TREATMENT PRODUCTS IN HEMOPHILIA Nairobi Kenya June 24

TREATMENT PRODUCTS IN HEMOPHILIA Nairobi, Kenya June 24, 2013

OBJECTIVES • Identify historical approaches used to treat hemophilia • Describe treatment products currently available for use in hemophilia • Distinguish classes of factor concentrates • Discuss donor screening and viral inactivation • List adjuvant therapies for treatment of hemophilia • Explore future therapies ADDITIONAL TEXT EXAMPLE

HISTORICAL TREATMENT OF HEMOPHILIA • Injections of adrenaline • Ingestion of such compounds as 1: − Strychnine − Turpentine − Lead − Female hormone − Bromide extracts of egg whites − Peanut flour 2 • Topical snake venom 3 • First blood transfusion in 1840 by Dr. Samuel Lane 4 1 Rosendaal FR, Smit C, Briët E. Ann Hematol. 1991; 62: 5 -15. 2 Mainwaring D, Keldon S. E. Lancet. 1964; 19: 647. 3 Mac. Farlane RG, Barnett, B. Lancet. 1934; ii: 985– 987. 4 Lane, S. Lancet. 1840; i: 185 -188.

CRYOPRECIPITATE DISCOVERED 1965: Discovery of cryoprecipitate Judith Graham Pool, MD File photo courtesy of HANDI, NHF Pool JG, Shannon AE. N Engl J Med. 1965: 273: 1443 -1447.

FACTOR CONCENTRATES SOON APPEAR 1966: Hyland announces commercial availability of FVIII concentrates 1969: FIX concentrate licensed 1 Allowed for greater independence 1. Hoag MS, et al. N Engl J Med. 1969; 280(11): 581 -6

THE PRICE OF INDEPENDENCE 1983: Suspicion that HIV threatened the worldwide blood supply 1983: Hemofil-T, first heat-treated FVIII concentrate in the US 1984: Montagnier 1 and Gallo 2 discover HTLV-3 (HIV) 1984: Efficacy of heat treatment for viral inactivation demonstrated 1984: Recall of blood products initiated 1985: ELISA test used to detect HIV antibodies among blood donors 1985: Safety net: 1. Donor deferral 2. Viral inactivation methods 3. Antibody and NAT testing 1. Barre-Sinoussi F, et al. Science 1983; 220(4599): 868 -71. 2. Gallo RC, et al. Science 1984; 4; 224(4648): 500 -3.

CLOTTING FACTOR CONCENTRATES AND OTHER PLASMA PRODUCTS Factor replacement therapy • Fresh frozen plasma (FFP) • Cryoprecipitate • Plasma-derived concentrates • Recombinant concentrates

WFH RECOMMENDATION The WFH strongly recommends the use of viral-inactivated plasma-derived or recombinant concentrates in preference to cryoprecipitate or fresh frozen plasma for the treatment of hemophilia Guidelines for the Management of Hemophilia, 2 nd edition, WFH 2012

CHOICE OF TREATMENT PRODUCT • • Choice of treatment product is an important decision • Important issues regarding infusion products: Infusion products should be chosen with provider, NMO, and patient/family input − − Efficacy Safety Purity Cost

PLASMA-DERIVED PRODUCTS: PURITY Concentrates on the market vary widely in their purity ● High purity − − ● Intermediate purity − − ● ● ● Just the clotting factor in the vial exclusive of added stabilizers Activity/protein ratio is very high More than just the clotting factor in the vial Activity/protein ratio mid-range Concentrates of lower purity may give rise to allergic reactions FVIII concentrates may contain variable amounts of VWF For treatment of FIX deficiency, a product containing only FIX is more appropriate than PCCs

PLASMA-DERIVED PRODUCTS: SAFETY Viral inactivation is the biggest contributor to safety of treatment products ● ● Heat treatment: effective against enveloped and nonenveloped viruses including (HIV, HAV, HBV and HBC) Solvent/detergent treatment: effective against nonenveloped viruses such as HIV, HBV, HCV but not HAV) Some viruses resistant to both types of process (e. g. parvovirus B 19) Products undergo one or two viral inactivation steps; if one, preferably one that is effective against viruses with and without lipid envelopes

RECOMBINANT PRODUCTS ● All recombinant products are high purity ● Not made from human plasma ● 1 st generation: Added albumin as stabilizer, human/animal protein exposure during production ● 2 nd generation: Albumin removed as stabilizer, human/animal protein exposure during production ● 3 rd generation: No added human or animal protein during production or in final formulation

FRESH FROZEN PLASMA • Contains all the coagulation factors • Due to concerns about safety and quality of FFP, it is not recommended for treatment of hemophilia, if avoidable • Possible to apply some forms of viral inactivation to packs of FFP but may have impact on coagulation factors • Large volumes of plasma must be transfused, which can lead to a complication called circulatory overload

