Trends in the Periodic Table Atomic Mass Atomic

Trends in the Periodic Table Atomic Mass Atomic Radius Ionization Energy Electronegativity Density Electron Affinity
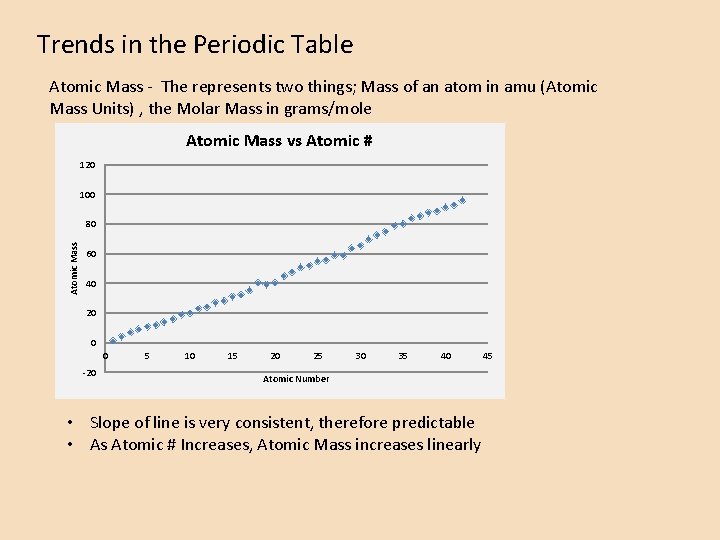
Trends in the Periodic Table Atomic Mass - The represents two things; Mass of an atom in amu (Atomic Mass Units) , the Molar Mass in grams/mole Atomic Mass vs Atomic # 120 100 Atomic Mass 80 60 40 20 0 0 -20 5 10 15 20 25 30 35 40 Atomic Number • Slope of line is very consistent, therefore predictable • As Atomic # Increases, Atomic Mass increases linearly 45
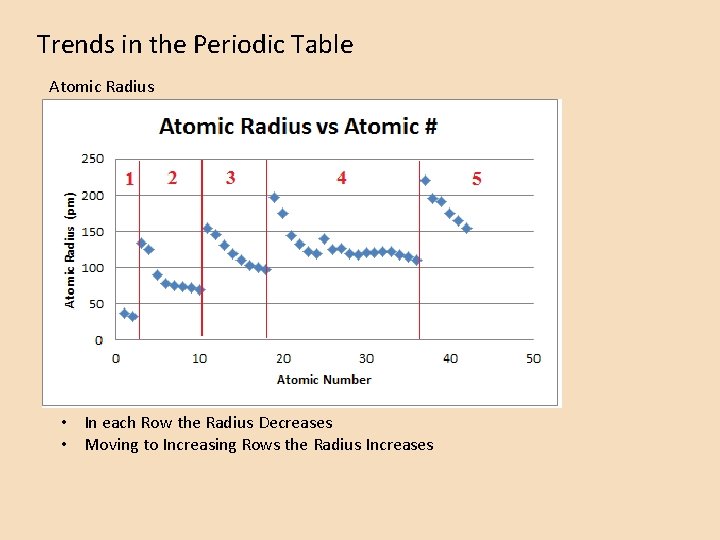
Trends in the Periodic Table Atomic Radius • In each Row the Radius Decreases • Moving to Increasing Rows the Radius Increases
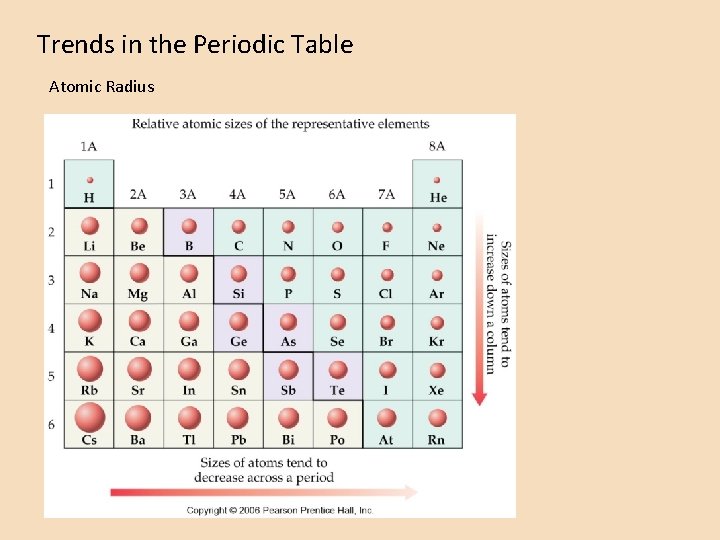
Trends in the Periodic Table Atomic Radius
