Treatment of CNS Degenerative Disorders By Imad M

ﺑﺴﻢ ﺍﻟﻠﻪ ﺍﻟﺮﺣﻤﻦ ﺍﻟﺮﺣﻴﻢ Treatment of CNS Degenerative Disorders By Imad M. Taj Eldin Faculty of Pharmacy 1

CNS Degenerative Disorders v. The central nervous system degenerative disorders are: ØParkinson’s disease (PD) ØAlzheimer’s disease (AD) ØHuntington’s disease (HD) and others v. They are characterized by progressive irreversible loss of neurons from specific regions of the brain. v. They are primarily disorders of later life. v. Parkinson’s disease (PD) is observed in more than 1% of individuals over the age of 65. v Alzheimer’s disease (AD) affects as many as 10% of the same population 2

CNS Degenerative Disorders v. Huntington’s disease (HD), which is a genetically determined autosomal dominant disorder. v. It is less frequent in the population as a whole but affects, on average, 50% of each generation in families carrying the gene. v. The pharmacotherapy of these neurodegenerative disorders is limited to symptomatic treatments that do not alter the course of the underlying disease. v. These disease are specific for particular types of neurons. ØIn Parkinson’s disease (PD) there are extensive destructions of the dopaminergic neurons of the substantia nigra. ØIn Alzheimer’s disease (AD), the neuronal injury is most severe in the hippocampus and neocortex ØIn Huntington’s disease (HD), the pathological changes are most prominent in the neostriatum. 3

Pharmacology of Parkinson’s Disease Overview v. Sometimes it is called paralysis agitans or shaking palsy. v. Parkinson’s disease or Parkinsonism, was first described by Dr. Parkinson in 1817. v. Most of the cases involve people over the 65 years between whom the incidence is about 1: 100. v. It is a progressive neurological disorder of muscle movement and it is characterized by: ØAn involuntary tremor of resting muscles and extremities. ØRigidity of muscles. ØHypokinesia (bradykinesia): it is slowness in initiating and carrying out voluntary movement and daily activities. 4

Pharmacology of Parkinson’s Disease v. It is also characterized by: ØPostural and gait abnormalities and backward falling. ØMasked facial expression. ØDysfunction of the autonomic nervous system: impaired gastrointestinal motility, bladder dysfunction, excessive head and neck sweating, and orthostatic hypotension ØAssociated with dementia ØDecrease in size of handwriting ØDifficulty turning over in bed, ØDifficulty getting out of a chair ØSlow thinking and slowed chewing and swallowing.

Pharmacology of Parkinson’s Disease Etiology v. The cause of Parkinson’s disease is unknown (idiopathic). v. This is called primary Parkinson’s disease (or type 1). v. The disease is correlated with a reduction of in the activity of inhibitory dopaminergic neurons in the substantia nigra and corpus striatum parts of the brain’s basal ganglia system that are responsible for motor control. v. The normally high concentration of dopamine in the basal ganglia of the brain is reduced in Parkinsonism. v. The substantia nigra is the source of dopaminergic neurons that terminate in the striatum. v. The corpus striatum is exceptionally rich in acetylcholine as well as dopamine v. Acetylcholine has excitatory effects whereas dopamine is mainly inhibitory, and the symptoms of Parkinsonism result from an imbalance between these two systems. 6
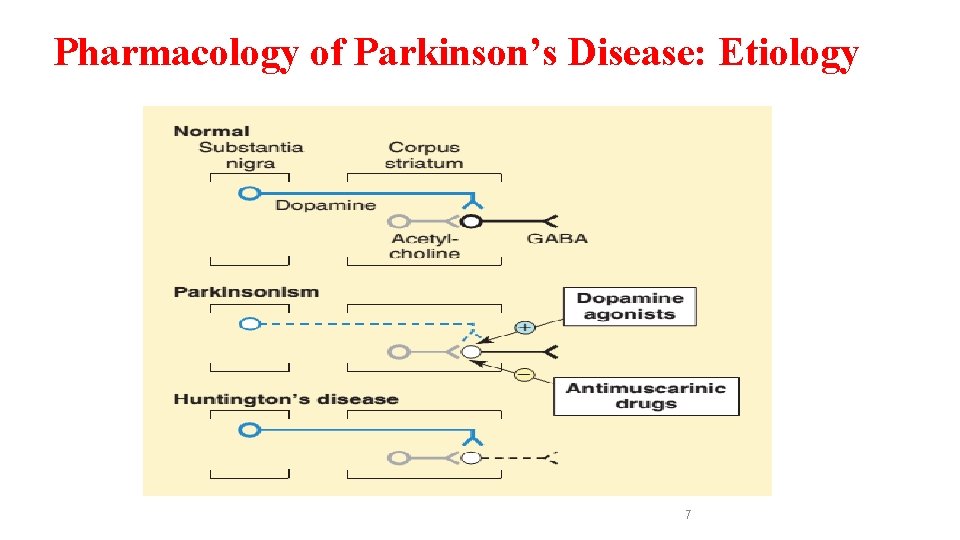
Pharmacology of Parkinson’s Disease: Etiology 7