CRYOPRECIPITATE • Prepared by slow thawing of FFP • Contains significant quantities of FVIII, VWF, fibrinogen and FXIII but no FIX • Less safe from viral contamination than factor concentrates; harder to store and administer • Virally-inactivated cryo has been described (S/D cryo) • Preferable to FFP for the treatment of hemophilia A • Cryo cannot be used for treatment of hemophilia B
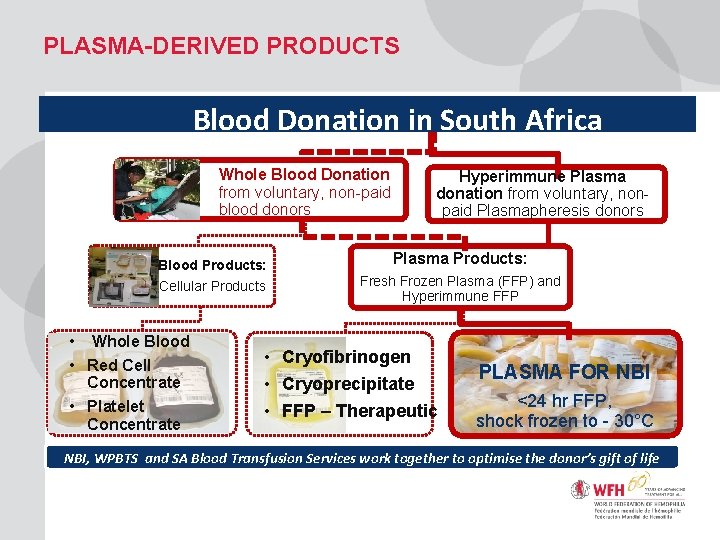
PLASMA-DERIVED PRODUCTS Blood Donation in South Africa Whole Blood Donation from voluntary, non-paid blood donors Blood Products: Cellular Products Hyperimmune Plasma donation from voluntary, nonpaid Plasmapheresis donors Plasma Products: Fresh Frozen Plasma (FFP) and Hyperimmune FFP • Whole Blood • Cryofibrinogen • Red Cell ADDITIONAL TEXT EXAMPLE Concentrate • Cryoprecipitate • Platelet • FFP – Therapeutic Concentrate PLASMA FOR NBI <24 hr FFP, shock frozen to - 30°C NBI, WPBTS and SA Blood Transfusion Services work together to optimise the donor’s gift of life

HEMOPHILIA A: EVOLUTION OF THERAPY Year Therapy 1950 Plasma (1 -FVIII U/ml) 1966 Cryoprecipitate (5 -FVIII U/ml) 1975 Lyophilized concentrates (30 -FVIII U/ml) 1983 Heat treatment of lyophilized concentrates for viral attenuation 1985 Introduction of a solvent detergent for viral inactivation 1988 Monoclonal antibody-purified FVIII concentrates with heat or solvent detergent treatment 1992 Recombinant DNA products: 1 st generation 2000 s 2 nd & 3 rd generation recombinant products

HEMOPHILIA B: EVOLUTION OF THERAPY Year Therapy 1950 Plasma (1 -FIX U/ml) 1975 Lyophilized prothrombin complex concentrates or PCC (30 -FIX U/ml) 1985 Heat treatment of lyophilized concentrates for viral attenuation Development of a solvent detergent for viral inactivation 1992 Chromatographic/monoclonal antibody-purified FIX concentrate with ultrafiltration and thiocyanate treatment 1997 Recombinant DNA product
![FACTOR REPLACEMENT PRODUCTS • FVIII products – – – – Advate [r-3 rd gen] FACTOR REPLACEMENT PRODUCTS • FVIII products – – – – Advate [r-3 rd gen]](http://slidetodoc.com/presentation_image_h2/69f43f8d47113dba6e2cc9d6ce0f406f/image-18.jpg)
FACTOR REPLACEMENT PRODUCTS • FVIII products – – – – Advate [r-3 rd gen] Xyntha [r-3 rd gen] Kogenate FS [r-2 nd gen] Helixate FS [r-2 nd gen] Recombinate [r-1 st gen] Hemophil-M [pd-HP] Monoclate-P [pd-HP] • VWF products • FIX products – – Bene. FIX [r-3 rd gen] Mononine [pd-HP] Profilnine [pd-PCC] Bebulin [pd-PCC] • Bypassing agents – Novo Seven [r-3 rd gen] – FEIBA [pd-APCC] – Humate-P [pd-HP] – Alphanate [pd-HP] – Wilate [pd-HP] http: //www. hemophilia. org/research/masac 151. htm MASAC document #151 & 106

OTHER TREATMENT PRODUCTS Desmopressin • Boosts plasma levels of FVIII and VWF • Does not affect FIX levels • May be treatment of choice for patients with mild or moderate hemophilia A and carriers • Lower cost than plasma products and no risk of viral transmission • Test patient response prior to use • Administration: - IV - SQ - Intranasal

OTHER TREATMENT PRODUCTS Antifibrinolytic agents • Promote clot stability • Useful as adjunctive therapy, particularly for skin and mucosal bleeding, e. g. oral bleeding, epistaxis, menorrhagia • Tranexamic acid available orally, IV, mouthwash • Epsilon aminocaproic acid (EACA) similar but less widely used • Do NOT use in patients with hemophilia B treated with PCCs
OTHER TREATMENT PRODUCTS Hormone therapy (women) • OCPs • IUDs Topical hemostatic agents • Fibrin sealant (fibrin glue) Replacement • Iron • Vitamin D

FUTURE TREATMENT THERAPIES FOR HEMOPHILIA • • Longer acting concentrates Recombinant therapy for VWD Alternate route therapies Gene transplantation Elimination of transfusion-associated infections Understanding and overcoming inhibitor development Quality of life issues − Elimination of joint morbidity − Optimizing the individual’s social and academic performance

SUMMARY • Treatment in hemophilia continues to progress • FFP and cryoprecipitate are still used in many parts of the world • Replacement therapies are available in a variety of forms • Choosing a factor concentrate is important to all involved in care • Efficacy, safety and cost remain important considerations when choosing a treatment product • Adjuvant therapies are available to assist in hemophilia treatment • The future of hemophilia treatment appears promising

WFH RESOURCES • Guide for the Assessment of Clotting Factor Concentrates • Registry of Clotting Factor Concentrates • Fibrinolytic Inhibitors in the Management of Bleeding Disorders • Desmopressin (DDAVP) in the Treatment of Bleeding Disorders • Guidelines for the Management of Hemophilia, 2 nd ed
- Slides: 24