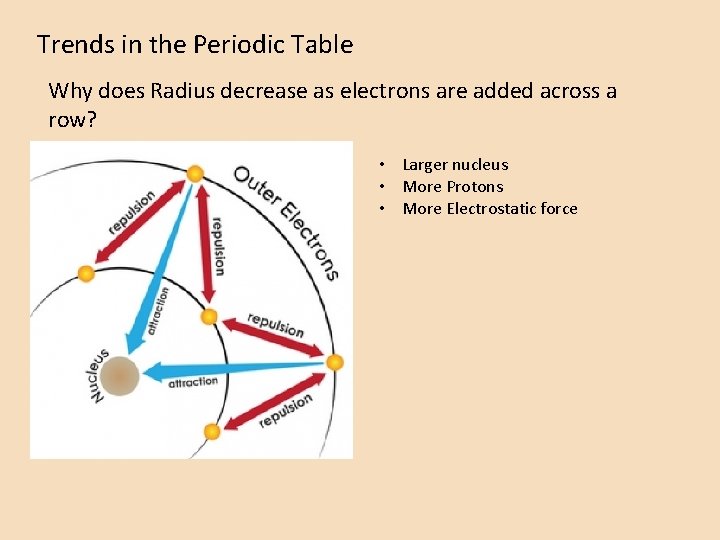
Trends in the Periodic Table Why does Radius decrease as electrons are added across a row? • Larger nucleus • More Protons • More Electrostatic force
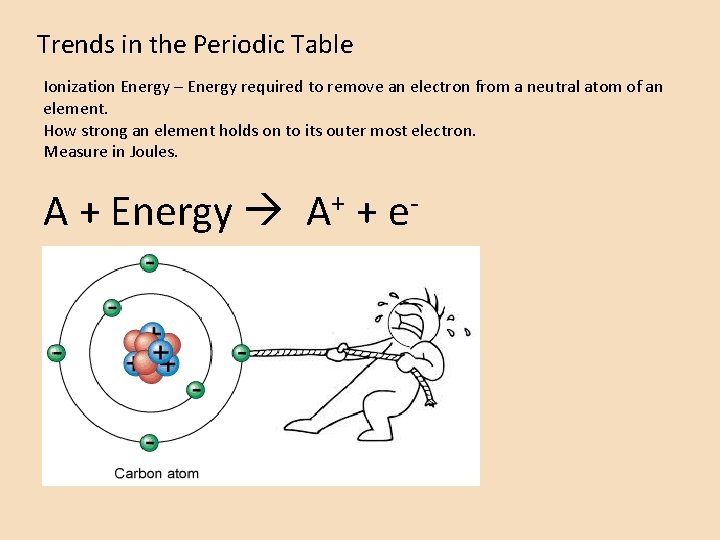
Trends in the Periodic Table Ionization Energy – Energy required to remove an electron from a neutral atom of an element. How strong an element holds on to its outer most electron. Measure in Joules. A + Energy + A + e
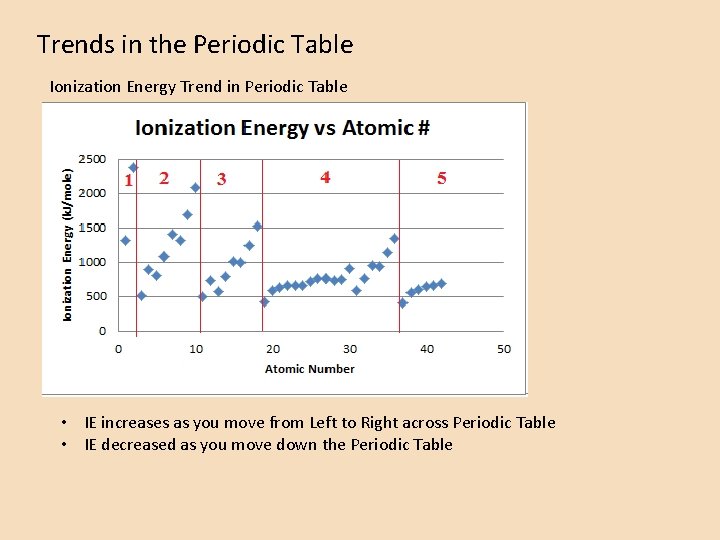
Trends in the Periodic Table Ionization Energy Trend in Periodic Table • IE increases as you move from Left to Right across Periodic Table • IE decreased as you move down the Periodic Table
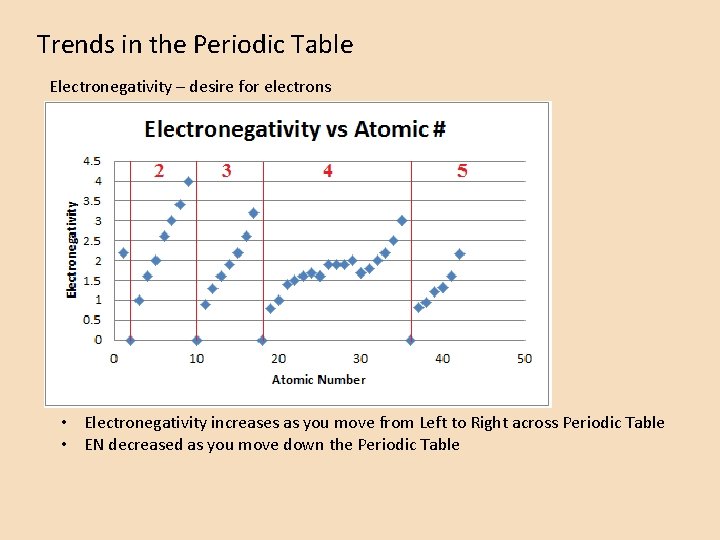
