TRATAMIENTO DEL CNCER DE PULMN AVANZADO Dr Claudio








- Slides: 38

TRATAMIENTO DEL CÁNCER DE PULMÓN AVANZADO Dr Claudio Martin Jefe del Servicio de Oncología Torácica Instituto Alexander Fleming Hospital de Rehabilitación Respiratoria Maria Ferrer Buenos Aires. Argentina

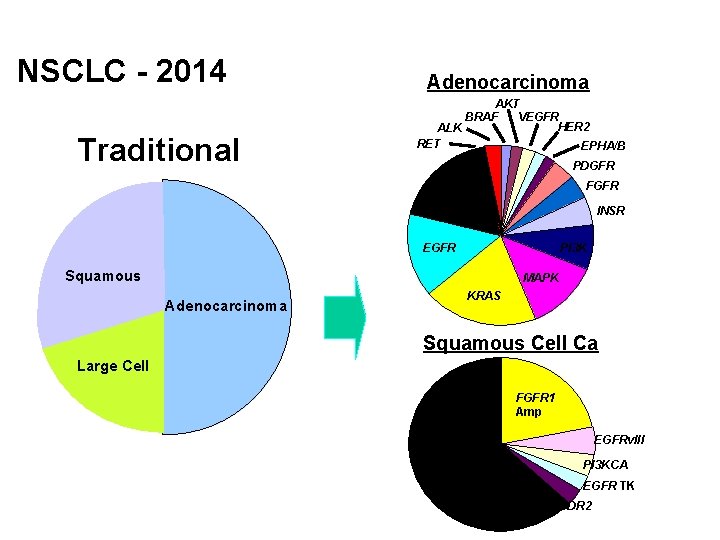
NSCLC - 2014 Traditional Adenocarcinoma AKT BRAF VEGFR HER 2 ALK RET EPHA/B PDGFR FGFR Unknown INSR EGFR PI 3 K Squamous MAPK Adenocarcinoma KRAS Squamous Cell Ca Large Cell FGFR 1 Amp EGFRv. III Unknown PI 3 KCA EGFR TK DDR 2 Adapted from W. Pao and N Girard, Lancet Oncol, 2011

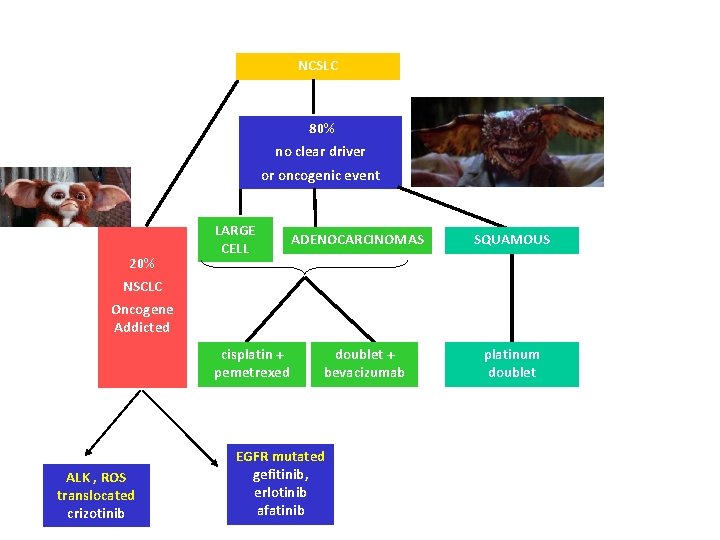
NCSLC 80% no clear driver or oncogenic event 20% LARGE CELL ADENOCARCINOMAS SQUAMOUS NSCLC Oncogene Addicted cisplatin + pemetrexed ALK , ROS translocated crizotinib doublet + bevacizumab EGFR mutated gefitinib, erlotinib afatinib platinum doublet

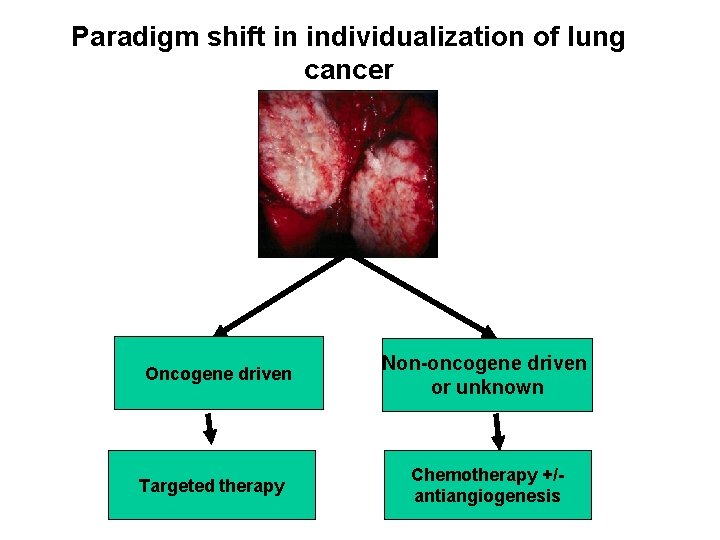
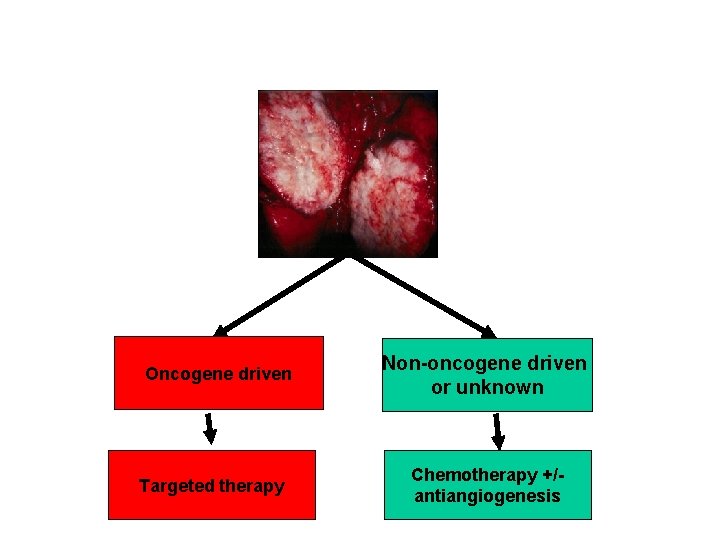
Paradigm shift in individualization of lung cancer Oncogene driven Targeted therapy Non-oncogene driven or unknown Chemotherapy +/antiangiogenesis

