Treatment of Ankylosing Spondylitis Enbrel RA MENA Speaker

Treatment of Ankylosing Spondylitis Enbrel RA MENA Speaker Bureau Dubai, United Arab Emirates January 2009 Prof. Joachim R. Kalden Director emeritus Medical Department III Div. for Molecular Immunology University of Erlangen-Nuremberg

Ankylosing Spondylitis Overview – A type of arthritis that causes inflammation and eventually fusion of the spine and the spinal joints. Involvement of peripheral joints and extraarticular manifestations. – AS causes pain, stiffness, disability, decreased spinal mobility, and decreased quality of life – The prevalence ranges from 0. 1 to 1 percent of the population – Men are affected three times more than women – Commonly develops between the ages of 15 and 40 – 95 percent of people with AS share the genetic marker HLA-B 27

Treatment of Ankylosing Spondylitis Overview • • Treatment goals Use of traditional therapies Clinical results with TNFalpha antagonists Guidelines for the management of TNFalpha antagonists

Treatment goals in Ankylosing Spondylitis § Reduce and or prevent deleterious effects of: • Inflammation • Ankylosis Inhibition of osteoblastogenesis • Abnormal posture Dougados et al. J. Rheumatol 2001; 28 -62: 16 -20.

Development of Ankylosis in Ankylosing Spondylitis • The earliest change seen was a sub acute osteitis in the immediately adjacent bone. • This was followed by extensive replacement of the fibrocartilage and adjacent bone by fibrous tissue in which there was often little remaining evidence of inflammation. • The late stage of the disease in this joint consisted of assification. From: Cruickshank and Path, Pathology of Ankylosing Spondylitis
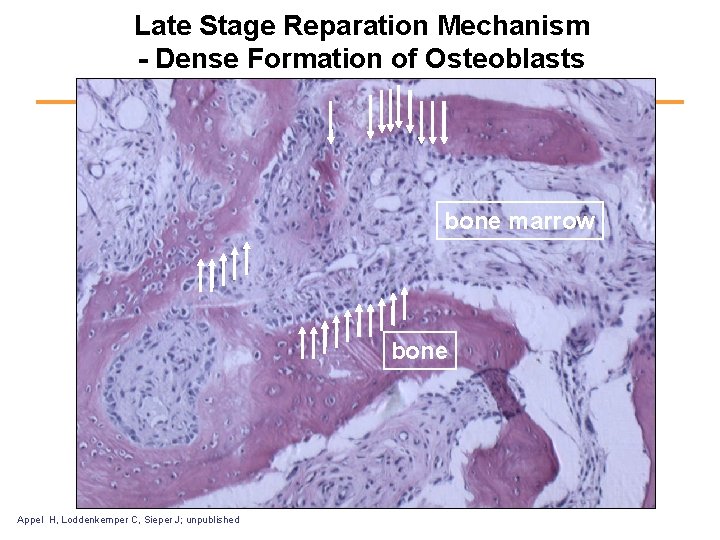
Late Stage Reparation Mechanism - Dense Formation of Osteoblasts bone marrow bone Appel H, Loddenkemper C, Sieper J; unpublished

