Trends and Outcomes of OffLabel Use of Transcatheter

Trends and Outcomes of Off-Label Use of Transcatheter Aortic Valve Replacement (TAVR): Insights from the NCDR® STS/ACC TVT Registry. Ravi S. Hira, MD, FACC, FAHA, FSCAI Assistant Professor of Medicine, University of Washington Director, Acute Cardiac & Cardiac Catheterization Service, Harborview Medical Center

Disclosures • I have no relevant disclosures. • This research was supported by the American College of Cardiology Foundation’s National Cardiovascular Data Registry (NCDR) and The Society of Thoracic Surgeons National Database. The views expressed in this presentation represent those of the author(s), and do not necessarily represent the official views of either organization.

Background • • • TAVR was approved by the US FDA for treatment of severe aortic stenosis in symptomatic inoperable and high-risk patients. Edwards Sapien FDA approved in Nov 2011 Core. Valve FDA approved in Jan 2014

Background Not currently recommended for: • low surgical risk • bicuspid aortic valve • severe mitral regurgitation (MR) • moderate AS • severe aortic regurgitation (AR) • subaortic stenosis

Study objectives • Evaluate the frequency, temporal trends, and adverse outcomes associated with off-label TAVR use. • Also evaluate factors associated with off-label TAVR, hospital-level variation, and the association with outcomes.

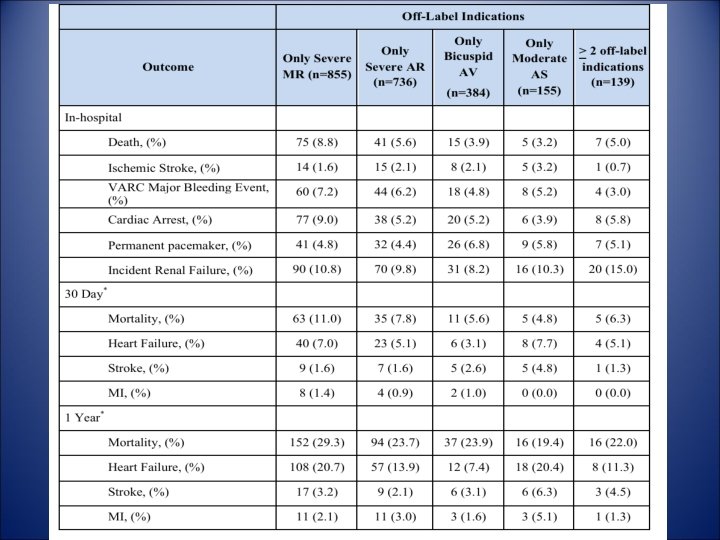
Methods • Patients receiving commercially funded TAVR in the US are included in the Transcatheter Valve Therapy (TVT) Registry launched in Nov 2011. • 23, 847 patients from 328 sites performing TAVR between 11/9/2011 -9/30/2014. • Off-label use defined as bicuspid or unicuspid valve, moderate AS (MG < 40 mm. Hg and AVA > 1 cm 2 when LV systolic function is > 55%) , severe mitral regurgitation, severe aortic regurgitation, or subaortic stenosis.

Methods • Exclusions - < 18 years of age, did not receive the Edward-Sapien or Core. Valve • We assumed that all patients met the NCD requirement i. e. deemed to be at high or severe risk for SAVR. • 30 -day and 1 -year adverse cardiovascular outcomes assessed in patients where CMS linked data was available (15, 397 patients)

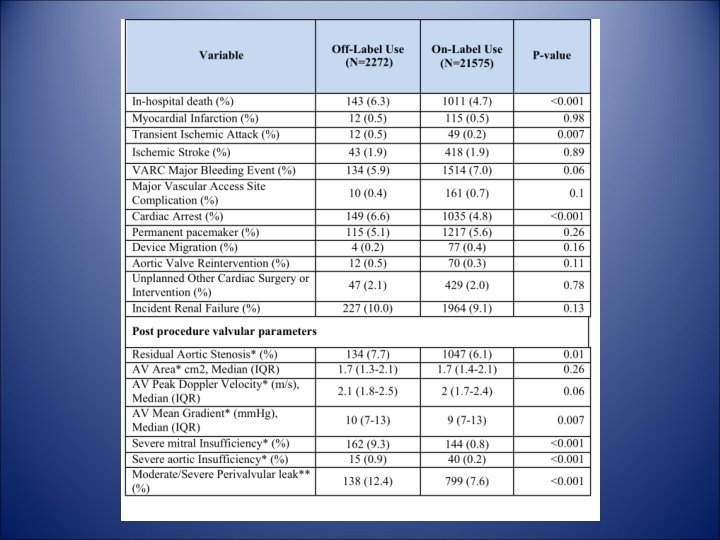
Primary outcomes 1. Frequency of off-label TAVR use in the registry 2. Association of off-label TAVR use with inhospital, and CMS-linked 30 -day and 1 -year mortality and adverse cardiovascular outcomes. Adverse outcomes were defined using standardized VARC and VARC-2 definitions

Statistical analysis • Frequency and quarterly frequency evaluated. • Baseline characteristics of off-label compared to on-label. • Proportional hazards models used to determine association between off-label TAVR use and adverse 30 -day and 1 -year outcomes.