Trends in the Periodic Table Electronegativity – desire for electrons • Electronegativity increases as you move from Left to Right across Periodic Table • EN decreased as you move down the Periodic Table
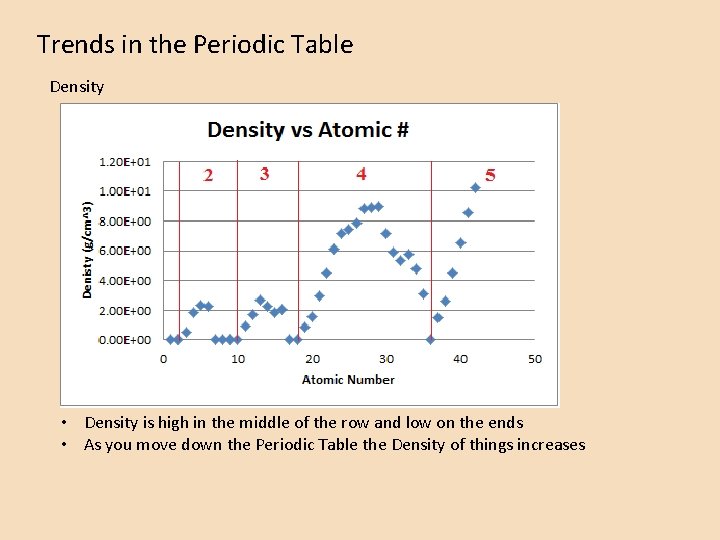
Trends in the Periodic Table Density • Density is high in the middle of the row and low on the ends • As you move down the Periodic Table the Density of things increases

Trends in the Periodic Table Electron Affinity – The energy change that occurs when an electron is acquired by a neutral atom. Most atoms give away energy when they acquire an electron. Energy given away is given the sign (-). A + e- A- + Energy Some atoms must be forced to gain an electron by the addition of Energy. A + Energy + e- A-
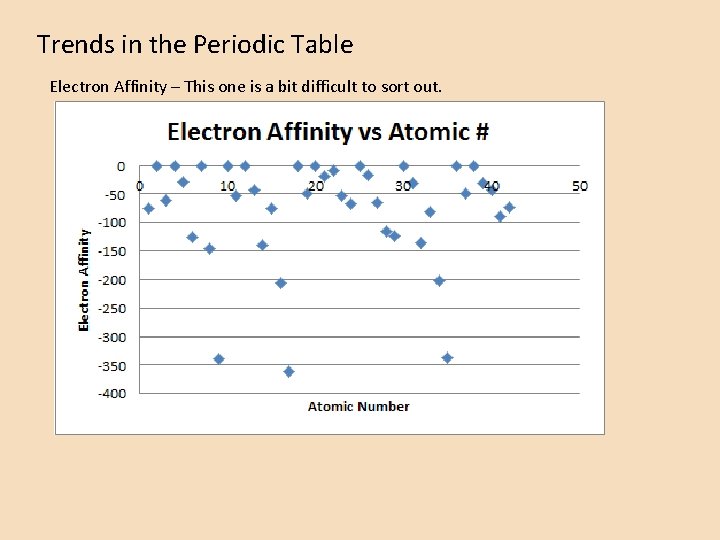
Trends in the Periodic Table Electron Affinity – This one is a bit difficult to sort out.