ASAS/EULAR Recommendations for the Management of AS Education, exercise, physical therapy, rehabilitation, patient associations, self help groups NSAIDs Axial disease Zochling J, et al. Ann Rheum Dis. 2006; 65: 442 -52 (excerpt) Peripheral disease Sulfasalazine Local corticosteroids TNF blockers A n a l g e s i c s S u r ge r y
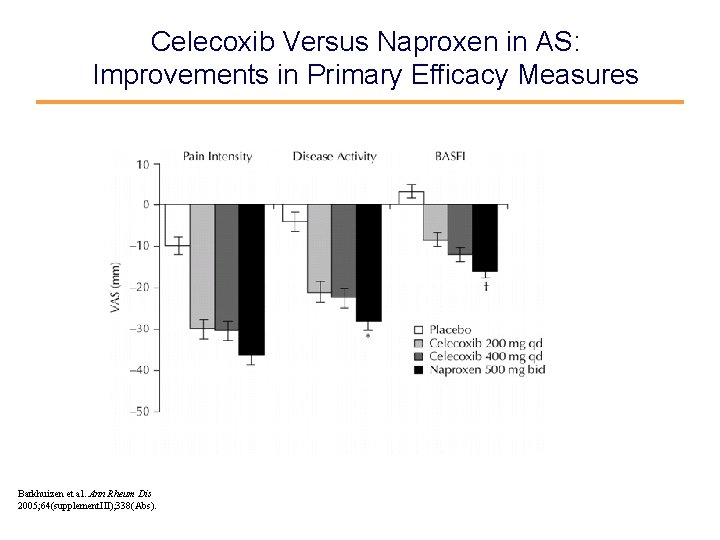
Celecoxib Versus Naproxen in AS: Improvements in Primary Efficacy Measures Barkhuizen et al. Ann Rheum Dis 2005; 64(supplement. III); 338(Abs).
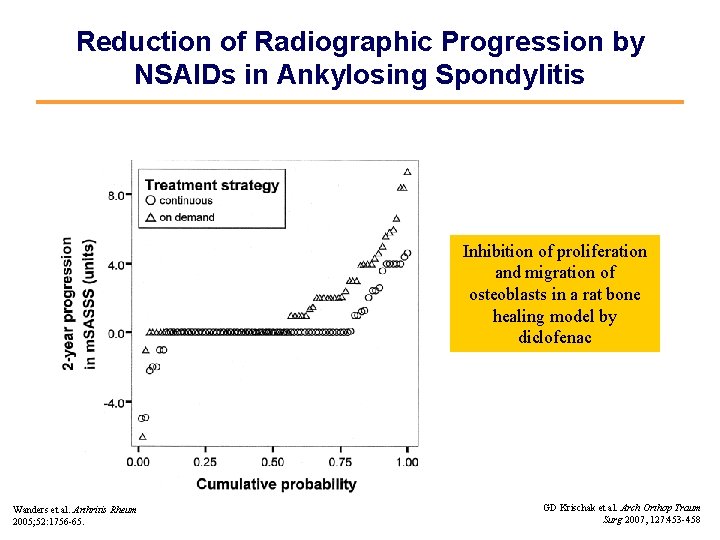
Reduction of Radiographic Progression by NSAIDs in Ankylosing Spondylitis Inhibition of proliferation and migration of osteoblasts in a rat bone healing model by diclofenac Wanders et al. Arthritis Rheum 2005; 52: 1756 -65. GD Krischak et al. Arch Orthop Traum Surg 2007, 127: 453 -458
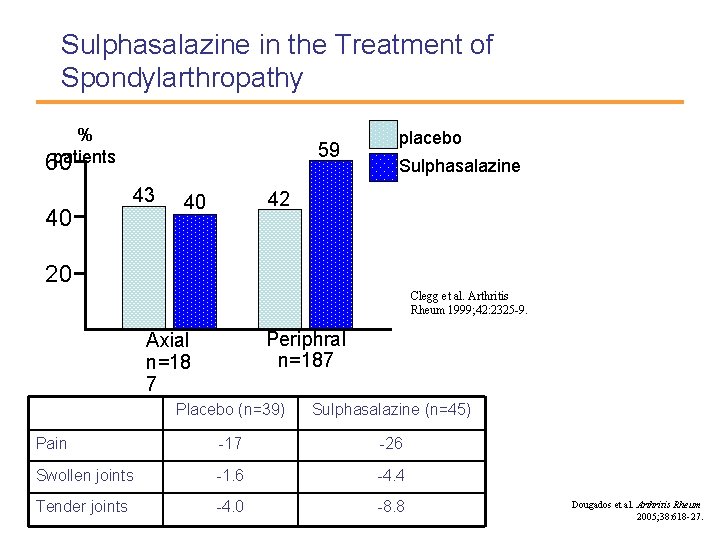
Sulphasalazine in the Treatment of Spondylarthropathy % patients 60 40 59 43 placebo Sulphasalazine 42 40 20 Clegg et al. Arthritis Rheum 1999; 42: 2325 -9. Periphral n=187 Axial n=18 7 Placebo (n=39) Sulphasalazine (n=45) Pain -17 -26 Swollen joints -1. 6 -4. 4 Tender joints -4. 0 -8. 8 Dougados et al. Arthritis Rheum 2005; 38: 618 -27.
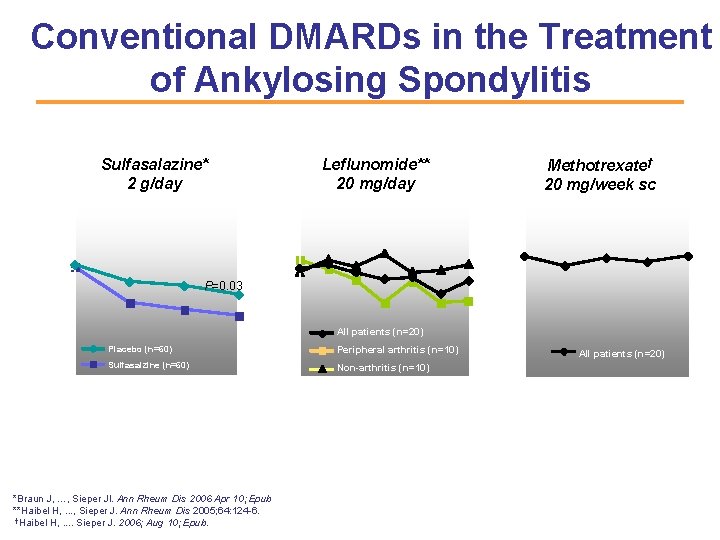
Conventional DMARDs in the Treatment of Ankylosing Spondylitis Methotrexate† 20 mg/week sc 8 8 P=0. 03 Placebo (n=60) Sulfasalzine (n=60) 0 4 8 12 16 20 24 Week *Braun J, …, Sieper Jl. Ann Rheum Dis 2006 Apr 10; Epub **Haibel H, . . . , Sieper J. Ann Rheum Dis 2005; 64: 124 -6. †Haibel H, . . Sieper J. 2006; Aug 10; Epub. 6 4 2 All patients (n=20) Peripheral arthritis (n=10) 0 Non-arthritis (n=10) 0 4 8 12 16 20 24 Week BASDAI (mean) 8 7 6 5 4 3 2 1 0 Leflunomide** 20 mg/day BASDAI (mean) Sulfasalazine* 2 g/day 6 4 2 All patients (n=20) 0 0 4 8 12 Week 16