Pharmacology of Parkinson’s Disease Etiology v. The secondary Parkinsonism often occurs with no obvious underlying cause, but it may be the result of cerebral ischemia, viral encephalitis or other types of pathological damage such as magnesium and carbon dioxide poisoning or by MPTP (1 -methyl -4 -phenyl, 1, 2, 5, 6 -tetrapyridine) which causes damage of the dopaminergic neuron. v. Also Parkinsonism can be one of the side effects of some drugs e. g. antipsychotic drug v. There is also familial Parkinson's disease due to genetic factors. 8
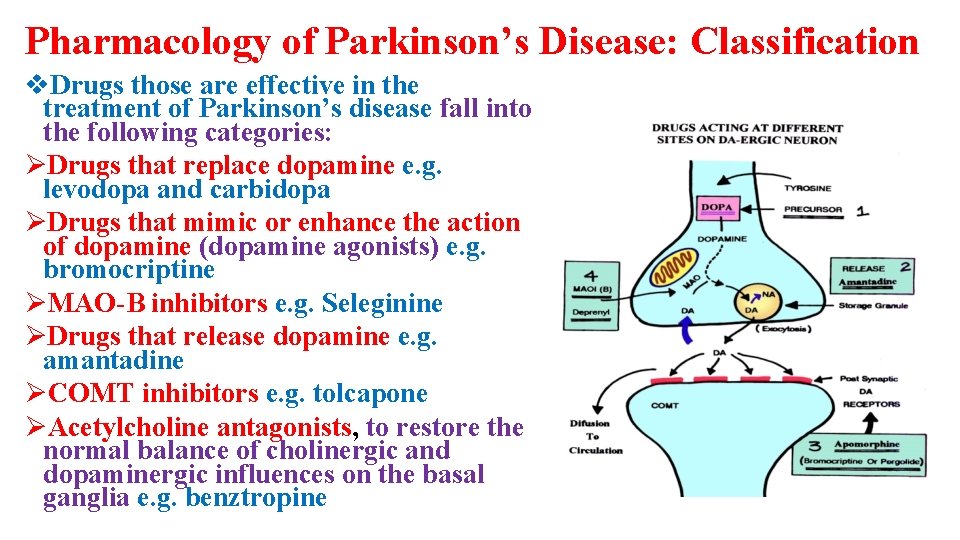
Pharmacology of Parkinson’s Disease: Classification v. Drugs those are effective in the treatment of Parkinson’s disease fall into the following categories: ØDrugs that replace dopamine e. g. levodopa and carbidopa ØDrugs that mimic or enhance the action of dopamine (dopamine agonists) e. g. bromocriptine ØMAO-B inhibitors e. g. Seleginine ØDrugs that release dopamine e. g. amantadine ØCOMT inhibitors e. g. tolcapone ØAcetylcholine antagonists, to restore the normal balance of cholinergic and dopaminergic influences on the basal ganglia e. g. benztropine

Pharmacology of Parkinson’s Disease v. There is no cure for Parkinsonism. v. The currently available drugs offer temporary relief from the symptoms of the disorder, but not arrest the neuronal degeneration caused by the disease. Therapeutic goal ØBecause it is not yet possible to reverse the degenerative process, drugs are used to increase dopamine activity or ØTo reduce excitatory interneuron acetylcholine activity, in order to restore their balance in the corpus striatum 10

Pharmacology of Parkinson’s Disease 1. Drugs That Replace Dopamine v. These are L-dopa and carbidopa and L-dopa and beserazide v. Levodopa is the most reliable and effective drug used in the treatment of Parkinsonism. v. Considered as a form of replacement therapy. v. Levodopa is a precursor of dopamine and is used to elevate dopamine level in extrapyramidal center (substantia nigra) of the parkinsonian patients. v. Dopamine itself does not cross the blood brain barrier (BBB)? ? ? . v. However levodopa is an amino acid, is converted to dopamine by the enzyme dopa decarboxylase (L-aromatic amino acid decarboxylase)