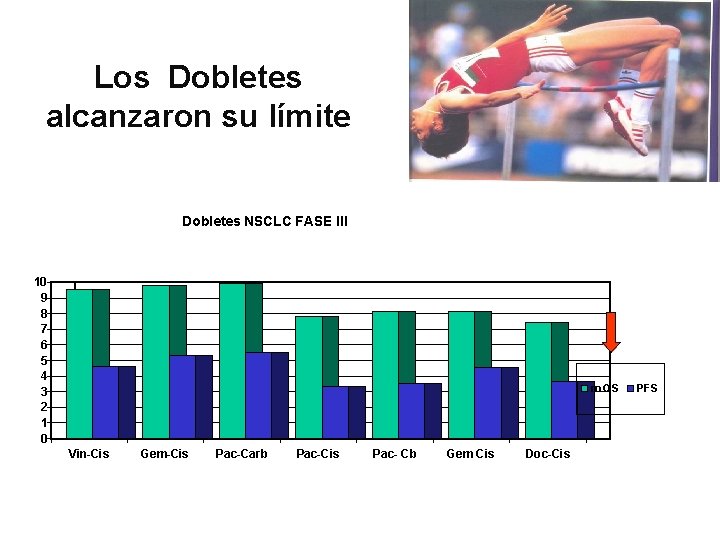
Los Dobletes alcanzaron su límite Dobletes NSCLC FASE III 10 9 8 7 6 5 4 3 2 1 0 m OS Vin-Cis Gem-Cis Pac-Carb Pac-Cis Pac- Cb Gem Cis Doc-Cis PFS

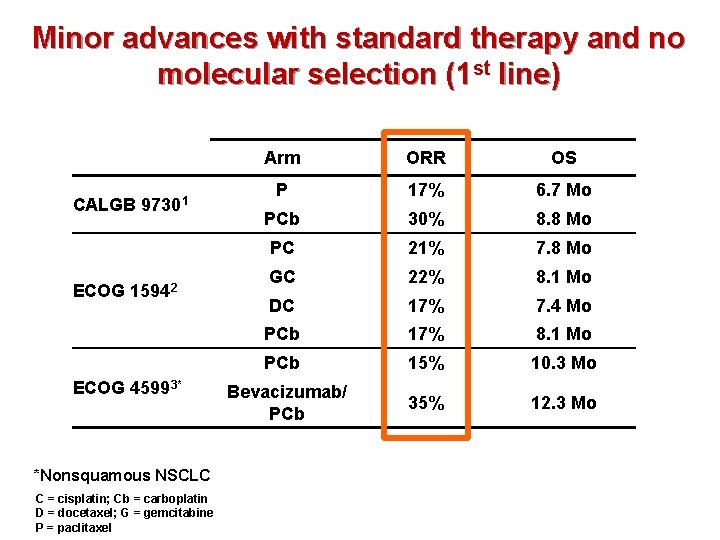
Minor advances with standard therapy and no molecular selection (1 st line) CALGB 97301 ECOG 15942 ECOG 45993* Arm ORR OS P 17% 6. 7 Mo PCb 30% 8. 8 Mo PC 21% 7. 8 Mo GC 22% 8. 1 Mo DC 17% 7. 4 Mo PCb 17% 8. 1 Mo PCb 15% 10. 3 Mo Bevacizumab/ PCb 35% 12. 3 Mo *Nonsquamous NSCLC C = cisplatin; Cb = carboplatin D = docetaxel; G = gemcitabine P = paclitaxel 1. Lilenbaum et al. , J Clin Oncol 2005; 23: 190 -196 2. Schiller et al. , New Engl J Med 2002; 346: 92 -98 3. Sandler et al. , New Engl J Med 2006; 355: 2542 -2550

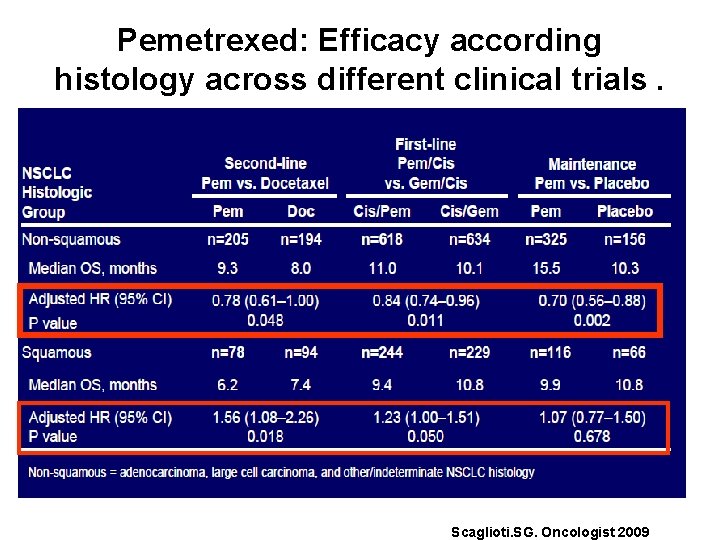
HISTOLOGY INTERACTION l Efficacy ( Pemetrexed) l Safety ( Bevacizumab )

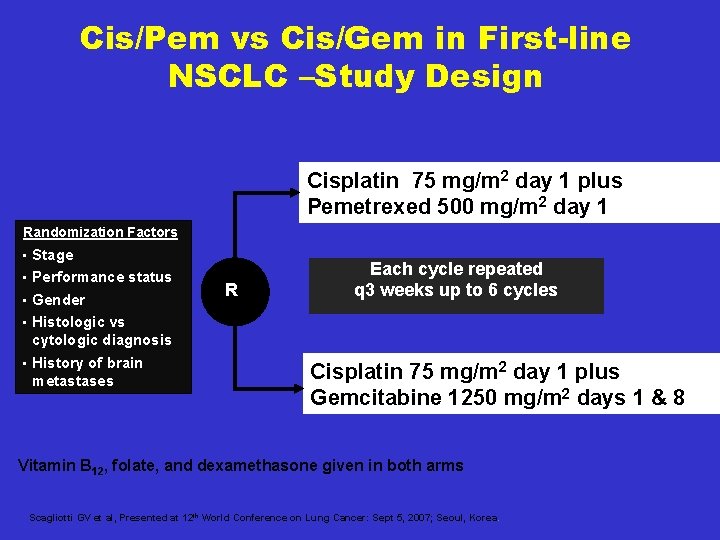
Cis/Pem vs Cis/Gem in First-line NSCLC –Study Design Cisplatin 75 mg/m 2 day 1 plus Pemetrexed 500 mg/m 2 day 1 Randomization Factors • • • Stage Performance status Gender Histologic vs cytologic diagnosis History of brain metastases R Each cycle repeated q 3 weeks up to 6 cycles Cisplatin 75 mg/m 2 day 1 plus Gemcitabine 1250 mg/m 2 days 1 & 8 Vitamin B 12, folate, and dexamethasone given in both arms Scagliotti GV et al, Presented at 12 th World Conference on Lung Cancer: Sept 5, 2007; Seoul, Korea.

