Trends the Periodic Table Trends more than 20

Trends & the Periodic Table

Trends • more than 20 properties change in predictable way based location of elements on PT • some properties: – Density – melting point/boiling point – atomic radius – ionization energy – electronegativity

Atomic Radius • Atomic radius: defined as ½ distance between neighboring nuclei in molecule or crystal • “size” varies a bit from substance to substance
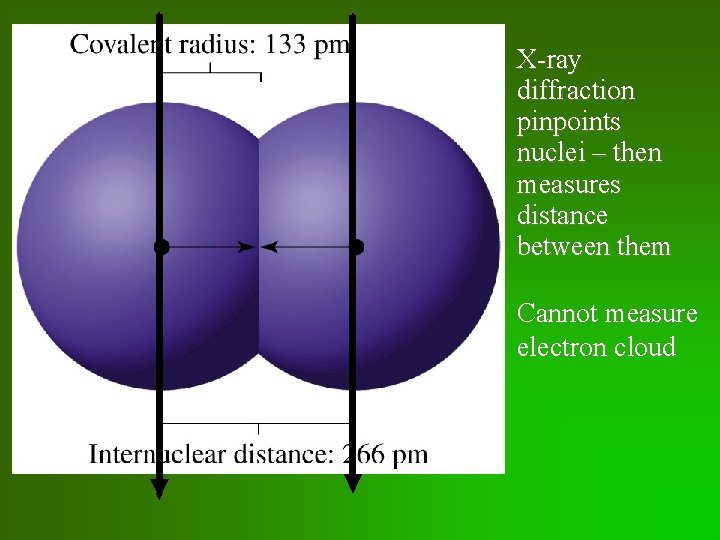
X-ray diffraction pinpoints nuclei – then measures distance between them Cannot measure electron cloud
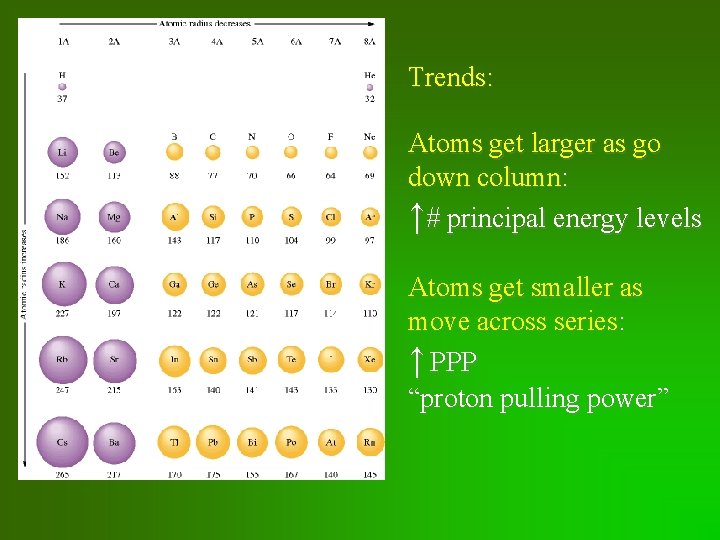
Trends: Atoms get larger as go down column: ↑# principal energy levels Atoms get smaller as move across series: ↑ PPP “proton pulling power”