Pharmacology of Parkinson’s Disease Levodopa and carbidopa v. When levodopa alone is used for the treatment of Parkinsonism most of it does not reach its intended site of action in the brain, because of its peripheral metabolism to dopamine by dopa decarboxylase in the liver, kidney, and GIT. v. This decarboxylation can be markedly reduced if levodopa is given in combination with peripheral decarboxylase inhibitor (carbidopa). v. However it is important that the peripheral decarboxylase inhibitor not enter the brain so that the needed decarboxylation of dopa to dopamine can occur at the site of its therapeutic action. v. This is achieved through the administration of the decarboxylase inhibitor, carbidopa, which is ionized at physiological p. H and therefore cannot readily penetrate the blood brain barrier. 12

Pharmacology of Parkinson’s Disease Levodopa and carbidopa v. The combination of levodopa-carbidopa has the following advantages: ØCarbidopa diminishes the metabolism of levodopa in the G. I. T and peripheral tissues, thus it increases the availability of levodopa to the CNS. ØThe addition of carbidopa lowers the dose of levodopa by 4 to 5 folds ØConsequently decreases the severity of the side effects of the peripherally formed dopamine (such as nausea, vomiting and cardiovascular effects) and it increases the availability of levodopa to the CNS. 13

Pharmacology of Parkinson’s Disease Mechanism of action v. Levodopa is a levorotatory isomer of dopa that is presynaptically decarboxylated in the CNS to dopamine in the corpus striatum, where it interacts with postsynaptic dopamine D 2 - and D 3 -receptors to activate inhibitory G proteins (Gi), inhibit adenylyl cyclase, decrease c. AMP levels, and open potassium channels

Pharmacology of Parkinson’s Disease Action if Levodopa v. Levodopa decreases the rigidity, tremors and other symptoms of Parkinsonism. v. Improvement of rigidity and bradykinesia are generally more complete and appear sooner than do improvement in tremor v. Levodopa is less effective in eliminating postural instability and shuffling gait Absorption and Metabolism v. The drug is absorbed rapidly from the small intestine (when empty of food) metabolized by the actions of MAO and COMT vlevodopa has an extremely short half- life (1 to 2 hours), which causes fluctuation in motor response, (on-off phenomenon). v. This will cause the patient to suddenly lose motility and experience tremor, crams, and immobility 15

Pharmacology of Parkinson’s Disease Therapeutic Uses v. Levodopa in combination with carbidopa is a potent and efficacious drug regimen currently available to treat Parkinsonism in approximately two third of patients with Parkinson’s disease. v. Levodopa/ carbidopa treatment substantially reduces the severity of the disease for the first few days of treatment. v. Patients then typically experience a decline in response during the third to fifth years of therapy. The dose v. The most appropriate carbidopa/levodopa preparation for initial therapy is the 25/100 mg formulation. v. This allows for enough of carbidopa to block the peripheral decarboxylase (usually 75 - 100 mg /day are required) low doses of (50/200/day in two divided doses) should be used for the first few days to avoid orthostatic hypotension. v. A maintenance dosage of 75/300/day may be effective 16 for year or more

Pharmacology of Parkinson’s Disease Adverse Effects v. G. I. T effects: anorexia, nausea and vomiting may occur v. Cardiovascular effects: cardiac arrhythmia (tachycardia and ventricular extrasystoles). Postural hypotension is common? ? ? v. Dyskinesia: it is a variety of repetitive involuntary abnormal movement of limbs, hands, trunk, and tongue. v. CNS effects: These are depression, anxiety, agitation, insomnia, confusion, hallucination, nightmares and other changes in mood. v. Fluctuation in response: this is called on-off phenomenon. In a matter of minutes, a patient enjoying normal or near normal mobility and may suddenly develop a severe degree of Parkinsonism (akinesia, a sudden freezing of movement). 17