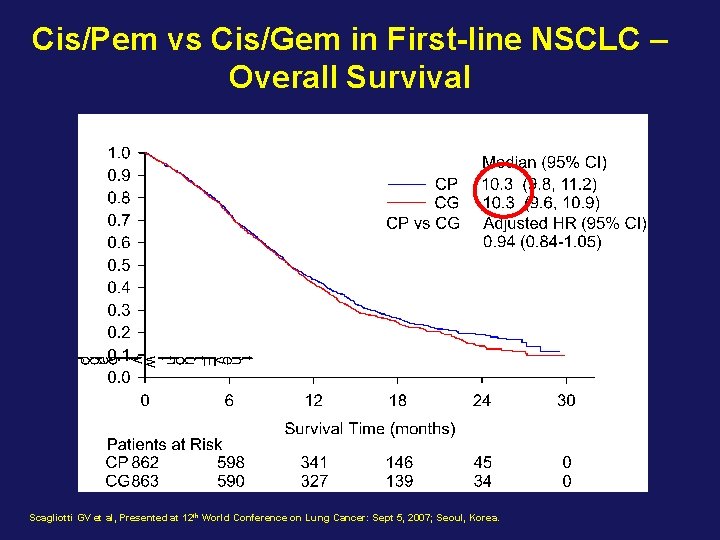
Cis/Pem vs Cis/Gem in First-line NSCLC – Overall Survival Scagliotti GV et al, Presented at 12 th World Conference on Lung Cancer: Sept 5, 2007; Seoul, Korea.

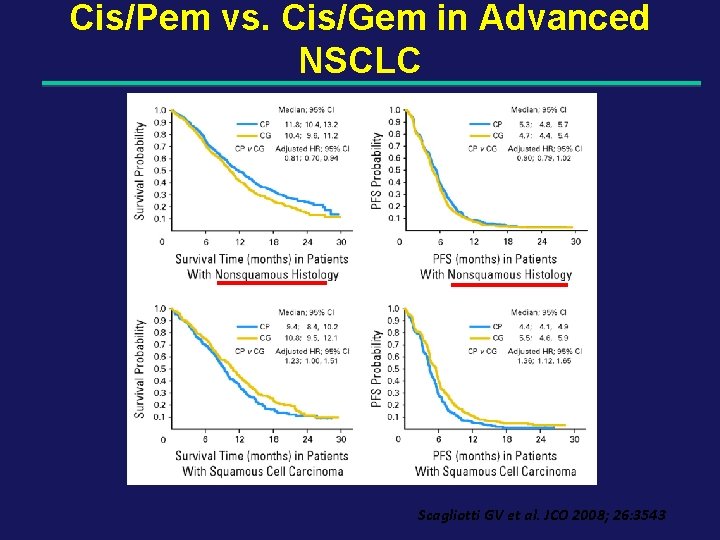
Cis/Pem vs. Cis/Gem in Advanced NSCLC Scagliotti GV et al. JCO 2008; 26: 3543

Pemetrexed: Efficacy according histology across different clinical trials. Scaglioti. SG. Oncologist 2009

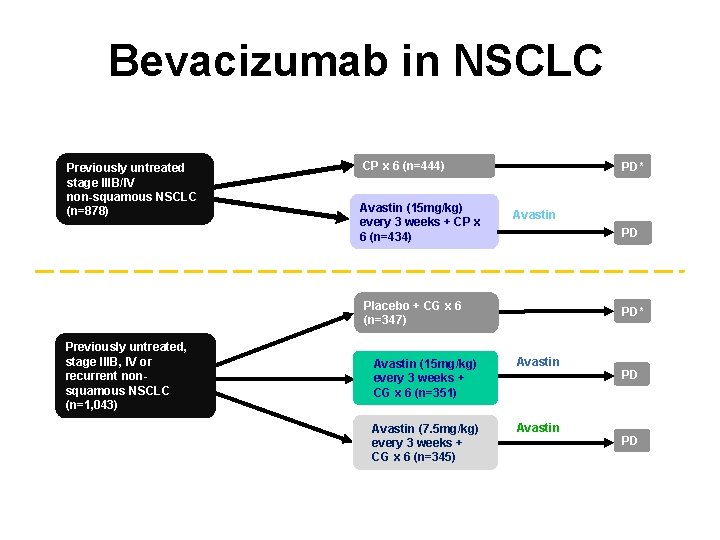
Histology and safety concern Bevacizumab

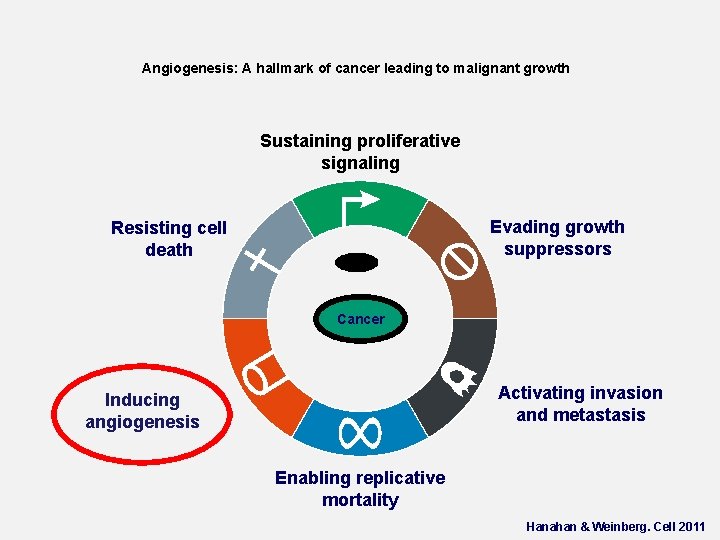
Angiogenesis: A hallmark of cancer leading to malignant growth Sustaining proliferative signaling Evading growth suppressors Resisting cell death Cancer Activating invasion and metastasis Inducing angiogenesis Enabling replicative mortality Hanahan & Weinberg. Cell 2011

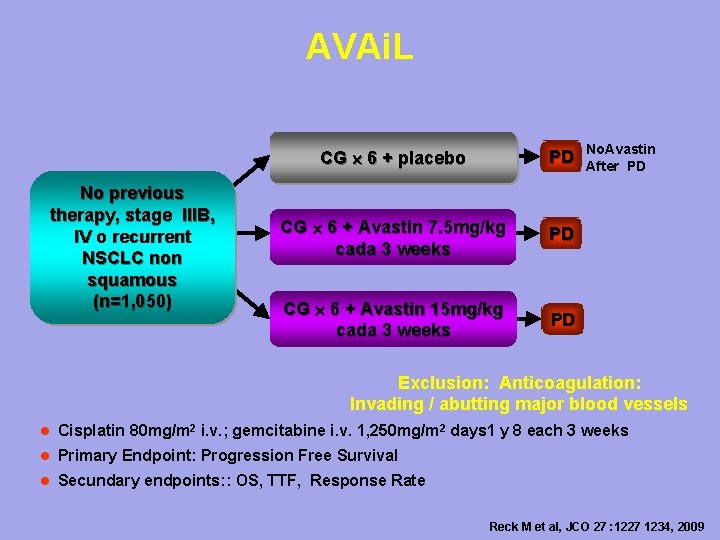
Bevacizumab in NSCLC E 4599 trial design 1 Previously untreated stage IIIB/IV non-squamous NSCLC (n=878) AVAi. L trial design 2 Previously untreated, stage IIIB, IV or recurrent nonsquamous NSCLC (n=1, 043) *No cross over permitted CP=carboplatin + paclitaxel CG=cisplatin + gemcitabine PD=progression of disease CP x 6 (n=444) Avastin (15 mg/kg) every 3 weeks + CP x 6 (n=434) PD* Avastin PD Placebo + CG x 6 (n=347) PD* Avastin (15 mg/kg) every 3 weeks + CG x 6 (n=351) Avastin (7. 5 mg/kg) every 3 weeks + CG x 6 (n=345) Avastin PD PD 1. Sandler, et al. N Engl J Med 2006; 355: 2542 -2550. 2. Reck, et al. J Clin Oncol 2009; 27: 1227 -1234.