Conclusions from treatment experiences in Ankylosing Spondylitis with traditional therapies § Traditional therapy options are not sufficient to treat active disease • Physiotherapy important to maintain function • NSAIDs alleviate pain in some but not all patients • Steroids and immunomodulators have little/no effect • No drugs which impede disease progression § Unmet medical need in the treatment of severe AS
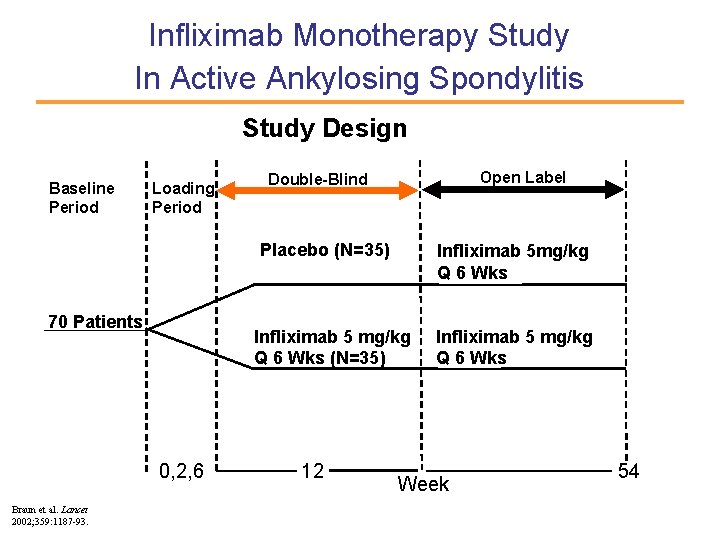
Infliximab Monotherapy Study In Active Ankylosing Spondylitis Study Design Baseline Period Loading Period Open Label Double-Blind Placebo (N=35) 70 Patients Infliximab 5 mg/kg Q 6 Wks (N=35) 0, 2, 6 Braun et al. Lancet 2002; 359: 1187 -93. Infliximab 5 mg/kg Q 6 Wks 12 Infliximab 5 mg/kg Q 6 Wks Week 54
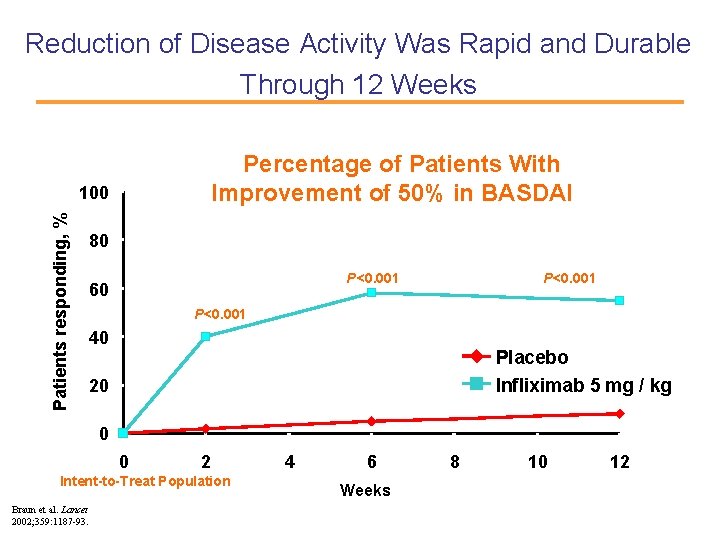
Reduction of Disease Activity Was Rapid and Durable Through 12 Weeks Percentage of Patients With Improvement of 50% in BASDAI Patients responding, % 100 80 P<0. 001 60 P<0. 001 40 Placebo Infliximab 5 mg / kg 20 0 0 2 Intent-to-Treat Population Braun et al. Lancet 2002; 359: 1187 -93. 4 6 Weeks 8 10 12
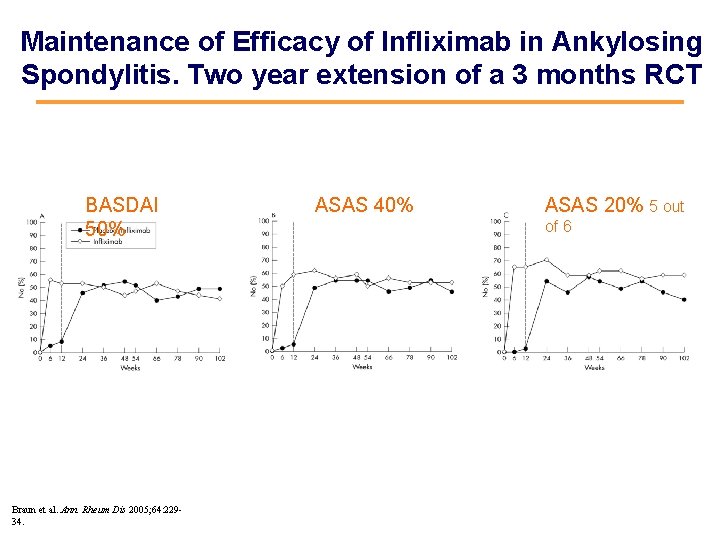
Maintenance of Efficacy of Infliximab in Ankylosing Spondylitis. Two year extension of a 3 months RCT BASDAI 50% Braun et al. Ann. Rheum Dis 2005; 64: 22934. ASAS 40% ASAS 20% 5 out of 6
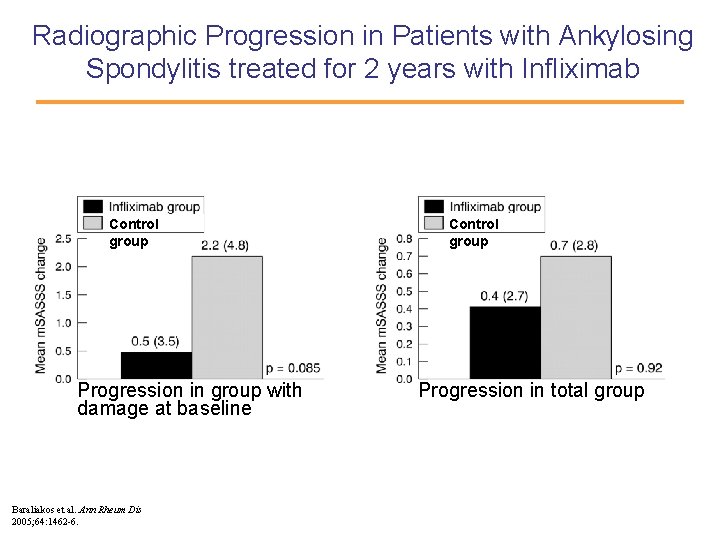
Radiographic Progression in Patients with Ankylosing Spondylitis treated for 2 years with Infliximab Control group Progression in group with damage at baseline Baraliakos et al. Ann Rheum Dis 2005; 64: 1462 -6. Control group Progression in total group
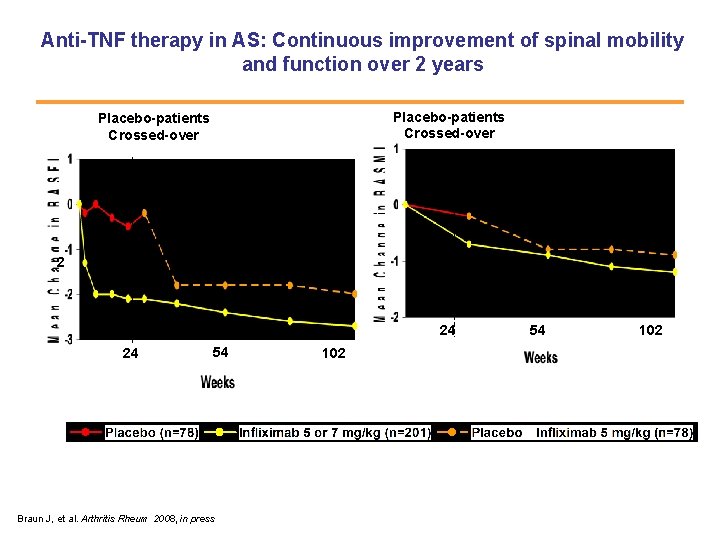
Anti-TNF therapy in AS: Continuous improvement of spinal mobility and function over 2 years Placebo-patients Crossed-over 2 24 24 54 Braun J, et al. Arthritis Rheum 2008, in press 102 54 102