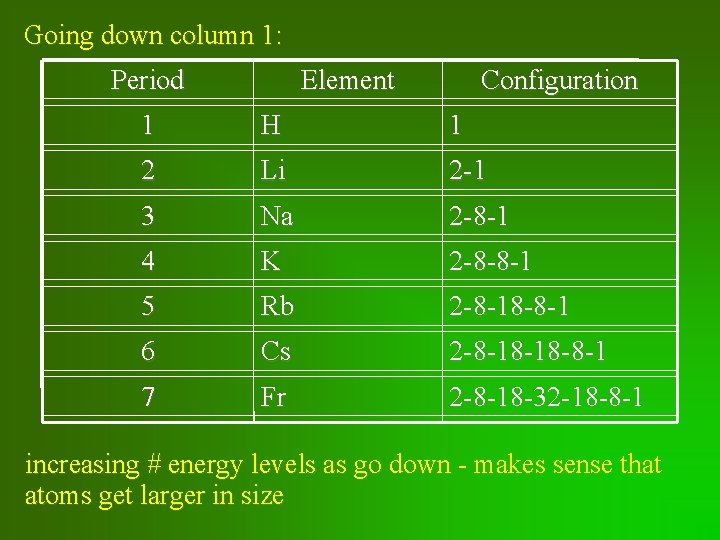
Going down column 1: Period Element Configuration 1 H 1 2 Li 2 -1 3 Na 2 -8 -1 4 K 2 -8 -8 -1 5 Rb 2 -8 -18 -8 -1 6 Cs 2 -8 -18 -18 -8 -1 7 Fr 2 -8 -18 -32 -18 -8 -1 increasing # energy levels as go down - makes sense that atoms get larger in size
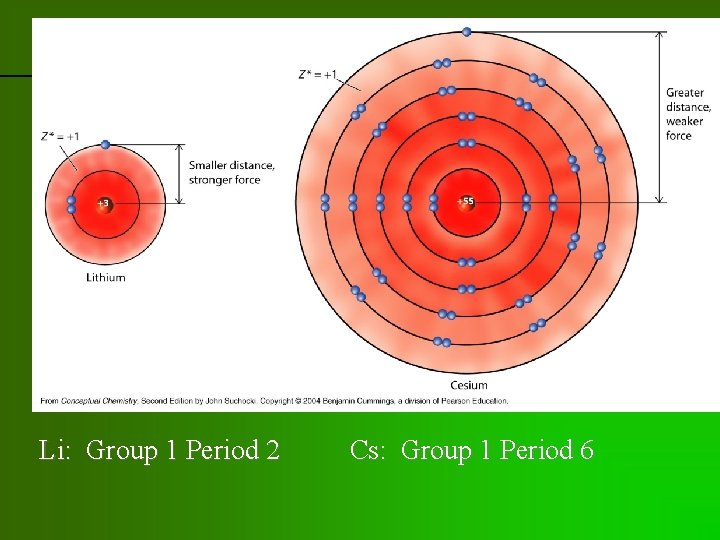
previous | index | next Li: Group 1 Period 2 Cs: Group 1 Period 6
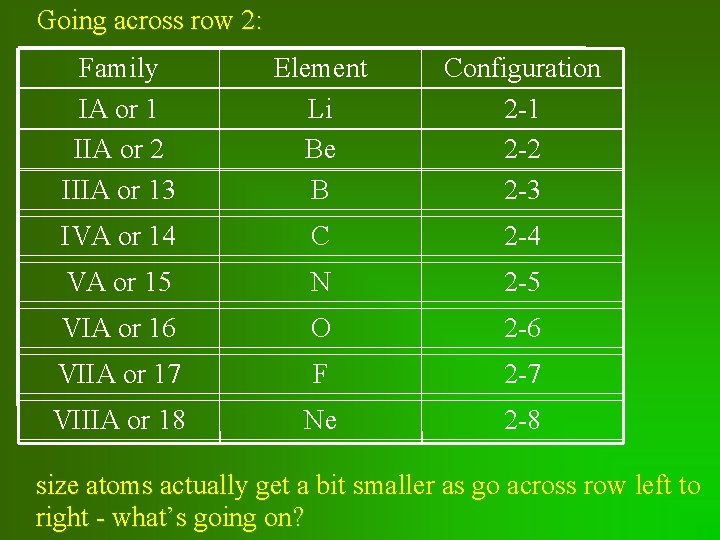
Going across row 2: Family IA or 1 IIA or 2 IIIA or 13 Element Li Be B Configuration 2 -1 2 -2 2 -3 IVA or 14 C 2 -4 VA or 15 N 2 -5 VIA or 16 O 2 -6 VIIA or 17 F 2 -7 VIIIA or 18 Ne 2 -8 size atoms actually get a bit smaller as go across row left to right - what’s going on?

