Trends the Periodic Table Ionization Energy Amount of

Trends & the Periodic Table

Ionization Energy • Amount of energy required to remove an electron from a gas-phase atom • First ionization energy or ionization energy = energy required to remove most loosely held valence electron

Trends in ionization energy • What do you think happens to the ionization energy as you go down a column of the periodic table? • As you go across a row?
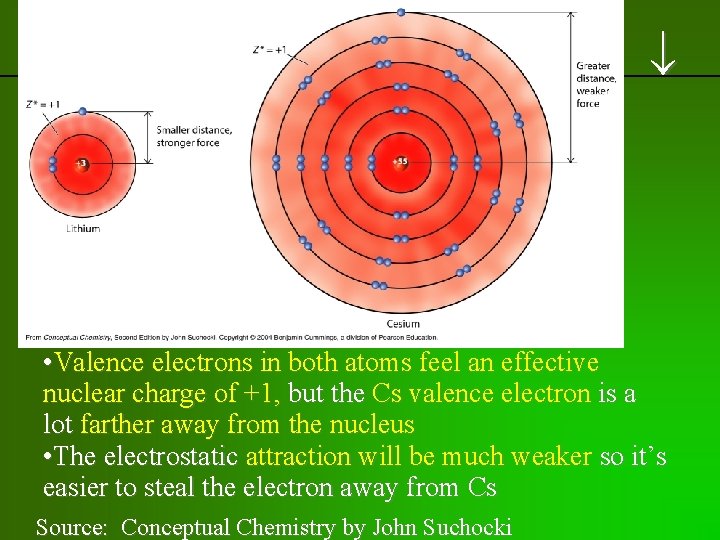
previous | index | next • Valence electrons in both atoms feel an effective nuclear charge of +1, but the Cs valence electron is a lot farther away from the nucleus • The electrostatic attraction will be much weaker so it’s easier to steal the electron away from Cs Source: Conceptual Chemistry by John Suchocki
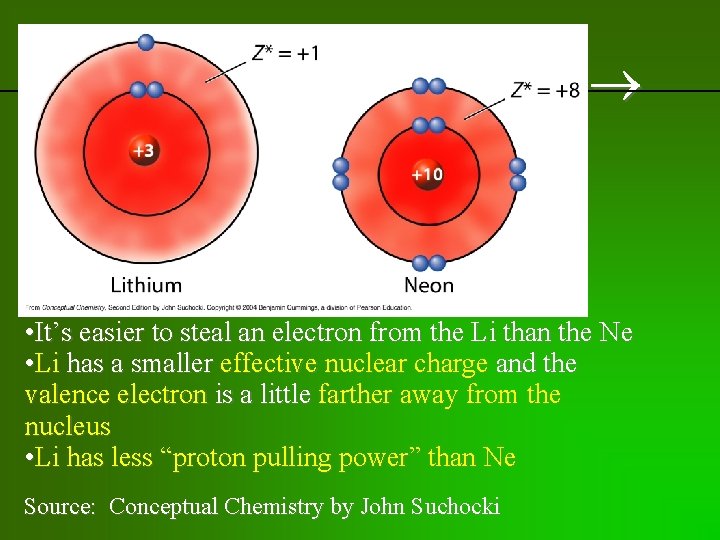
previous | index | next • It’s easier to steal an electron from the Li than the Ne • Li has a smaller effective nuclear charge and the valence electron is a little farther away from the nucleus • Li has less “proton pulling power” than Ne Source: Conceptual Chemistry by John Suchocki

Trends in ionization energy • Ionization energy decreases as you go down a column – it gets easier & easier to remove the valence electron • Ionization energy increases as you go across a row – it gets more difficult to remove the valence electron
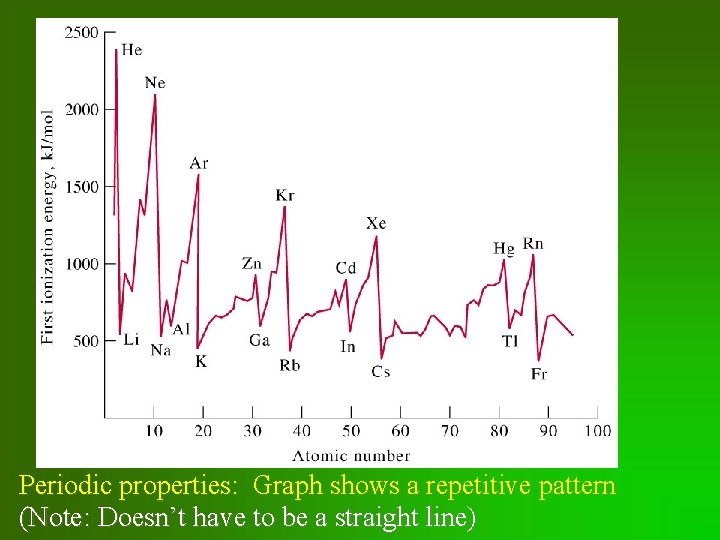
Periodic properties: Graph shows a repetitive pattern (Note: Doesn’t have to be a straight line)

Electronegativity • Relative ability of an atom to attract electrons in a bond • The noble gases tend not to form bonds, so they don’t have electronegativities • Unit = Pauling • Fluorine is the most electronegative element at 4. 0 Paulings

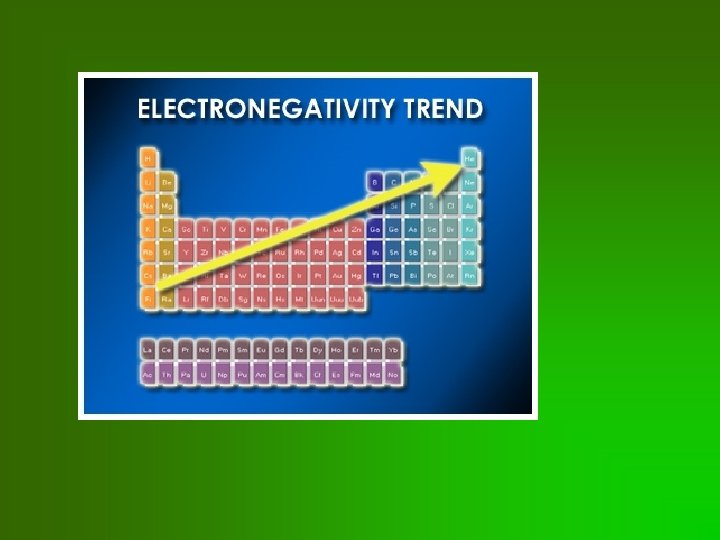
Trends in electronegativity • • Related to “proton pulling power” Increases as you go across a row Decreases as you go down a column Remember: F is the most electronegative element!

Reactivity of Metals • Metals are losers! • We judge the reactivity of metals by how easily they give up electrons • The most active metals are Cs and Fr • For metals, reactivity goes up as ionization energy goes down

Trends for Reactivity of Metals or Metallic Character • Increases as you go down – Easier to lose electrons! • Decreases as you go across – Harder to lose electrons!

Reactivity of Non-metals • Non-metals are winners! • We judge the reactivity of non-metals by how easily they gain electrons • The most active non-metal is fluorine • For non-metals, reactivity increases as electronegativity increases

Trend for Reactivity of Non-metals: Depends on “proton pulling power” • Increases as you go across • Decreases as you go down (Shielded by more inner-shell electrons)
- Slides: 14