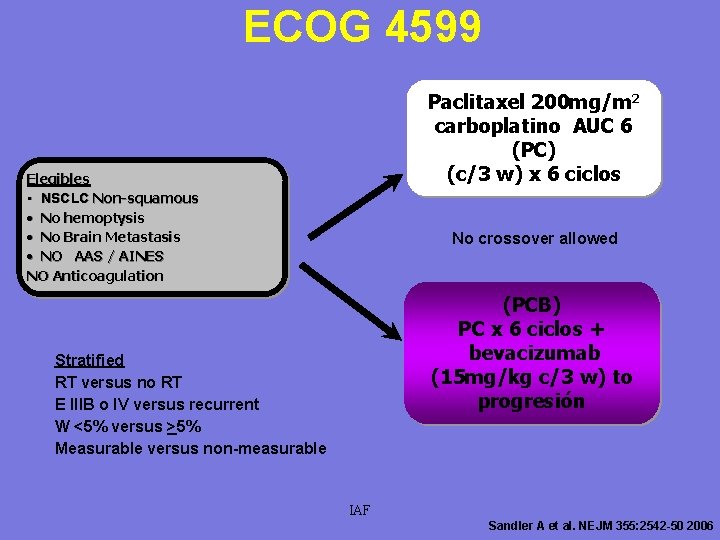
ECOG 4599 Paclitaxel 200 mg/m 2 carboplatino AUC 6 (PC) (c/3 w) x 6 ciclos Elegibles • NSCLC Non-squamous • No hemoptysis • No Brain Metastasis • NO AAS / AINES NO Anticoagulation No crossover allowed (PCB) PC x 6 ciclos + bevacizumab (15 mg/kg c/3 w) to progresión Stratified RT versus no RT E IIIB o IV versus recurrent W <5% versus >5% Measurable versus non-measurable IAF Sandler A et al. NEJM 355: 2542 -50 2006

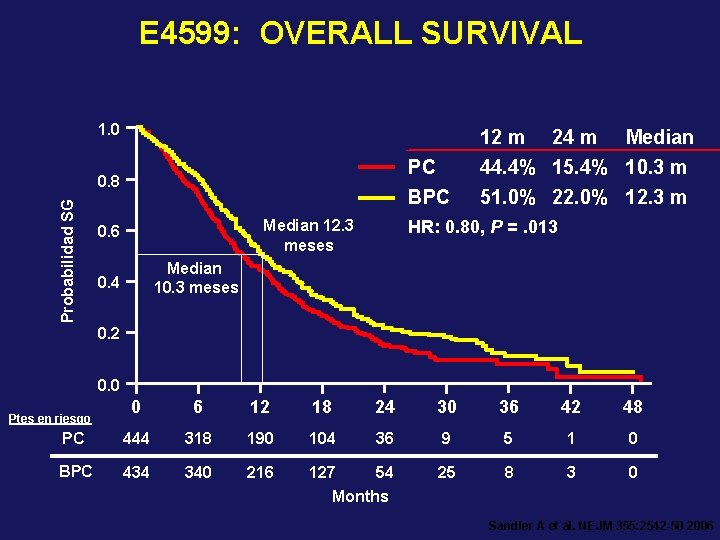
E 4599: OVERALL SURVIVAL 1. 0 12 m Probabilidad SG 0. 8 Median 12. 3 meses 0. 6 24 m Median PC 44. 4% 15. 4% 10. 3 m BPC 51. 0% 22. 0% 12. 3 m HR: 0. 80, P =. 013 Median 10. 3 meses 0. 4 0. 2 0. 0 0 6 12 18 24 30 36 42 48 PC 444 318 190 104 36 9 5 1 0 BPC 434 340 216 54 127 Months 25 8 3 0 Ptes en riesgo Sandler A et al. NEJM 355: 2542 -50 2006

AVAi. L No previous therapy, stage IIIB, IV o recurrent NSCLC non squamous (n=1, 050) CG 6 + placebo PD CG 6 + Avastin 7. 5 mg/kg cada 3 weeks PD CG 6 + Avastin 15 mg/kg cada 3 weeks PD No. Avastin After PD Exclusion: Anticoagulation: Invading / abutting major blood vessels l Cisplatin 80 mg/m 2 i. v. ; gemcitabine i. v. 1, 250 mg/m 2 days 1 y 8 each 3 weeks l Primary Endpoint: Progression Free Survival l Secundary endpoints: : OS, TTF, Response Rate Reck M et al, JCO 27 : 1227 1234, 2009

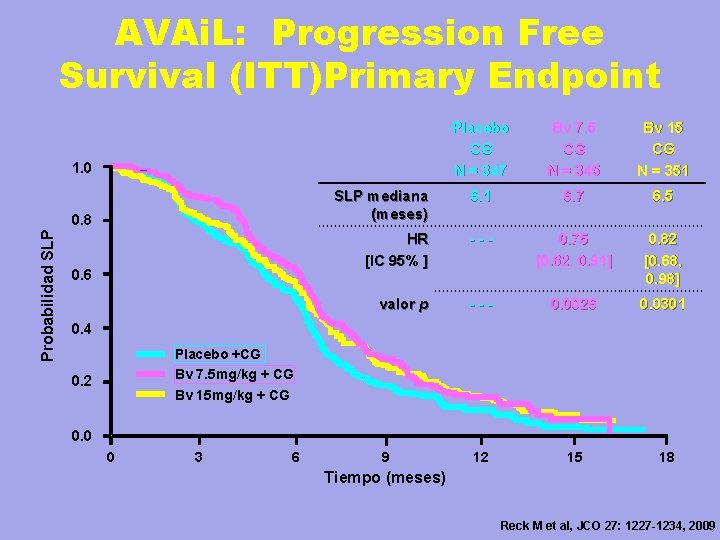
AVAi. L: Progression Free Survival (ITT)Primary Endpoint Placebo CG N = 347 Bv 7. 5 CG N = 345 Bv 15 CG N = 351 SLP mediana (meses) 6. 1 6. 7 6. 5 HR [IC 95% ] --- 0. 75 [0. 62, 0. 91] 0. 82 [0. 68, 0. 98] valor p --- 0. 0026 0. 0301 9 12 15 18 1. 0 Probabilidad SLP 0. 8 0. 6 0. 4 Placebo +CG Bv 7. 5 mg/kg + CG Bv 15 mg/kg + CG 0. 2 0. 0 0 3 6 Tiempo (meses) Reck M et al, JCO 27: 1227 -1234, 2009