What do you remember about charge? • opposites attract • like charges repel • largest influence on atomic size in order: • # principal energy levels • “proton pulling power (PPP)”

Effective nuclear charge • Charge actually felt by valence electrons • = Atomic Number minus # inner shell electrons • Not same as nuclear charge or # protons in nucleus • Charge felt by valence electrons is attenuated (shielded) by inner shell electrons
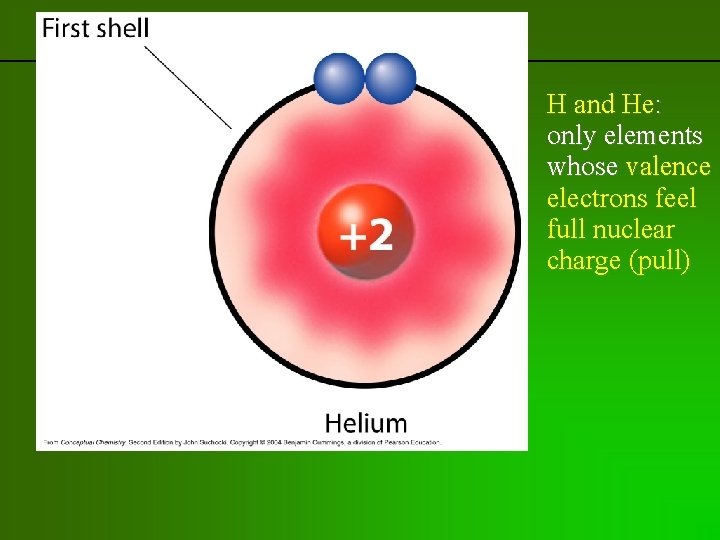
previous | index | next H and He: only elements whose valence electrons feel full nuclear charge (pull)
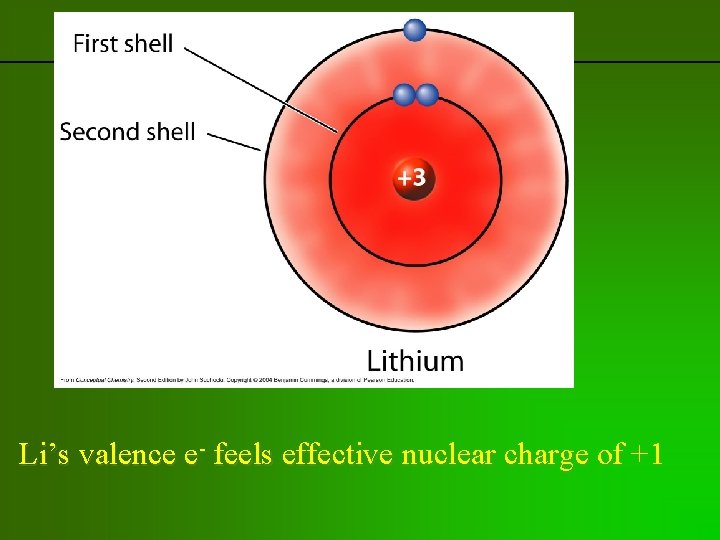
previous | index | next Li’s valence e- feels effective nuclear charge of +1
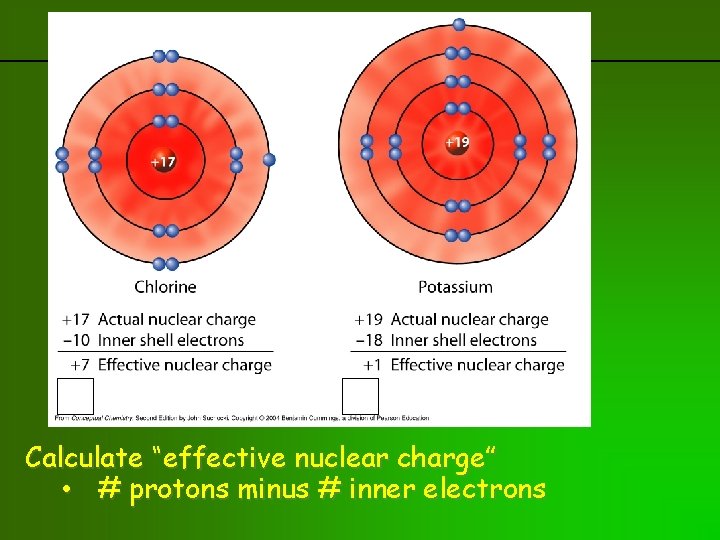
previous | index | next Calculate “effective nuclear charge” • # protons minus # inner electrons