A randomized controlled clinical trial of infliximab shows clinical and MRI efficacy in patients with pos. HLA B 27 and very early AS • 49 pat. with early inflammatory back pain, HLA B 27 pos. and known oedema on MRI were randomized • No demographic changes between the two groups Result • Infliximab appears to be an effective therapy in very early inflammatory back pain (significant change from baseline: MRI score of sacroiliac joints resolving of SIjoint lesions. Improvement of clinical parameters) N. Barkham et al. ACR 2007. L 11
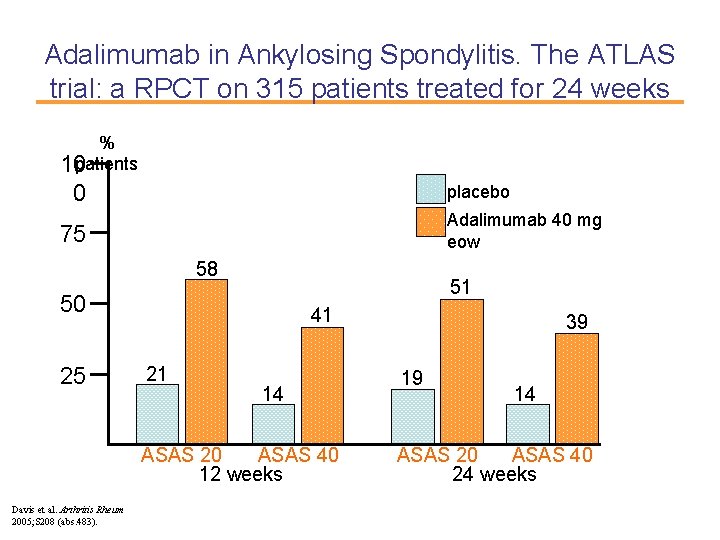
Adalimumab in Ankylosing Spondylitis. The ATLAS trial: a RPCT on 315 patients treated for 24 weeks % 10 patients 0 placebo Adalimumab 40 mg eow 75 58 51 50 25 41 21 14 ASAS 20 ASAS 40 12 weeks Davis et al. Arthritis Rheum 2005; S 208 (abs. 483). 39 19 14 ASAS 20 ASAS 40 24 weeks
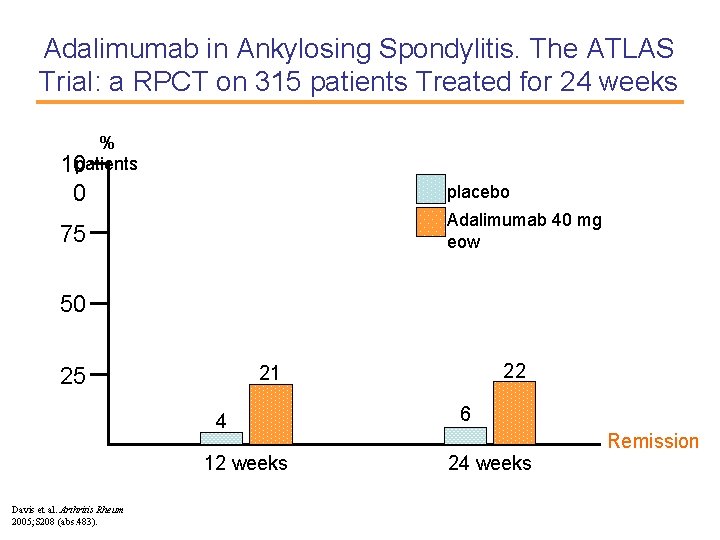
Adalimumab in Ankylosing Spondylitis. The ATLAS Trial: a RPCT on 315 patients Treated for 24 weeks % 10 patients 0 placebo Adalimumab 40 mg eow 75 50 4 12 weeks Davis et al. Arthritis Rheum 2005; S 208 (abs. 483). 22 21 25 6 Remission 24 weeks