Pharmacology of Parkinson’s Disease Drug Interactions v. The therapeutic action of levodopa is reduced by antiemetic or antipsychotic drugs that block dopamine receptors. v. Natural aromatic amino acids (tryptophan, histidine, leucine, phenylalanine, tyrosine) decrease the absorption of levodopa. v. Levodopa should not be used with MAO-A inhibitors. v. This combination cause a severe hypertensive crisis. v. In the absence of carbidopa, peripheral levels of levodopa are decreased by pyridoxine (vitamin B 6), which increases the activity of dopa decarboxylase and increases conversion of levodopa to dopamine in the periphery. 18

Pharmacology of Parkinson’s Disease Contraindications v. Carbidopa/levodopa preparation are contraindicated in: ØPsychotic patients. ØPatients with a history of cardiac arrhythmias. ØPatients with active peptic ulcer disease. ØPatients with narrow-angle glaucoma v. The use of levodopa may produce mydriasis and precipitation of an attack of acute glaucoma. This due to adrenergic action on iris. v. Brownish discoloration of saliva, urine or vaginal secretion, because of melanine pigmentation produces from catecholamine oxidation ( additional adverse effect) 19

Pharmacology of Parkinson’s Disease 2. Dopamine Agonists The dopamine agonists are two types: v. Ergot derivatives like bromocriptine, apomorphine, pergolide and lisuride v. Nonergot derivatives such as pramipexole and ropinirole Ergot Dopamine Agonists v. Bromocriptine: is a potent agonist at dopamine D 2 -repetoers in the CNS and a weak partial agonist at D 1 -receptors v. The activity of bromocriptine in D 2 -receptors is thought to underline its efficacy in Parkinson’s disease. v. It was first introduced for the treatment of hyperprolactinemia and gynecomastia. 20

Pharmacology of Parkinson’s Disease Ergot Dopamine Agonists v. Pergolide and lisuride: directly stimulates D 1 and D 2 recepetors v More active than bromocriptine in relieving the symptoms and signs of Parkinsonism v. The ergot derivatives often induce profound hypotension, nausea and fatigue, so they should be initiated at low dosage. Adverse effects v. These drugs have the same adverse effects, cautions, and contraindications as levodopa, although the severity of their effects may differ 21

Pharmacology of Parkinson’s Disease Ergot Dopamine Agonists v. Apomorphine: is administered by subcutaneous injection. v. It has high affinity for D 4 receptors v. Apomorphine is highly emetogenic and requires pre- and posttreatment with antiemetic therapy. v. Also apomorphine induces QT prolongation and development of a pattern of abuse. 22

Pharmacology of Parkinson’s Disease Nonergot Dopamine Agonists v. Pramipexole and ropinirole are newer dopamine agonists and both are potent antiparkinsonian agents v. Pramipexole has preferential affinity for D 3 family of receptors v. It is effective when used as monotherapy for mild parkinsonism or given in combination with levodopa for optimal treatment. v. Ropinirole is relative pure D 2 -receptor agonist. v. It is effective as monotherapy in-patient with mild disease v. Pramipexole is excreted unchanged; dosage adjustments may be necessary in patients with renal insufficiency. v. Ropinirole, which is also approved to treat restless leg syndrome v. These drugs have the same adverse effects, cautions, and contraindications as levodopa 23

Pharmacology of Parkinson’s Disease 3. Monoamine Oxidase Inhibitors v. Two types of monoamine oxidase have been distinguished. v. Monoamine oxidase A (MAO-A) metabolized noradrenaline and serotonin; monoamine oxidase B (MAO-B) metabolizes dopamine. v. Selegiline (deprenyl) and rasagiline are selective inhibitor of monoamine oxidase-B that decrease dopamine metabolism in the CNS and prolong its action. v. It is used as adjunctive therapy for patient with a declining or fluctuation response to levodopa. v. Rasagiline is more potent than seleginine in preventing MPTPinduced Parkinsonism. v. The combined administration of levodopa and inhibitors of monoamine oxidase must be avoided, since it may lead to hypertensive crises, probably because of the peripheral accumulation of noradrenaline. 24

Pharmacology of Parkinson’s Disease 4. Drug that release dopamine v. Amantadine, an antiviral agent, was discovered by chance, found to have antiparkinsonism properties. v. Its mode of action in Parkinsonism is unclear, but it may potentiate dopaminergic function by influencing the synthesis, release or inhibiting the uptake of dopamine. v. Release of catecholamines from peripheral has been documented. v. Amantadine is less effective than levodopa or bromocriptine v. Adverse effects v. Amantadine is associated with a reversible occasional headache, insomnia, confusion, hallucinations, and peripheral edema. v. Long-term use of amantadine may lead to reversible discolouration of the skin (livedo reticularis) or, more rarely, congestive heart failure. v. Overdose may cause a toxic psychosis and seizures. 25