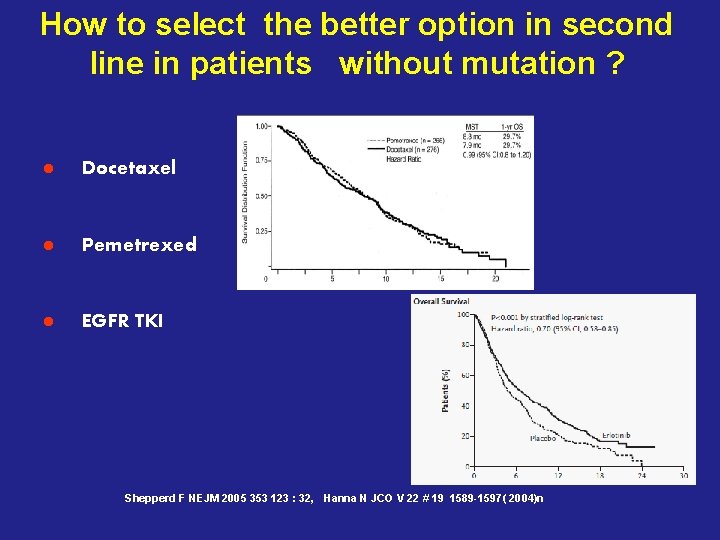
How to select the better option in second line in patients without mutation ? l Docetaxel l Pemetrexed l EGFR TKI Shepperd F NEJM 2005 353 123 : 32, Hanna N JCO V 22 # 19 1589 -1597( 2004)n

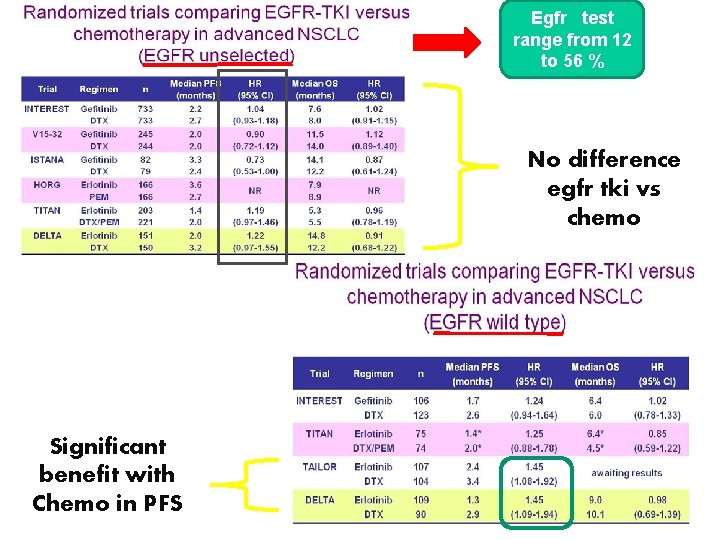
Egfr test range from 12 to 56 % No difference egfr tki vs chemo Significant benefit with Chemo in PFS

Oncogene driven Targeted therapy Non-oncogene driven or unknown Chemotherapy +/antiangiogenesis

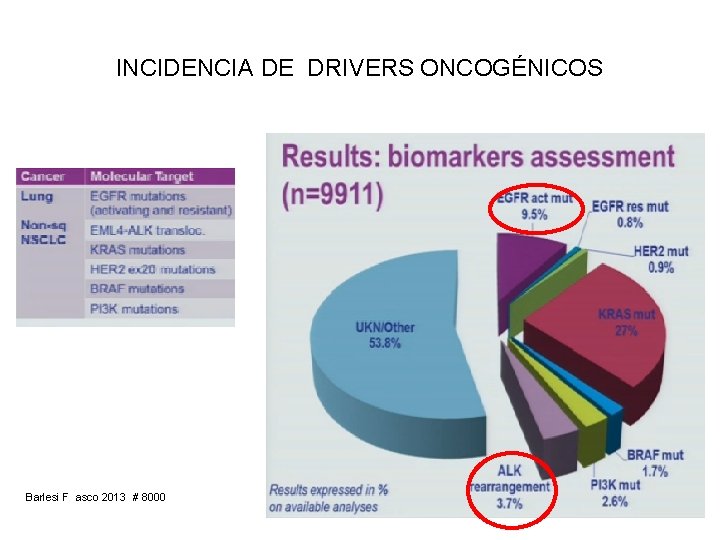
INCIDENCIA DE DRIVERS ONCOGÉNICOS Barlesi F asco 2013 # 8000

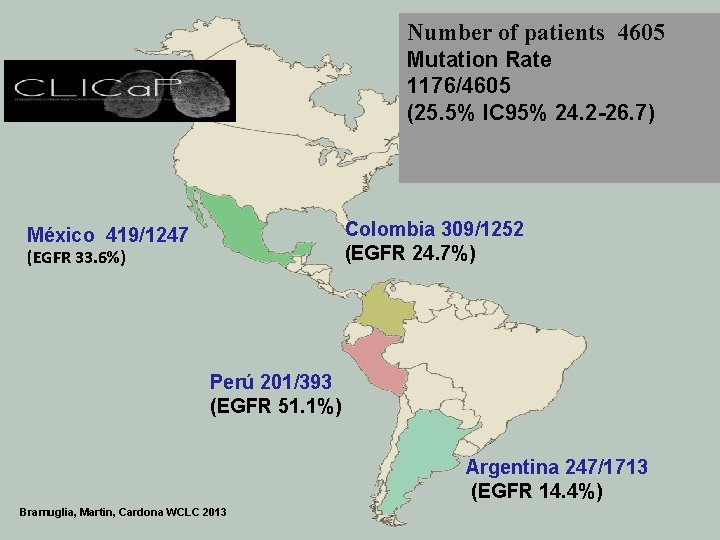
Number of patients 4605 Mutation Rate 1176/4605 (25. 5% IC 95% 24. 2 -26. 7) Colombia 309/1252 (EGFR 24. 7%) México 419/1247 (EGFR 33. 6%) Perú 201/393 (EGFR 51. 1%) Argentina 247/1713 (EGFR 14. 4%) Bramuglia, Martin, Cardona WCLC 2013

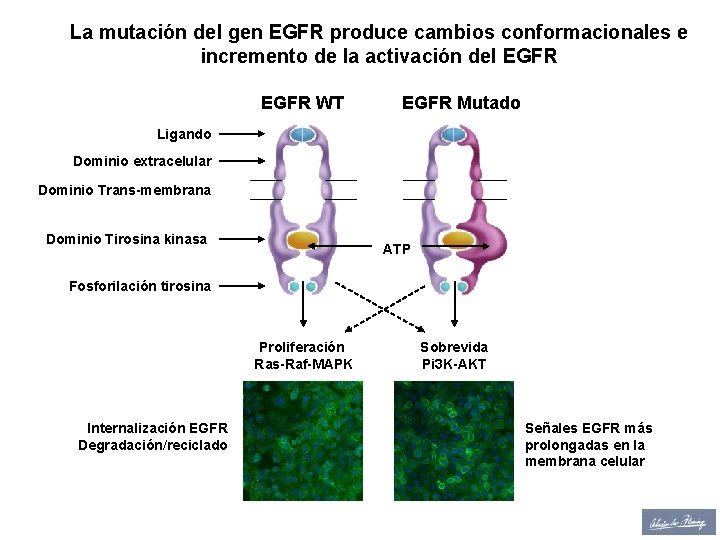
La mutación del gen EGFR produce cambios conformacionales e incremento de la activación del EGFR WT EGFR Mutado Ligando Dominio extracelular Dominio Trans-membrana Dominio Tirosina kinasa ATP Fosforilación tirosina Proliferación Ras-Raf-MAPK Internalización EGFR Degradación/reciclado Sobrevida Pi 3 K-AKT Señales EGFR más prolongadas en la membrana celular