previous | index | next across row size tends to decrease as go a bit because of greater PPP “proton pulling power”
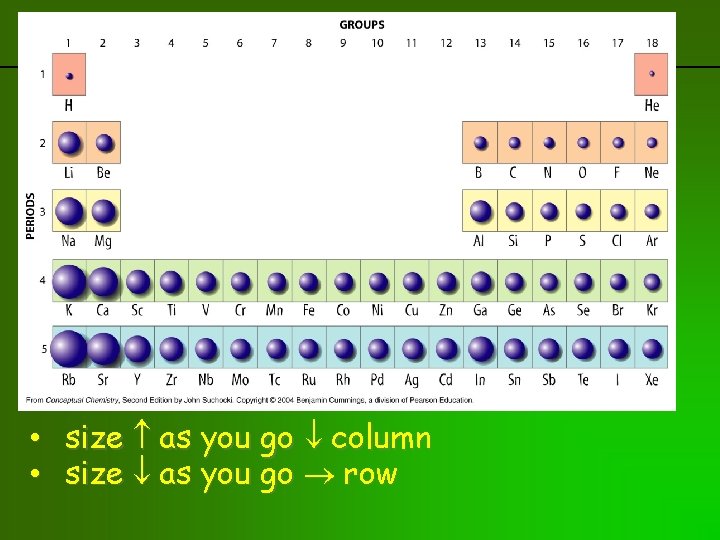
previous | index | next • size as you go column • size as you go row

Ionization Energy • = amount energy required to remove valence electron from atom in gas phase • 1 st ionization energy = energy required to remove most loosely held valence electron (e- farthest from nucleus)

Trends in ionization energy • What do you think happens to the ionization energy as go down column of PT? decreases • As go across row? increases

previous | index | next • Valence electrons in atoms: effective nuclear charge of +1 • Cs valence electron lot farther away from nucleus than Li • electrostatic attraction much weaker so easier to steal electron away from Cs
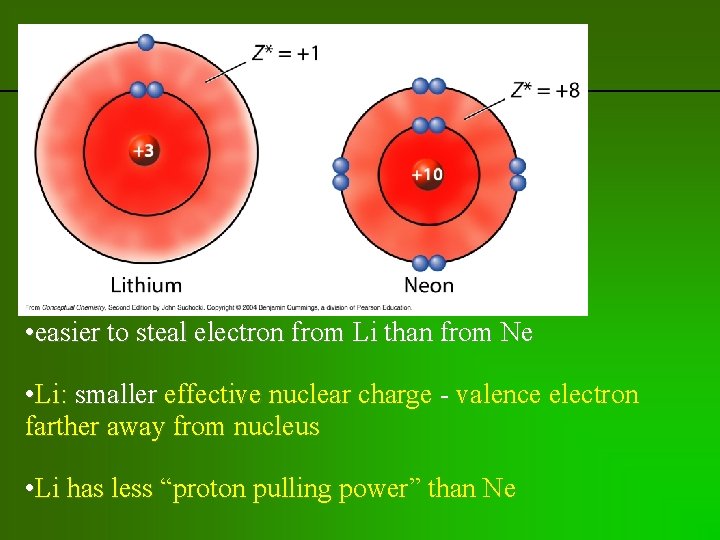
previous | index | next • easier to steal electron from Li than from Ne • Li: smaller effective nuclear charge - valence electron farther away from nucleus • Li has less “proton pulling power” than Ne