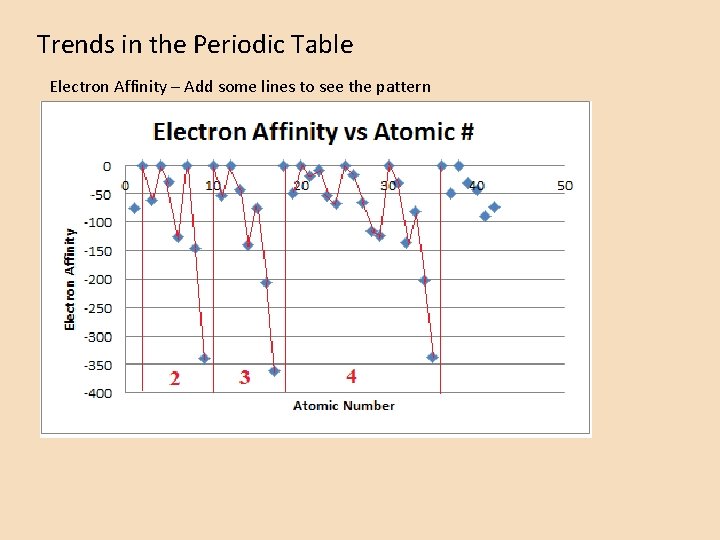
Trends in the Periodic Table Electron Affinity – Add some lines to see the pattern
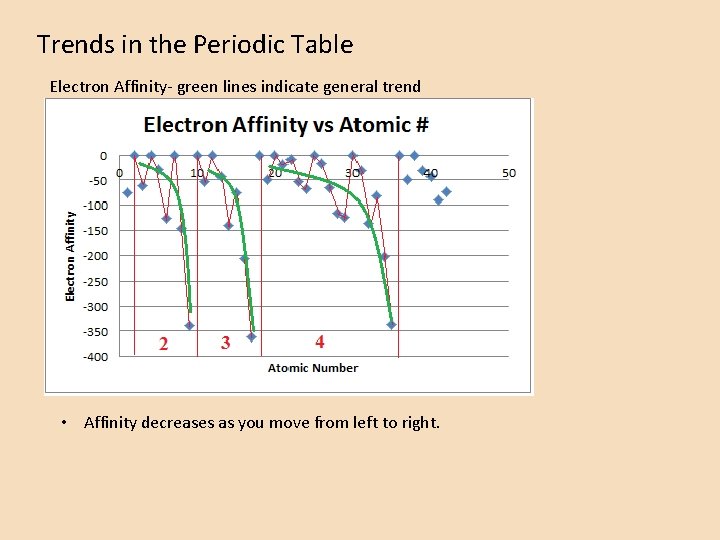
Trends in the Periodic Table Electron Affinity- green lines indicate general trend • Affinity decreases as you move from left to right.
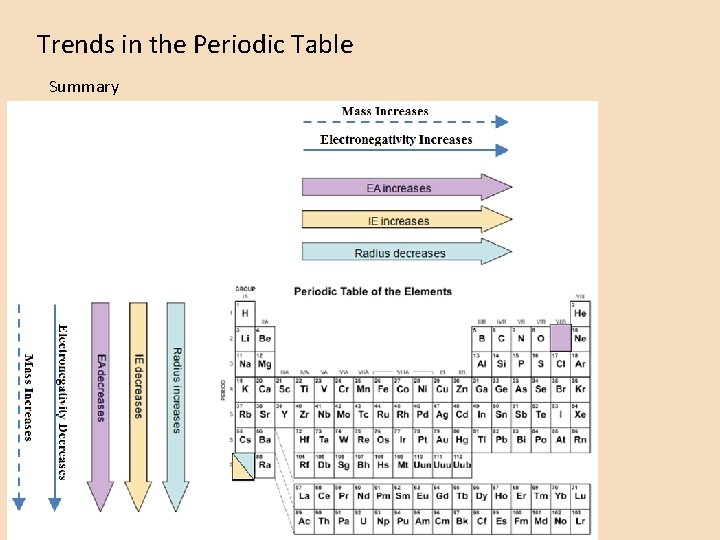
Trends in the Periodic Table Summary
- Slides: 14