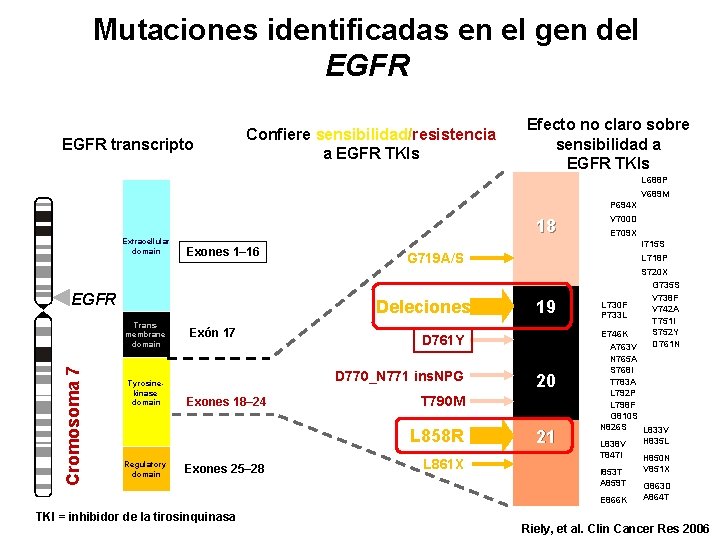
Mutaciones identificadas en el gen del EGFR transcripto Confiere sensibilidad/resistencia a EGFR TKIs Efecto no claro sobre sensibilidad a EGFR TKIs L 688 P V 689 M P 694 X 18 Extracellular domain Exones 1– 16 EGFR Cromosoma 7 Tyrosinekinase domain Exón 17 Exones 18– 24 G 719 A/S Exones 25– 28 L 718 P 19 D 761 Y 18 D 770_N 771 ins. NPG 20 T 790 M L 858 R Regulatory domain E 709 X I 715 S Deleciones Transmembrane domain V 700 D L 861 X 21 S 720 X G 735 S V 738 F L 730 F V 742 A P 733 L T 751 I S 752 Y E 746 K D 761 N A 763 V N 765 A S 768 I T 783 A L 792 P L 798 F G 810 S N 826 S L 833 V L 838 V T 847 I I 853 T A 859 T E 866 K TKI = inhibidor de la tirosinquinasa H 835 L H 850 N V 851 X G 863 D A 864 T Riely, et al. Clin Cancer Res 2006

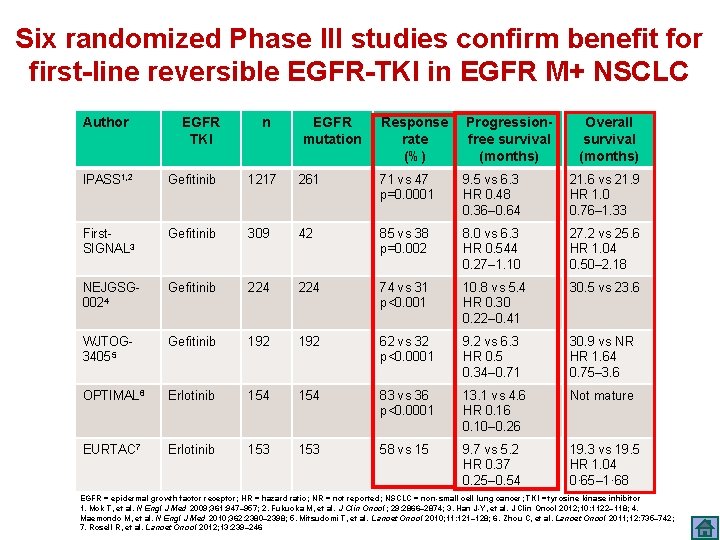
Six randomized Phase III studies confirm benefit for first-line reversible EGFR-TKI in EGFR M+ NSCLC Author EGFR TKI n EGFR mutation Response rate (%) Progressionfree survival (months) Overall survival (months) IPASS 1, 2 Gefitinib 1217 261 71 vs 47 p=0. 0001 9. 5 vs 6. 3 HR 0. 48 0. 36‒ 0. 64 21. 6 vs 21. 9 HR 1. 0 0. 76– 1. 33 First. SIGNAL 3 Gefitinib 309 42 85 vs 38 p=0. 002 8. 0 vs 6. 3 HR 0. 544 0. 27– 1. 10 27. 2 vs 25. 6 HR 1. 04 0. 50– 2. 18 NEJGSG 0024 Gefitinib 224 74 vs 31 p<0. 001 10. 8 vs 5. 4 HR 0. 30 0. 22– 0. 41 30. 5 vs 23. 6 WJTOG 34055 Gefitinib 192 62 vs 32 p<0. 0001 9. 2 vs 6. 3 HR 0. 5 0. 34– 0. 71 30. 9 vs NR HR 1. 64 0. 75– 3. 6 OPTIMAL 6 Erlotinib 154 83 vs 36 p<0. 0001 13. 1 vs 4. 6 HR 0. 16 0. 10– 0. 26 Not mature EURTAC 7 Erlotinib 153 58 vs 15 9. 7 vs 5. 2 HR 0. 37 0. 25– 0. 54 19. 3 vs 19. 5 HR 1. 04 0· 65– 1· 68 EGFR = epidermal growth factor receptor; HR = hazard ratio; NR = not reported; NSCLC = non-small cell lung cancer; TKI = tyrosine kinase inhibitor 1. Mok T, et al. N Engl J Med 2009; 361: 947– 957; 2. Fukuoka M, et al. J Clin Oncol; 29: 2866‒ 2874; 3. Han J-Y, et al. J Clin Oncol 2012; 10: 1122‒ 118; 4. Maemondo M, et al. N Engl J Med 2010; 362: 2380– 2398; 5. Mitsudomi T, et al. Lancet Oncol 2010; 11: 121– 128; 6. Zhou C, et al. Lancet Oncol 2011; 12: 735‒ 742; 7. Rosell R, et al. Lancet Oncol 2012; 13: 239– 246