Pharmacology of Parkinson’s Disease 5. COMT inhibitors v. Inhibition of dopa decarboxylase is associated with compensatory activation of other pathways of levodopa metabolism, especially catechol-o-methyltransferase (COMT), and this increases plasma level of 3 -o-methyldopa (3 OMD). v. Elevated levels of 3 OMD have been associated with a poor therapeutic response to levodopa, perhaps in part because 3 OMD competes with levodopa for an active carrier mechanism that governed its transport across intestinal mucosa and the blood brain barrier. v. Selective COMT inhibitors, tolcapone and entacapone prolong the duration of levodopa by diminishing its peripheral metabolism. v. The pharmacological effects of tolcapone and entacapone are similar

Pharmacology of Parkinson’s Disease v. Tolcapone has both central and peripheral effects, whereas the effect of entacapone is peripheral v. The half-life of both drugs is approximately 2 hours, but tolcapone is slightly more potent and has longer duration of action. v. Entacapone is generally preferred because it has not been associated with hepatotoxicity. v. Adverse effects of the COMT inhibitors related in part to increased levodopa exposure and include dyskinesia, nausea, and confusion. v. Other side effects include diarrhea, abdominal pain, orthostatic hypotension, sleep disturbance, and orange discoloration of urine. v. Tolcapone may cause hepatotoxicity with increase in the liver enzyme level. 27

Pharmacology of Parkinson’s Disease 6. Acetylcholine antagonists v. They were used widely for the treatment of Parkinson’s disease before the discovery of levodopa v. Centrally acting antimuscarinic agents are benztropine, biperiden, procyclidine, and trihexyphenidyl (benzhexol). v. All have modest antiparkinsonian activity that is useful in the treatment of early Parkinson’s disease or as an adjunct to dopamimetic therapy. v. Antimuscarinic agents improve tremor and rigidity of Parkinsonism, but have little effects on bradykinesia. v. Side effects including dryness of the mouth, blurred vision, mydriasis, urinary retention and constipation. 28

Nonpharmacological Approaches to the Treatment of Parkinsonism v. Additional approaches to the treatment of Parkinson’s disease include: ØSurgical procedures ØBrain stimulation and ØTransplantation of dopaminergic cells. v. In general, surgical procedures are reserved for patients who are refractive to levodopa or who have profound dyskinesias or fluctuations in response to levodopa.

Alzheimer's Disease (AD) v. Alzheimer’s disease characterized functionally by a loss of memory and impairment of cognitive abilities that is gradual in onset but relentless in progression. v. Impairment of short-term memory usually is the first clinical feature. As the condition progresses, additional cognitive abilities are impaired, among them the ability to calculate, exercise and the use common objects and tools. v. AD is characterized by marked atrophy of the cerebral cortex and loss of cortical and subcortical neurons. v. In patients with AD there are senile plaques, which are spherical accumulations of the protein b-amyloid accompanied by degenerating neuronal processes. v. In advanced AD, senile plaques are numerous and most in the hippocampus and associative regions of the cortex 30

Alzheimer's Disease (AD) Neurochemistry v. Direct analysis of neurotransmitter content in the cerebral cortex shows a reduction of acetylcholine (due to atrophy and degeneration of subcortical cholinergic neurons ), which given rise to the "cholinergic hypothesis, ". v. Cholinergic hypothesis proposes that a deficiency of acetylcholine is critical in the genesis of the symptoms of AD 31

Alzheimer's Disease (AD) Treatment of Alzheimer's Disease v. A major approach to the treatment of AD has involved attempts to augment the cholinergic function of the brain ØPrecursors of acetylcholine synthesis: such as choline chloride and phosphatidyl choline were used for the treatment but randomized trials failed to demonstrate any clinically significant efficacy. ØFour inhibitors of acetylcholinesterase (ACh. E) currently are approved for treatment of Alzheimer's disease: v. Tacrine v. Donepezil v. Rivastigmine and v. Galantamine. 32