Trends in ionization energy • Ionization energy decreases as go down a column – gets easier to remove valence electron • Ionization energy increases as go across a row – more difficult to remove valence electron
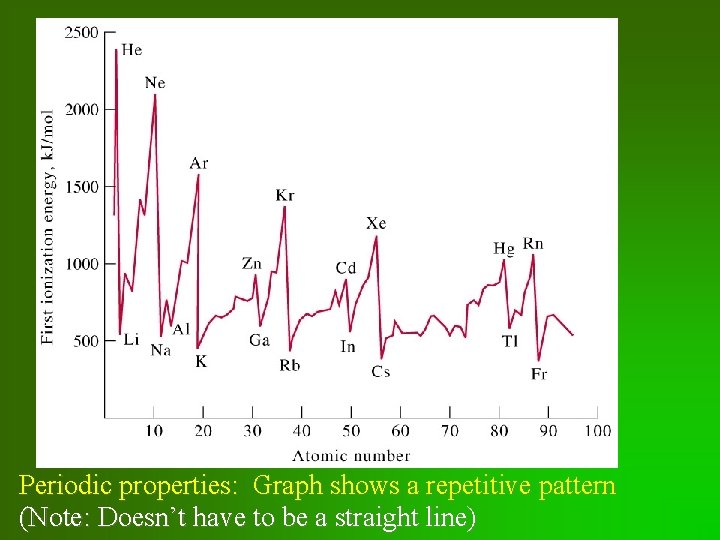
Periodic properties: Graph shows a repetitive pattern (Note: Doesn’t have to be a straight line)

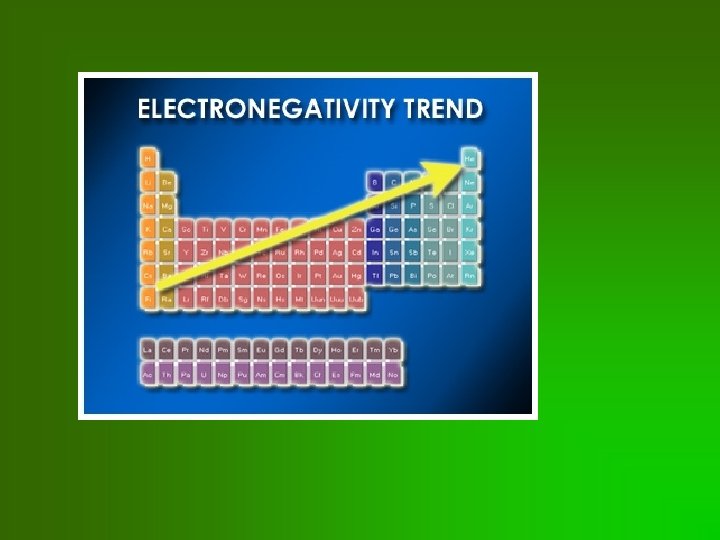
Electronegativity • ability of atom to attract electrons in bond • noble gases tend not to form bonds, so don’t have electronegativity values • Unit = Pauling • Fluorine: most electronegative element = 4. 0 Paulings

Trends in electronegativity • • • Related to PPP Increases as go across row Decreases as go down column • Remember: F most electronegative element!

Reactivity of Metals • Metals are losers! • judge reactivity of metals by how easily give up electrons • most active metals: Fr (then Cs) • For metals, reactivity increases as ionization energy goes down

Trends for Reactivity of Metals or Metallic Character • Increases as go down column – easier to lose electrons! • Decreases as go across row – more difficult to lose electrons!

Reactivity of Non-metals • Non-metals are winners! • judge reactivity of non-metals by how easily gain electrons • • F: most active non-metal For non-metals: – reactivity ↑ as electronegativity ↑

Trend for Reactivity of Non-metals: Depends on PPP • Increases as go across row • Decreases as go down column – (shielded by more inner-shell electrons)

Ionic Size Relative to Parent Atom • Depends if (+) ion or (-) ion • How do you make a positive ion? Remove electrons • How do you make a negative ion? Add electrons

How do you know if an atom gains or loses electrons? • Think back to the Lewis structures of ions • Atoms form ions to get a valence of 8 (or 2 for H) • Metals tend to have 1, 2, or 3 valence electrons – It’s easier to lose them • Nonmetals tend to have 5, 6, or 7 valence electrons – It’s easier to add some • Noble gases already have 8 so they don’t form ions very easily

Positive ions (cations) • Formed by loss of electrons • Cations always smaller than parent atom

Negative ions or (anions) • Formed by gain of electrons • Anions always larger than parent atom

Allotropes • Different forms of element in same phase – different structures and properties

O 2 and O 3 - both gas phase O 2 (oxygen) - necessary for life O 3 (ozone) - toxic to life

Graphite, diamond: both carbon in solid form

Graphite and Diamond
- Slides: 36