Adalimumab in patients with total spine ankylosis • Randomized, placebo-controlled trial (ratio 2: 1) • In patients with TSA adalimumab treatment resulted in rapid and clinically significant improvement in signs and symptoms of active disease – At week 12: 50% of adalimumab treated patients achieved ASAS 20, 33% ASAS 40, ASAS 5/6 and BASDAI 50 as compared to non of placebo treated patients – After 1 year: 8/11 adalimumab treated patients achieved ASAS 20 – After 2 years: 6/11 adalimumab treated patients achieved ASAS 10 van der Heijde D et al, Ann Rheum Dis, Dec 2007
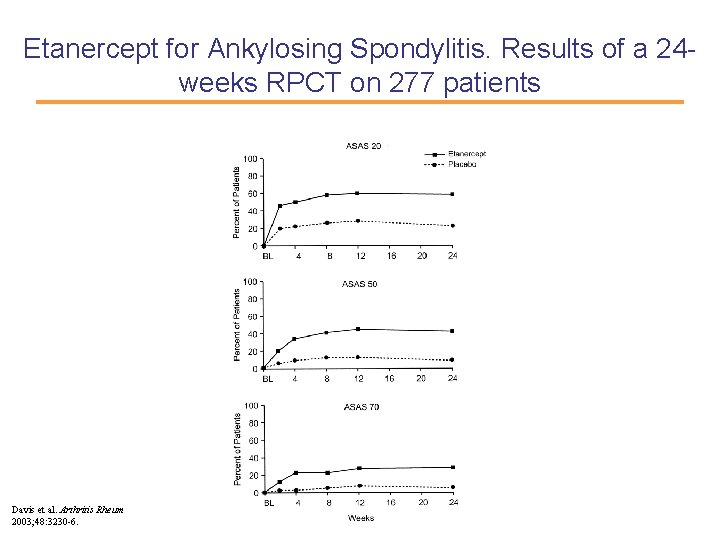
Etanercept for Ankylosing Spondylitis. Results of a 24 weeks RPCT on 277 patients Davis et al. Arthritis Rheum 2003; 48: 3230 -6.
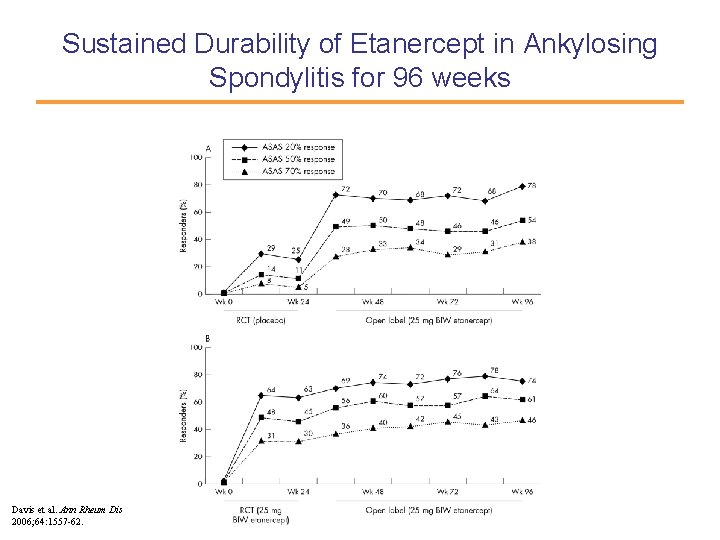
Sustained Durability of Etanercept in Ankylosing Spondylitis for 96 weeks Davis et al. Ann Rheum Dis 2006; 64: 1557 -62.
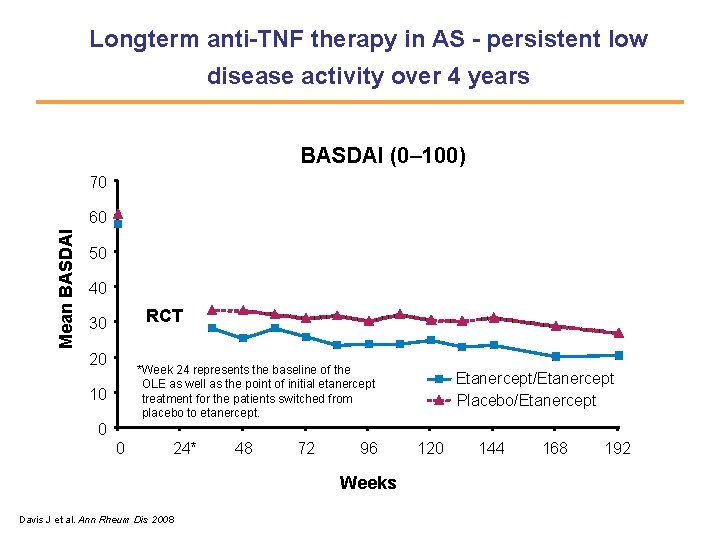
Longterm anti-TNF therapy in AS - persistent low disease activity over 4 years BASDAI (0– 100) 70 Mean BASDAI 60 50 40 RCT 30 20 *Week 24 represents the baseline of the OLE as well as the point of initial etanercept treatment for the patients switched from placebo to etanercept. 10 Etanercept/Etanercept Placebo/Etanercept 0 0 24* 48 72 96 Weeks Davis J et al. Ann Rheum Dis 2008 120 144 168 192
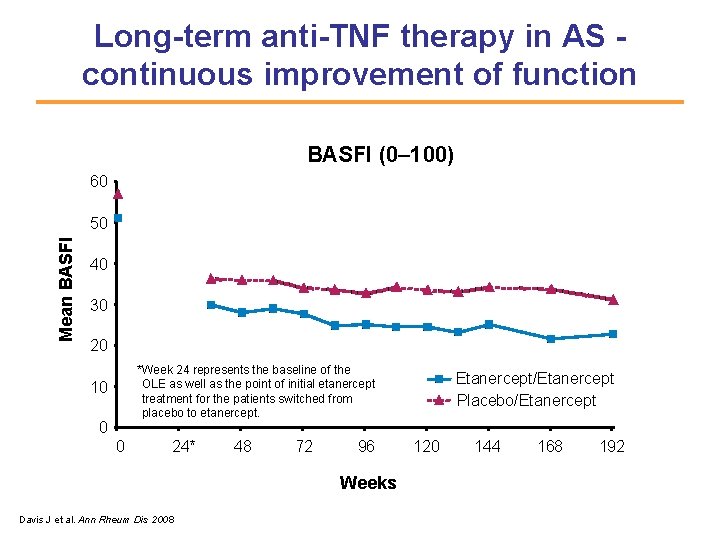
Long-term anti-TNF therapy in AS continuous improvement of function BASFI (0– 100) 60 Mean BASFI 50 40 30 20 *Week 24 represents the baseline of the OLE as well as the point of initial etanercept treatment for the patients switched from placebo to etanercept. 10 0 0 24* 48 72 96 Weeks Davis J et al. Ann Rheum Dis 2008 Etanercept/Etanercept Placebo/Etanercept 120 144 168 192