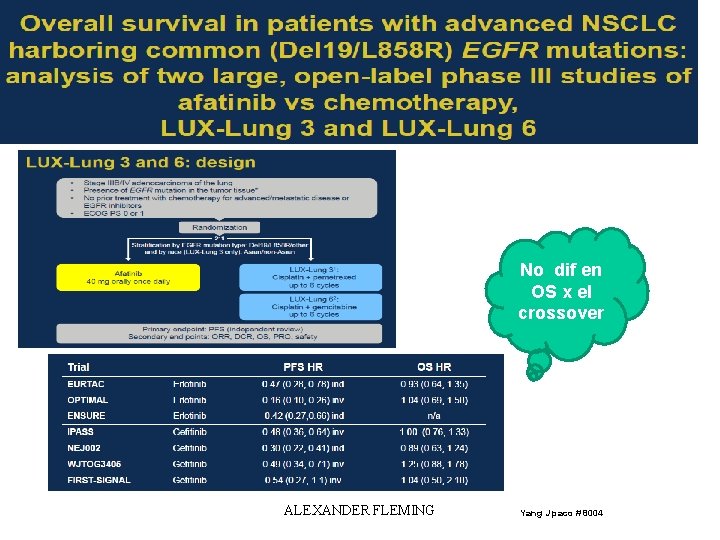
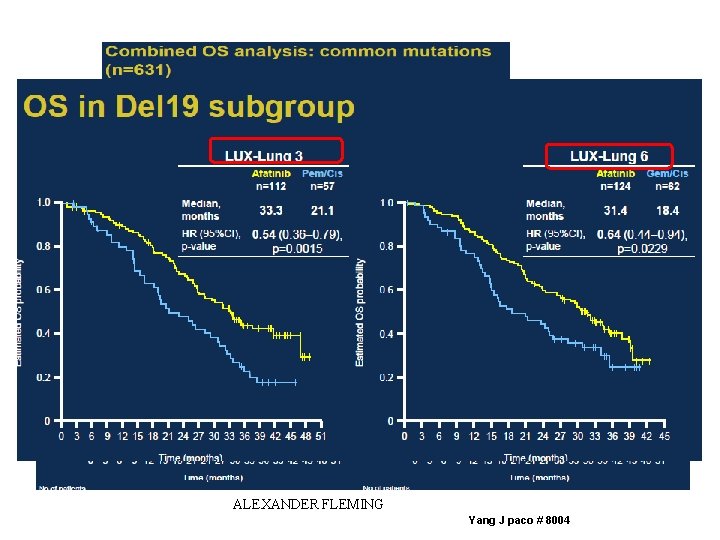
No dif en OS x el crossover ALEXANDER FLEMING Yang J paco # 8004

ALEXANDER FLEMING Yang J paco # 8004

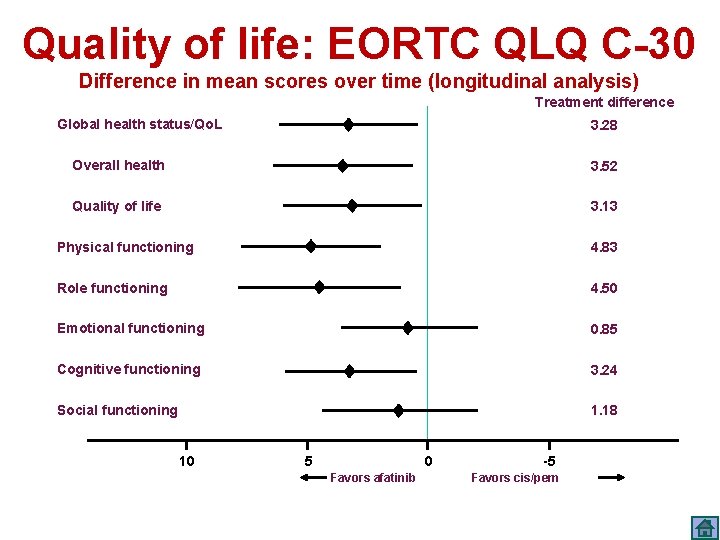
Quality of life: EORTC QLQ C-30 Difference in mean scores over time (longitudinal analysis) Treatment difference Global health status/Qo. L 3. 28 Overall health 3. 52 Quality of life 3. 13 Physical functioning 4. 83 Role functioning 4. 50 Emotional functioning 0. 85 Cognitive functioning 3. 24 Social functioning 1. 18 10 5 0 Favors afatinib -5 Favors cis/pem Yang JC, et al.

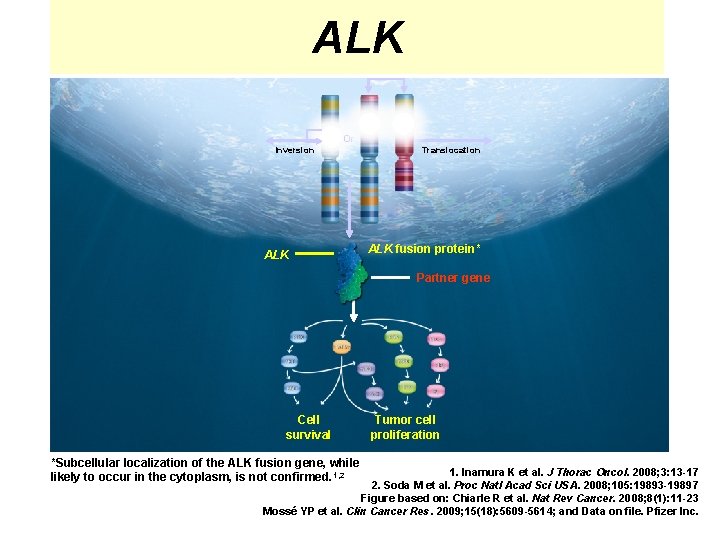
ALK Or Inversion ALK Translocation ALK fusion protein* Partner gene Cell survival *Subcellular localization of the ALK fusion gene, while likely to occur in the cytoplasm, is not confirmed. 1, 2 Tumor cell proliferation 1. Inamura K et al. J Thorac Oncol. 2008; 3: 13 -17 2. Soda M et al. Proc Natl Acad Sci USA. 2008; 105: 19893 -19897 Figure based on: Chiarle R et al. Nat Rev Cancer. 2008; 8(1): 11 -23 Mossé YP et al. Clin Cancer Res. 2009; 15(18): 5609 -5614; and Data on file. Pfizer Inc.

EML 4 -ALK fusion variants in NSCLC ● Several EML 4 -ALK fusion variants have been identified in NSCLC that demonstrate gain-of-function properties. ● ALK tyrosine kinase activity is required for transforming activity. ● Evidence shows ALK inhibitors lead to tumor shrinkage in vivo, and suggests oncogene addiction, and the potential target for therapeutic intervention. EML 4 -ALK E 13; A 20 E 6; A 20 E 20; A 20 E 14; A 20 E 18; A 20 E 15; A 20 EML 4 TFG-ALK KIF 5 B-ALK unknown ALK E 17; 20 ALK EML 4 E 13; A 20 Variant 1 33% ALK EML 4 E 2; A 20 E 17; A 20 ALK EML 4 TFG KIF 5 B Coiled-coil domain E 6 a/b; A 20 Variant 3 a/b 29% E 2; 20 E 15; 20 E 18; 20 E 14; 20 E 20; 20 ALK E 13; 20 ALK E 6 a/b; 20 Tyrosine kinase domain Adapted from Sasaki et al. , Eur J Cancer 2010; 46: 1773 -1780