Statistical analyses • Hospital variation was evaluated by calculating rates of off-label TAVR use for each hospital. • Hospitals were stratified into tertiles based on the proportion of off-label use. • Proportional hazard models used to determine association between hospital tertiles and 30 day and 1 -year composite of death, HF, MI, and stroke.

Results 9. 5% 75. 7% Severe MR and/or AI
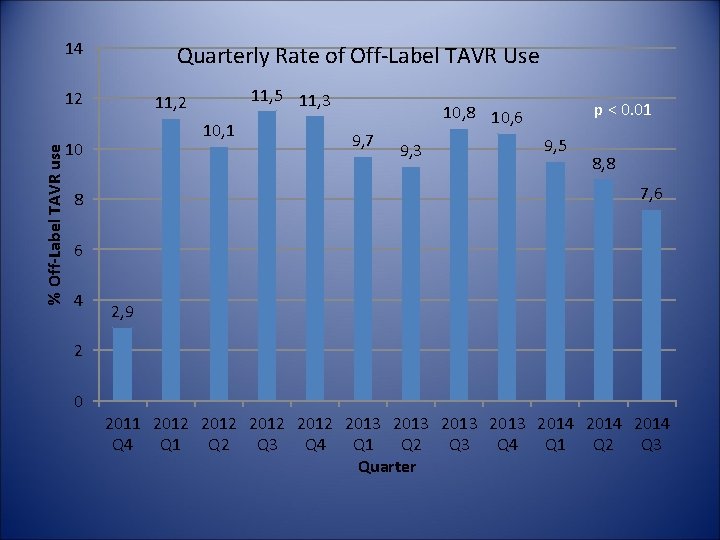
14 Quarterly Rate of Off-Label TAVR Use % Off-Label TAVR use 12 11, 5 11, 3 11, 2 10, 1 10 p < 0. 01 10, 8 10, 6 9, 7 9, 3 9, 5 8, 8 7, 6 8 6 4 2, 9 2 0 2011 2012 2013 2014 Q 1 Q 2 Q 3 Q 4 Q 1 Q 2 Q 3 Quarter
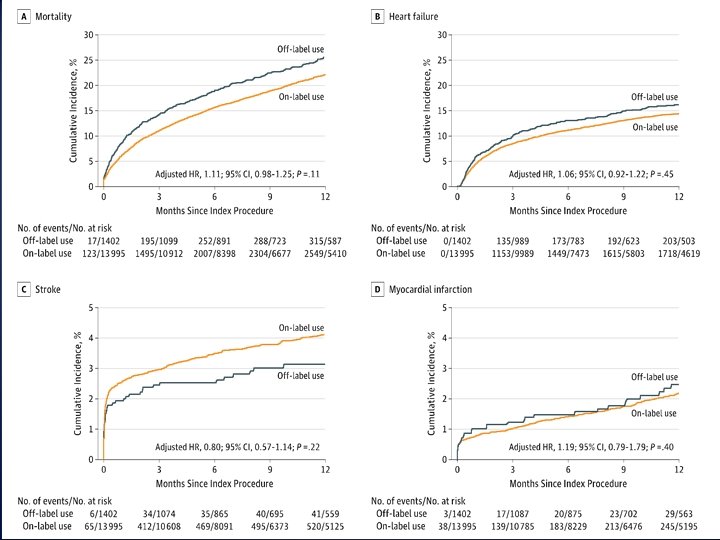
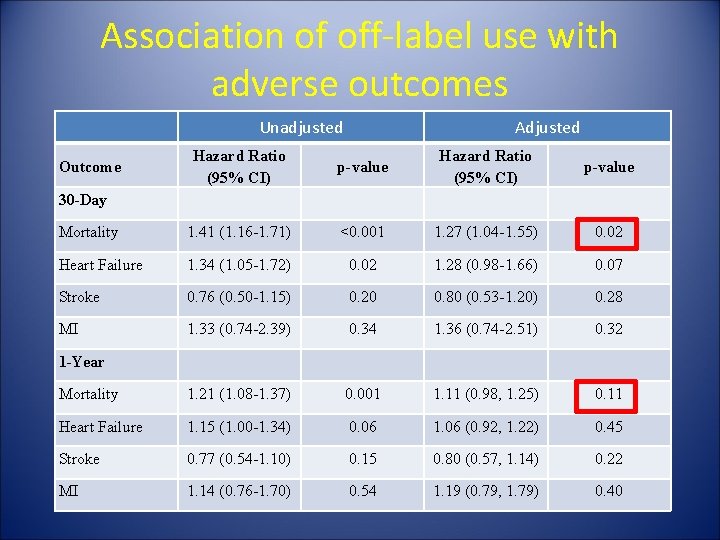
Association of off-label use with adverse outcomes Unadjusted Adjusted Hazard Ratio (95% CI) p-value Mortality 1. 41 (1. 16 -1. 71) <0. 001 1. 27 (1. 04 -1. 55) 0. 02 Heart Failure 1. 34 (1. 05 -1. 72) 0. 02 1. 28 (0. 98 -1. 66) 0. 07 Stroke 0. 76 (0. 50 -1. 15) 0. 20 0. 80 (0. 53 -1. 20) 0. 28 MI 1. 33 (0. 74 -2. 39) 0. 34 1. 36 (0. 74 -2. 51) 0. 32 Mortality 1. 21 (1. 08 -1. 37) 0. 001 1. 11 (0. 98, 1. 25) 0. 11 Heart Failure 1. 15 (1. 00 -1. 34) 0. 06 1. 06 (0. 92, 1. 22) 0. 45 Stroke 0. 77 (0. 54 -1. 10) 0. 15 0. 80 (0. 57, 1. 14) 0. 22 MI 1. 14 (0. 76 -1. 70) 0. 54 1. 19 (0. 79, 1. 79) 0. 40 Outcome 30 -Day 1 -Year