Treatment of Alzheimer's Disease Tacrine v. It is a potent centrally acting inhibitor of acetylcholinesterase. v. The side effects of tacrine are; abdominal cramping, anorexia, nausea, vomiting, diarrhea and hepatotoxicity (elevations of serum transaminases) are observed in 50% of those treated. v. Because of significant side effects, tacrine is not used widely clinically. Donepezil v. It is a selective inhibitor of acetylcholinesterase in the CNS with little effect on ACh. E in peripheral tissues. v. It produces modest improvements in cognitive scores in Alzheimer's disease patients and has a long half-life, allowing once-daily dosing. 33

Treatment of Alzheimer's Disease Rivastigmine and galantamine v. Rivastigmine and galantamine are dosed twice daily and produced a similar degree of cognitive improvement. v. Adverse effects associated with donepezil, rivastigmine, and galantamine are similar in character but generally less frequent and less severe than those observed with tacrine. v. They include nausea, diarrhea, vomiting, and insomnia. v. Donepezil, rivastigmine, and galantamine are not associated with the hepatotoxicity that limits the use of tacrine. 34

Treatment of Alzheimer's Disease NMDA glutamate-receptor antagonist (memantine) v. An alternative strategy for the treatment of AD is the use of the NMDA glutamate-receptor antagonist memantine v. Memantine produces a use-dependent blockade of NMDA receptors. v. In patients with moderate to severe Alzheimer’s disease, use of memantine is associated with a reduced rate of clinical deterioration. v Adverse effects of memantine usually are mild and reversible and may include headache or dizziness. 35

Huntington's Disease (HD) Clinical Features v. Huntington’s disease was first described by George Huntington, a New York physician, in 1872. v. It is an inherited disease of the CNS characterized by progressive dementia and involuntary choreic movements, resulting from degeneration of the caudate and putamen nuclei with prominent neuronal loss in the striatum of the brain v. Other areas of the brain also are affected like cerebral cortex, hypothalamus, and thalamus but less severely. v. The symptoms develop insidiously, either as a movement disorder manifest by brief jerk like movements of the extremities, trunk, face, and neck (chorea) or as personality changes or both. v. Huntington’s disease is also characterized by the gradual onset of motor incoordination and cognitive decline in midlife. 36
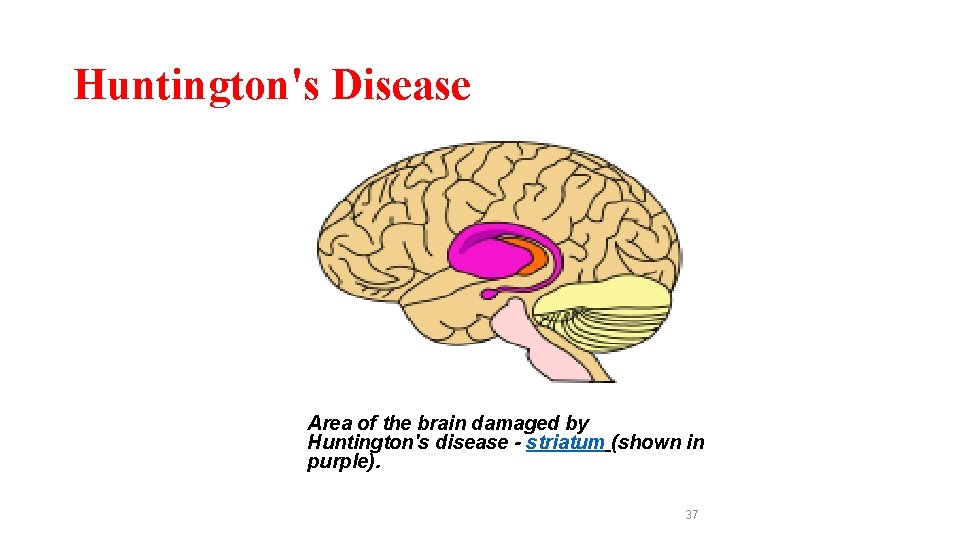
Huntington's Disease Area of the brain damaged by Huntington's disease - striatum (shown in purple). 37

Symptomatic Treatment of Huntington's Disease v. No current medication slows the progression of the disease, and many medications can impair function because of side effects. v. Treatment is needed for patients who are depressed, irritable, paranoid, excessively anxious, or psychotic. v. Depression can be treated effectively with SSRIs v. Paranoia, delusional states, and psychosis usually require treatment with antipsychotic drugs, but the doses required often are lower than those usually used in primary psychiatric disorders. v. Over a course of 15 to 30 years, the affected person becomes totally disabled and unable to communicate, requiring full-time care; death ensues from the complications of immobility. 38
- Slides: 38