Treatment of Antibody Mediated Rejection in Kidney Transplant

Treatment of Antibody Mediated Rejection in Kidney Transplant Recipients in an Urban Academic Medical Center Diana N. Hoang, Pharm. D Candidate 2021 Parichhya Luitel, Pharm. D Candidate 2021 Nicole M. Sifontis, Pharm. D Temple University School of Pharmacy, Philadelphia, PA

Learning Objectives ● Define antibody mediated rejection ● Discuss current treatment options for antibody mediated rejection ● Present data from this investigator initiated research study evaluating outcomes in kidney transplant recipients with antibody mediated rejection

What is Antibody Mediated Rejection? ● Antibody mediated rejection (AMR) in kidney transplantation is defined as histological changes caused by circulating donorspecific antibodies. ● It occurs in about 5 -7% of kidney recipients and is responsible for 20 -48% of acute rejection episodes. ● It occurs more commonly within the first 3 months post-transplant but continues to pose challenges for long-term graft survival. ● Currently, there are no standardized published guidelines for treatment of AMR in kidney transplantation.
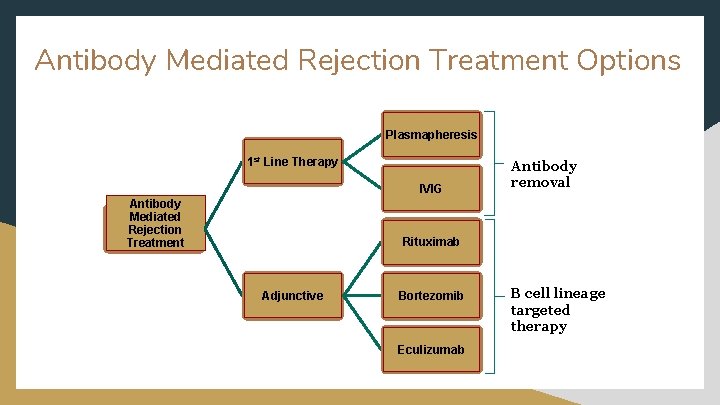
Antibody Mediated Rejection Treatment Options Plasmapheresis 1 st Line Therapy IVIG Antibody Mediated Rejection Treatment Antibody removal Rituximab Adjunctive Bortezomib Eculizumab B cell lineage targeted therapy

Study Purpose ● To describe the different regimens used to reverse antibody mediated rejection in kidney transplant recipients at our institution, and assess 1 year outcomes in these recipients.
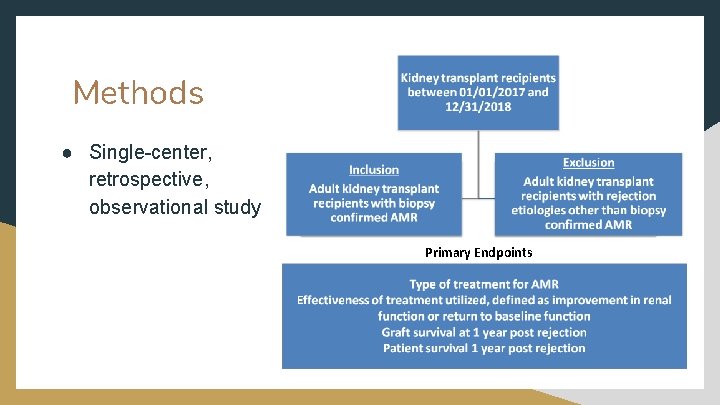
Methods ● Single-center, retrospective, observational study Primary Outcomes Primary Endpoints

Baseline Characteristics Results A total of 24 renal transplants patients with biopsy confirmed antibody mediated rejection were included and 19 patients were evaluable. Mean age at transplant, years + SD 48. 5 + 12 Gender, n (%) Male Female 9 (47) 10 (53) Ethnicity, n (%) African American Caucasian Hispanic 10 (53) 5 (26) 4 (21) Median % c. PRA 10 (IQR 0 -98) Previous Transplant, n (%) 4 (21) Median time to AMR diagnosis, days 92 (IQR 54 -350)

Results- Immunosuppression Induction agent, n (%) Thymoglobulin Basiliximab Alemtuzumab 15 (79) 2 (11) Mean tacrolimus trough level at the time of AMR diagnosis, ng/m. L + SD 10 + 3 Mean mycophenolic acid dose at time of AMR diagnosis, mg/day + SD 1501 + 497 Maintenance corticosteroid at time of diagnosis, n (%) 11 (58)
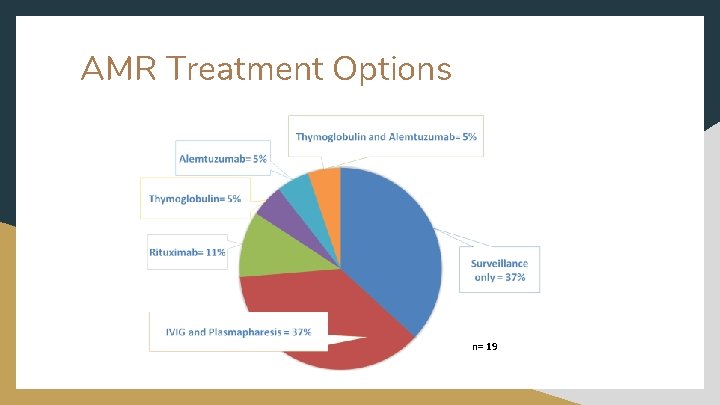
AMR Treatment Options n= 19

Primary Endpoints

Discussion ● This study is limited by its retrospective, single-center design ● The treatment regimens employed were similar to previous studies but of note about ⅓ of the cohort were managed by surveillance only, which supports theory that in some cases a pharmacologic agent may not be necessary to change outcomes. ● Analysis of adverse events associated with the use of these biologic regimen should be considered. ● Cost benefit analysis is warranted.

Conclusion ● The study findings suggest favorable outcomes in kidney transplant recipients at our institution who experienced antibody mediated rejection during the study period. ● Longer follow up is warranted to assess the long-term impact on graft and patient survival.

References 1. 2. 3. Puttarajappa C, Shapiro R, Tan HP. Antibody-mediated rejection in kidney transplantation: a review. Journal of Transplantation 2012; 193724: 1 -9 Fehr T, Gaspert A. Antibody‐mediated kidney allograft rejection: therapeutic options and their experimental rationale. Transplant International 2012; 25: 623 -632. Nickerson, PW. What have we learned about how to prevent and treat antibody ‐mediated rejection in kidney transplantation? . Am J Transplantation 2020; 20: 12 -22.

QUESTIONS? Questions?
- Slides: 14