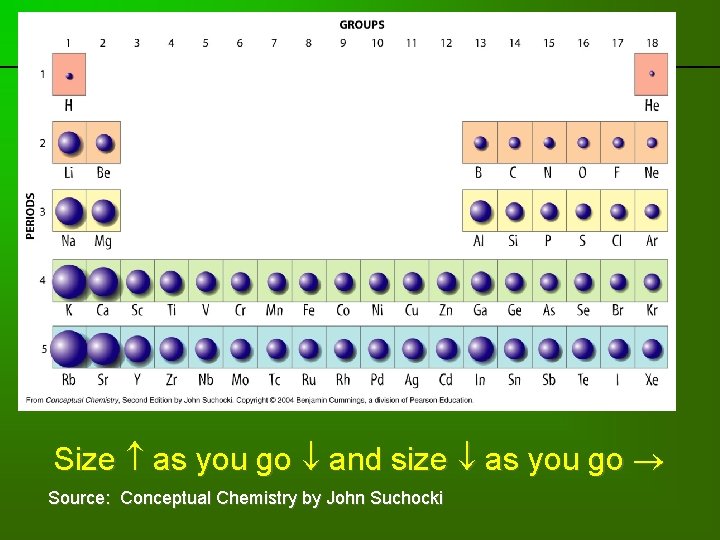
previous | index | next Size as you go and size as you go Source: Conceptual Chemistry by John Suchocki

IONS • An ion is a charged particle formed by the gain or loss of electrons. – CATION has a positive charge – ANION has a negative charge

Ionic Size Relative to Parent Atom • Depends on if it’s a positive ion or a negative ion • How do you make a positive ion? Remove electrons • How do you make a negative ion? Add electrons
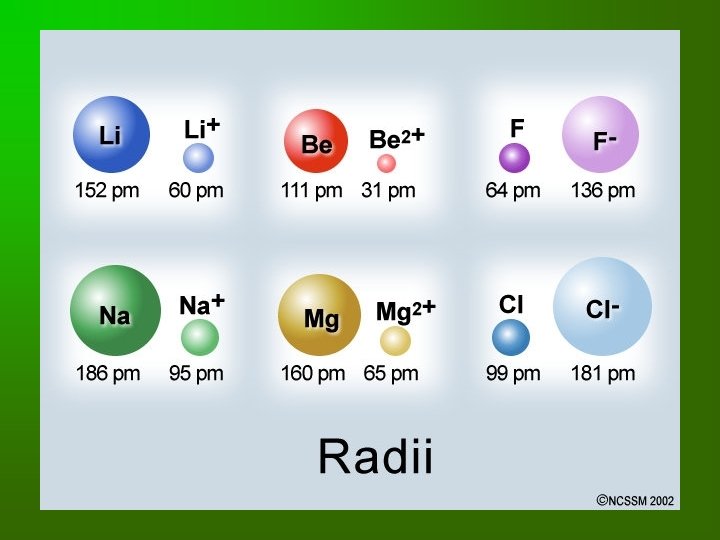

Oxidation Number • Oxidation Number (State) – number assigned to keep track of electron gain or loss in redox reactions. – Positive number means electrons LOST – Negative number means electrons GAINED
- Slides: 23