Assessment of Clinical Efficacy in a Randomized Double-Blind Study of Etanercept and Sulfasalazine in Patients With Ankylosing Spondylitis October 27, 2008 American College of Rheumatology Annual Scientific Meeting ACR 2008 J. Braun, 1 F. Huang, 2 R. Burgos-Vargas, 3 I. E. van der Horst-Bruinsma, 4 B. Freundlich, 5 B. Vlahos, 5 A. S. Koenig 5 1 Ruhr University, Bochum, Germany; 2 Chinese PLA General Hospital, Beijing China; 3 Hospital General de México and Universidad Nacional Autónoma de México, Mexico City, Mexico; 4 VU University Medical Center, Department of Rheumatology, Amsterdam, Netherlands; 5 Wyeth Research, Collegeville, PA, USA

Objective To compare the efficacy and safety of etanercept 50 mg once weekly with sulfasalazine 1. 5 to 3 g daily over 16 weeks in patients with active ankylosing spondylitis (AS)1 1 van der Linden S. et al. Arthritis Rheum 1984; 27: 361– 8.

Key Endpoints • Primary – Proportion of patients achieving ASAS 20 (20% improvement by Assessment of AS criteria) at 16 weeks • Select Secondary – – – – 1 Anderson ASAS 20, ASAS 40, ASAS 5/6, partial remission 1 BASMI (Bath Ankylosing Spondylitis Metrology Index) BASFI (Bath Ankylosing Spondylitis Functional Index) BASDAI (Bath Ankylosing Spondylitis Disease Activity Index) C-Reactive Protein through 16 weeks Back pain Modified Schober’s test et al. Arthritis Rheum 2001; 441876 -86
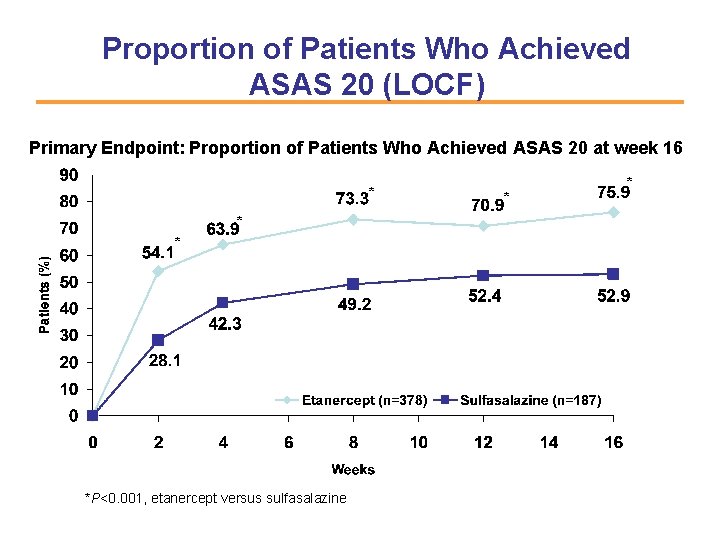
Proportion of Patients Who Achieved ASAS 20 (LOCF) Primary Endpoint: Proportion of Patients Who Achieved ASAS 20 at week 16 * *P<0. 001, etanercept versus sulfasalazine * *
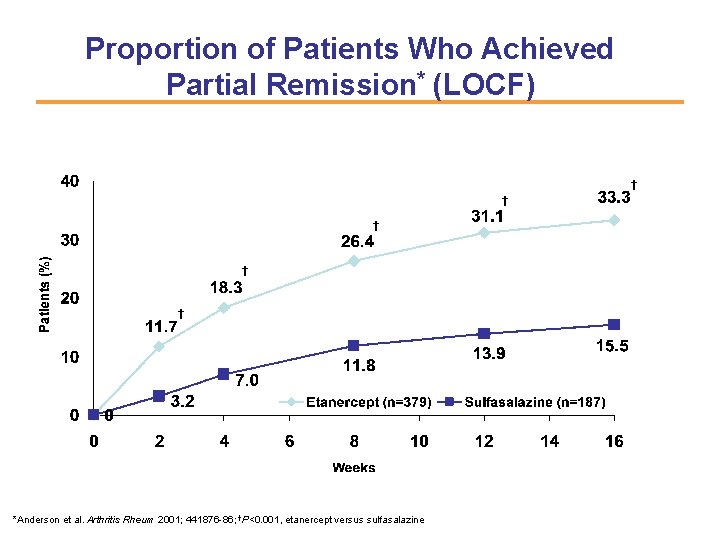
Proportion of Patients Who Achieved Partial Remission* (LOCF) † † † *Anderson et al. Arthritis Rheum 2001; 441876 -86; †P<0. 001, etanercept versus sulfasalazine
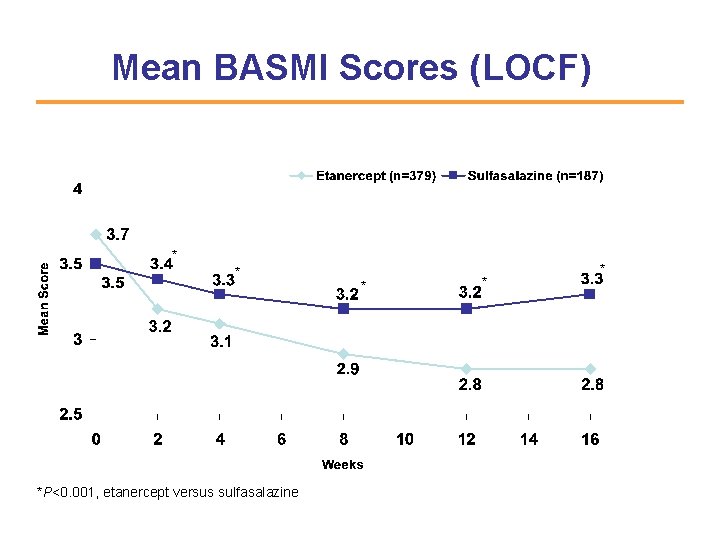
Mean BASMI Scores (LOCF) * * *P<0. 001, etanercept versus sulfasalazine * * *