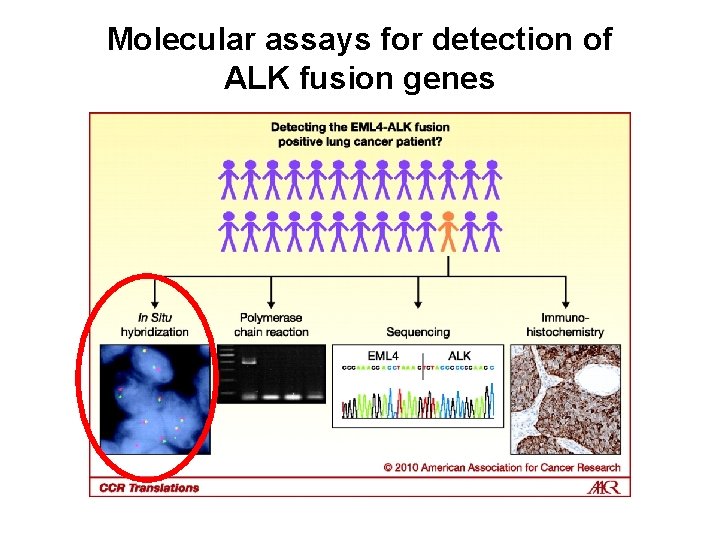
Molecular assays for detection of ALK fusion genes Hirsch et al. , Clin Cancer Res 2010; 16: 4909– 4911.

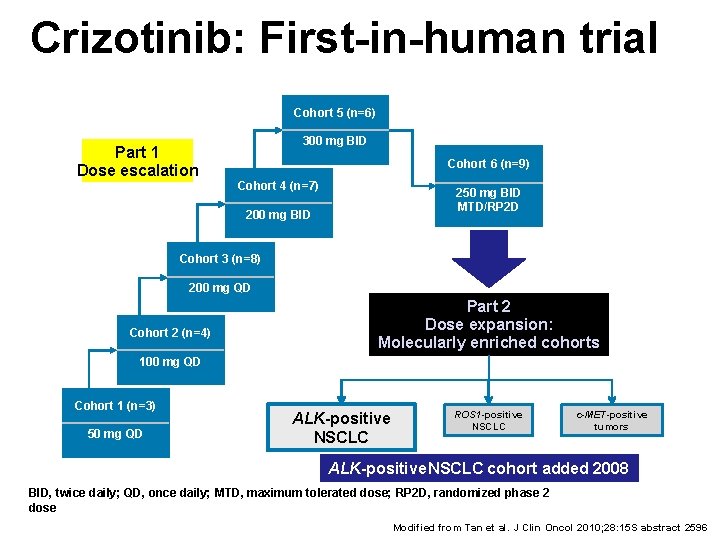
Crizotinib: First-in-human trial Cohort 5 (n=6) Part 1 Dose escalation 300 mg BID Cohort 6 (n=9) Cohort 4 (n=7) 250 mg BID MTD/RP 2 D 200 mg BID Cohort 3 (n=8) 200 mg QD Cohort 2 (n=4) Part 2 Dose expansion: Molecularly enriched cohorts 100 mg QD Cohort 1 (n=3) 50 mg QD ALK-positive NSCLC ROS 1 -positive NSCLC c-MET-positive tumors ALK-positive NSCLC cohort added 2008 BID, twice daily; QD, once daily; MTD, maximum tolerated dose; RP 2 D, randomized phase 2 dose Modified from Tan et al. J Clin Oncol 2010; 28: 15 S abstract 2596

Open-label, Multicenter, Phase II Study of Crizotinib in Advanced ALK-positive Non-Small Cell Lung Cancer PROFILE 1005 • ALK+ NSCLC with measureable lesions • ECOG PS: 0– 3 • Not eligible for Phase 3 study (A 8081007) • PD in chemo arm of study A 8081007 • ≥ 1 prior platinumbased chemotherapy • Stable/controlled brain metastases allowed N=400 (planned) Crizotinib 250 mg BID administered continuously (21 -day cycle) Primary endpoints: ORR, safety, and tolerability Secondary endpoints: OS, TTR, duration of response, disease control rate, PK, biomarkers, PRO/HRQo. L (EORTC QLQ-C 30 and LC-13) ALEXANDER FLEMING/ MARIA FERRER

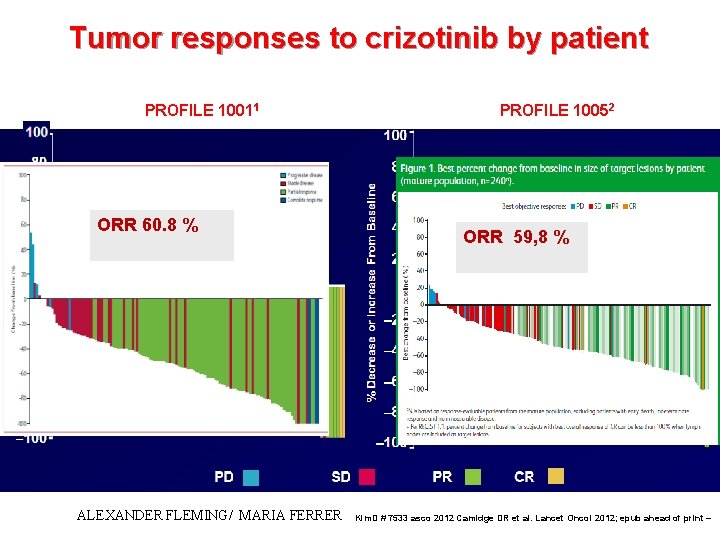
Tumor responses to crizotinib by patient PROFILE 10011 PROFILE 10052 Median time to response: 8 wk ORR 60. 8 % RR 61% ALEXANDER FLEMING/ MARIA FERRER ORR 59, 8 % RR 51 % 1. Camidge et al. , ASCO 2011; Abs #2501 2. Riely et al. , IASLC 2011; Abs #O 31. 05 Kim. D # 7533 asco 2012 Camidge DR et al. Lancet Oncol 2012; epub ahead of print –

CONCLUSIONS Platinum Doublets remains as standart treatment for patients without driver mutations Pemetrexed and Bevacizumab are candidates for non squamous cell carcinoma No clear benefit to add pemetrexed to bevacizumab in first line Chemotherapy is first option for EGFR WT in first and second line

CONCLUSIONES LA DETECCIÓN DE LOS GENES DE ALK Y EGFR Y SU TRATAMIENTO MODIFICA DRÁSTICAMENTE LA EXPECTATIVA DE VIDA EN PACIENTES CON CPNCP. TODOS LOS MÉDICOS QUE PARTICIPAN DEL CUIDADO DEL PACIENTE CON CÁNCER PULMONAR DEBEN SER PROACTIVOS EN LA BÚSQUEDA DE TEJIDO PARA REALIZACÍON DE ESTUDIOS MOLECULARES. EN UN FUTURO CERCANO NUEVAS TERAPIAS CONTRA DRIVERS ONCOGÉNICOS ESTARÁN DISPONIBLES Y SERÁ UN DESAFÍO LA OBTENCIÓN Y OPTIMIZACIÓN DEL MATERIAL

AAMR 2014 Claudio. martin@fibertel. com. ar ALEXANDER FLEMING