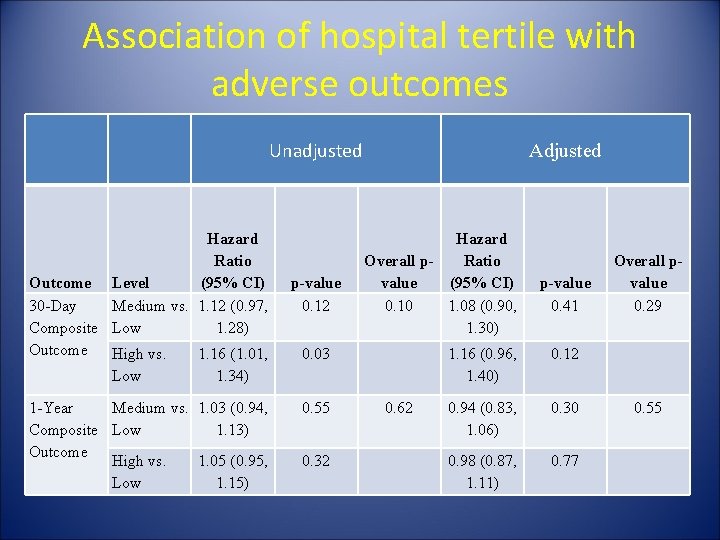
Association of hospital tertile with adverse outcomes Unadjusted Hazard Ratio Outcome Level (95% CI) 30 -Day Medium vs. 1. 12 (0. 97, Composite Low 1. 28) Outcome High vs. 1. 16 (1. 01, Low p-value 0. 12 Adjusted Hazard Ratio Overall pvalue (95% CI) 0. 10 1. 08 (0. 90, 1. 30) 0. 03 1. 34) 1 -Year Medium vs. 1. 03 (0. 94, Composite Low 1. 13) Outcome High vs. 1. 05 (0. 95, Low 1. 15) 0. 55 0. 32 0. 62 p-value 0. 41 1. 16 (0. 96, 1. 40) 0. 12 0. 94 (0. 83, 1. 06) 0. 30 0. 98 (0. 87, 1. 11) 0. 77 Overall pvalue 0. 29 0. 55

Limitations • Retrospective and cannot exclude confounding. • Majority with severe MR or AR which are often load-dependent and may change. • No core laboratory assessment was performed. • Labeling of the Edwards-Sapien and Core. Valve systems may be different from other countries. • Analysis based on valve type was not conducted. • Safety, efficacy, and cost effectiveness of TAVR compared to alternative therapies including SAVR or medical therapy could not be performed.

Conclusions • Approximately 1 in 10 patients in the US have received TAVR for an off-label indication. • After adjustment, 30 -day mortality was higher but 1 -year mortality was similar to patients with on-label TAVR use.

- Slides: 20