Conclusions • In this 16 -week study – Etanercept therapy was superior to sulfasalazine therapy in the treatment of subjects with ankylosing spondylitis. – There were no unexpected safety findings

Etanercept Shows Persistent clinical Efficacy over 5 Years Clinical Remission BASDAI < 3 BASFI < 4 BASDAI + global physician‘s < 4 No. Achieving at 12 weeks No. Achieving at < 90% of all visits (%) 6 5 (83) 11 9 11 12 8 (73) 8 (89) 11 (100) 10 (83) According to: Baraliakos X et al. , FRI 0291, EULAR 2008 At 5 Years (n = 18): § 33% were in partial remission according to ASAS criteria § 38% achieved a BASDAI 50% response § 62% achieved a ASAS 40% response § 65% achieved a ‘ 5 out of 6’ response

Effectiveness of Adalimumab after failure of infliximab or etanercept in patients with Ps. A and AS • Open-label phase IIb studies • Ps. A „STEREO“ 66 of 442 patients discontinued and were switched to adalimumab • AS „RAPSODY“ 309 of 1186 patients discontinued and were switched to adalimumab • Ps. A: Significant improvement in ACR 20, 50, 70 and HAQ • AS: Significant improvement in ASAS 20, 50, 70 and BASDAI Burmester et al. Arthritis Rheum 2007; 56, 393
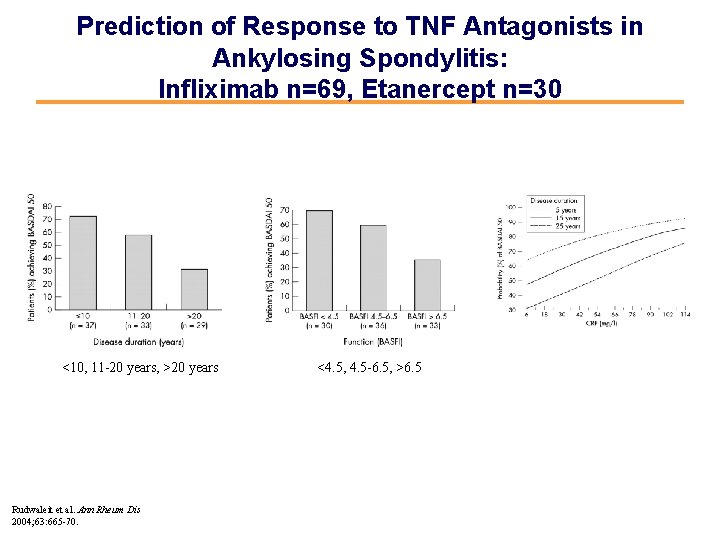
Prediction of Response to TNF Antagonists in Ankylosing Spondylitis: Infliximab n=69, Etanercept n=30 <10, 11 -20 years, >20 years Rudwaleit et al. Ann Rheum Dis 2004; 63: 665 -70. <4. 5, 4. 5 -6. 5, >6. 5
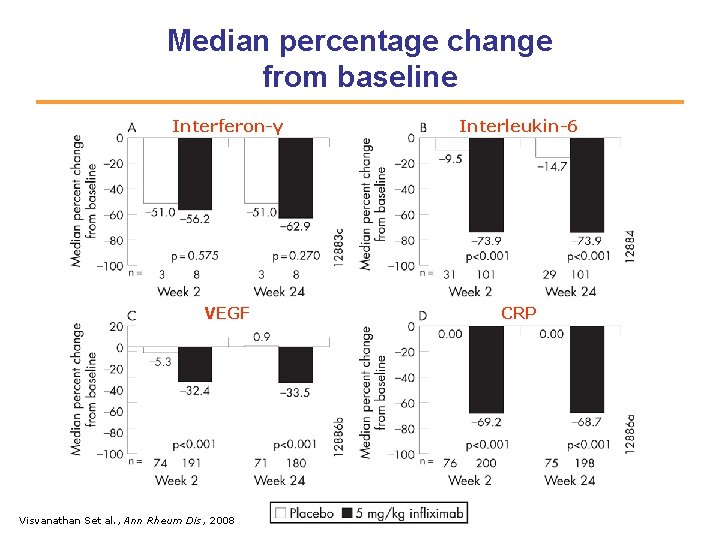
Median percentage change from baseline Interferon-γ Interleukin-6 VEGF CRP Visvanathan Set al. , Ann Rheum Dis, 2008
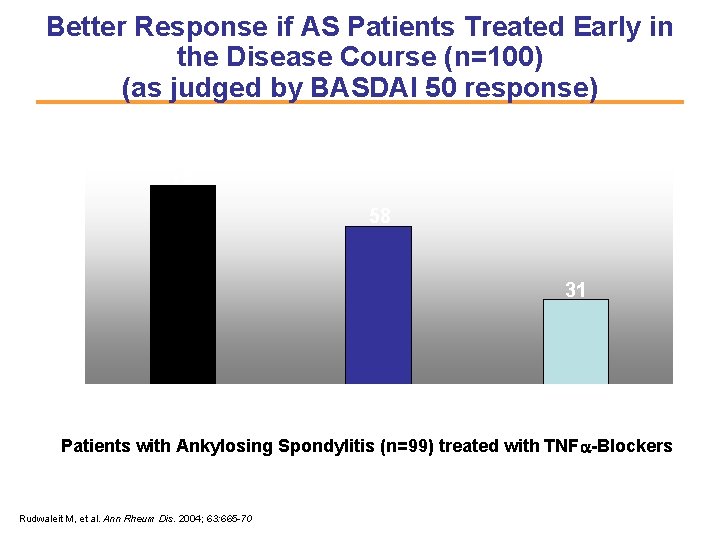
Better Response if AS Patients Treated Early in the Disease Course (n=100) (as judged by BASDAI 50 response) 80 73 58 % 60 40 31 20 0 <10 years, N=37 11 -20 years, N=33 >20 years, N=29 Patients with Ankylosing Spondylitis (n=99) treated with TNFa-Blockers Rudwaleit M, et al. Ann Rheum Dis. 2004; 63: 665 -70
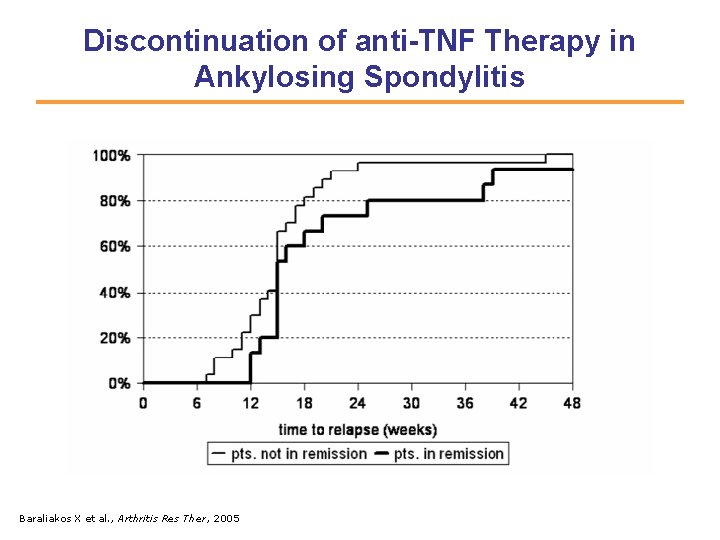
Discontinuation of anti-TNF Therapy in Ankylosing Spondylitis Baraliakos X et al. , Arthritis Res Ther, 2005

Effects of TNFalpha antagonists on extra-articular manifestations Reduction in the number of enthesetic regions - Infliximab Braun et al. , Lancet 2002; 359: 1187 -93 - Etanercept Marzo-Ortego et al. , Arthritis Rheum 2001; 44: 2112 -17 - Adalimumab Decreased incidence of anterior uveiitis - Infliximab Braun et al. , Arthritis Rheum 2005; 52: 2447 -51 - Adalimumab Lyndell LL et al, Arthritis & Rheumatism (2007)56: 3248 - Etanercept less effective -3252
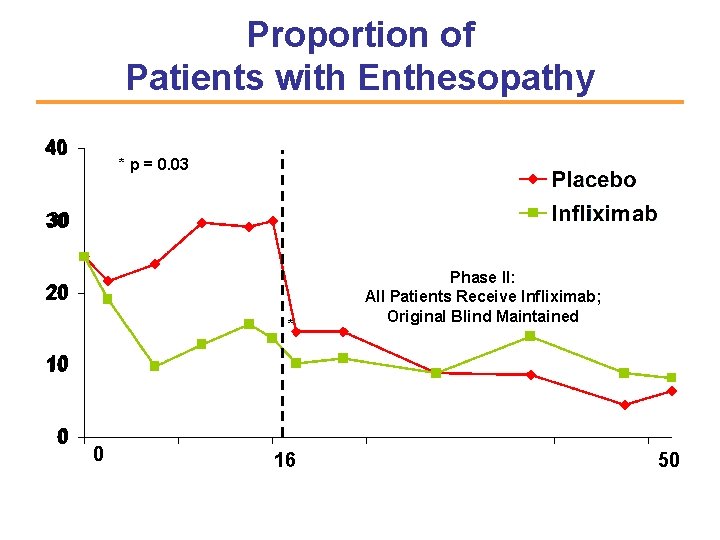
Proportion of Patients with Enthesopathy The Impact Study Placebo cross-over to Infliximab Percent of Patients 40 00 * p = 0. 03 30 10 20 * Phase II: All Patients Receive Infliximab; Original Blind Maintained 10 0 16 16 5050 Weeks C. Antoni et al. Arth Rheum 2003

International ASAS Consensus Statement for the Use of anti-TNF in Patients with Ankylosing Spondylitis • Indication AS Active disease - BASDAI ≥ 4 - expert opinion - acute phase response - imaging modalities Failure of ≥ 2 NSAIDs In case of peripheral arthritis failure of intraarticular corticosteroids/sulphasalazine Absence of contraindication • Monitoring BASDAI ASAS core set Responder: improvement of ≥ 2 units on BASDAI Baeten et al. Ann Rheum Dis 2003; 62: 829 -34. . Definitive

News from the ACR Meeting 2008 • Long-term efficacy for up to 7 years for all 3 TNF antagonists being licenzed for the treatment of AS. No new safety signals • Re-Treatment of AS patients who flaired after ADA treatment was stopped is possible • Early effective treatment of AS (as shown for ADA) might significantly improve the work productivity

Summary • TNF antagonists proven in Ps. A, AS to have a long-term clinical efficacy and acceptable safety profile • No new side effects reported in long-term clinical trials as compared to previous studies and registry data • Identical recommendation for screening patients before TNFalpha antagonist should be started • Psoriasis induced by TNFalpha antagonists – (Sfakis PP et al. Arthritis Rheum 2005) – (Kary S. Ann Rheum Dis 2006; 65: 405 -407) – (Massara A et al. Rheumatology 2006; 45: 730 -733)
- Slides: 43