TRANSPORT PROCESSES OF ACTIVE AGENTS INDUCING THE BIFLUX

TRANSPORT PROCESSES OF ACTIVE AGENTS INDUCING THE BI-FLUX PHENOMENON Luiz Bevilacqua – COPPE/UFRJ Maosheng Jiang - COPPE/UFRJ

INVADING SPECIES
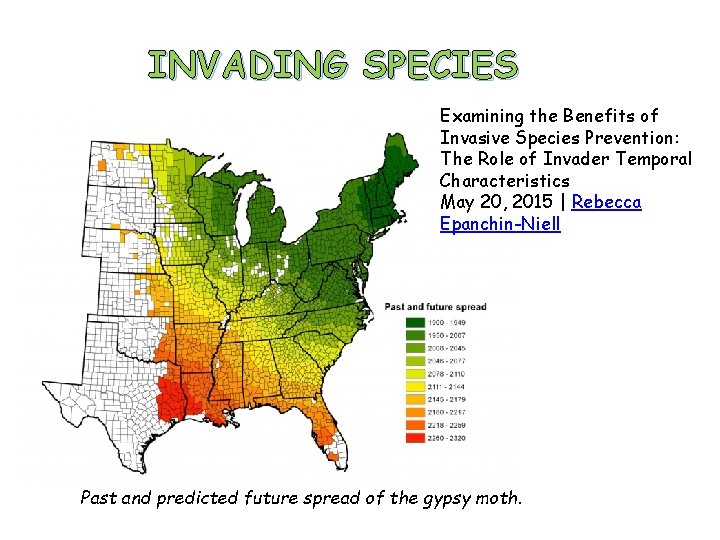
INVADING SPECIES Examining the Benefits of Invasive Species Prevention: The Role of Invader Temporal Characteristics May 20, 2015 | Rebecca Epanchin-Niell Past and predicted future spread of the gypsy moth.
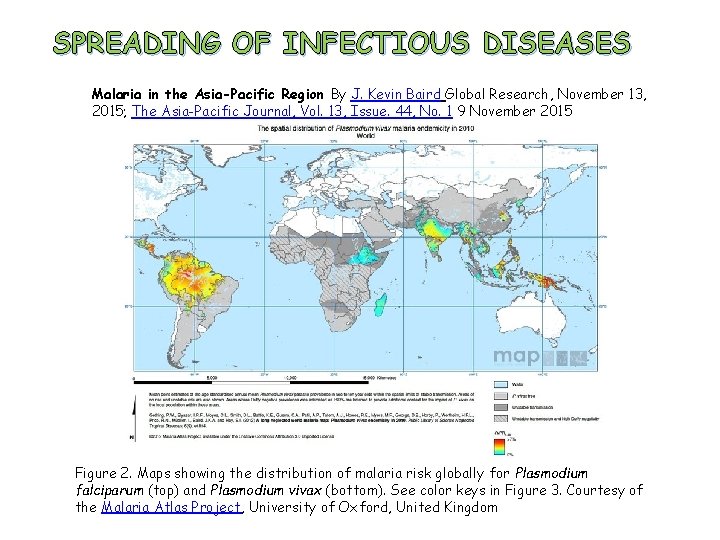
SPREADING OF INFECTIOUS DISEASES Malaria in the Asia-Pacific Region By J. Kevin Baird Global Research, November 13, 2015; The Asia-Pacific Journal, Vol. 13, Issue. 44, No. 1 9 November 2015 Figure 2. Maps showing the distribution of malaria risk globally for Plasmodium falciparum (top) and Plasmodium vivax (bottom). See color keys in Figure 3. Courtesy of the Malaria Atlas Project, University of Oxford, United Kingdom

CLASSICAL APPROACH: REGULAR AND ANOMALOUS PROCESSES 1. Extra terms intended to introduce appropriates corrections in theory to match experimental results 2. Expand the flux potential D∂p/∂x to include higher order terms: D 1 ∂p/∂x + D 2 ∂2 p/∂x 2 +(D 3 ∂p/∂x)2 + D 4 ∂4 p/∂x 4 + … Other types of phenomena: MBE (thin films deposition), lubrication, etc.

ARE THERE REFERENCES OR EXPERIMENTAL EVIDENCE OF RETENTION PHENOMENA IN ANY PHYSIC-CHEMISTRY OR BIOLOGICAL PROCESS?

SOME EXAMPLES OF DIFFUSION WITH RETENTION 1. Diffusion and Retention Processes in a Bentonite Near Field (WP 2. 5) NF-PRO Annual Meeting, 2006, El Escorial, Spain (Euro-Comm) 2. Extracellular space structure revealed by diffusion analysis; C. Nicholson, E. Syková 3. Retention phenomena in non-Newtonian fluids flow, 2003, D’Angelo, Fontana, Chertco, Rosen 4. Hydrogen bulk retention in graphite and kinetics of diffusion, 2002, H. Atsumi 5. Effect of Liquid-Phase Diffusion Resistance on Retention Time in Gas-Liquid Chromatography, Jan-Chan Huang. Richard Madey 6. Laboratory determination of water retention and diffusion coefficient in unsaturated sand, 2004, K. Badv and M. R. Faridfard

where N is the local concentration of free hydrogen atom, N 1 is that of trapped hydrogen atom, C is the concentration of vacant trap, D is the true diffusion coefficient of free hydrogen, ∑d is thermal detrapping rate constant for trapped hydrogen and ∑t is the trappingrate constant for free hydrogen


Hydraulic, Diffusion, and Retention Characteristics of Inorganic Chemicals in Bentonite (Ph. D Thesis) Naim Muhammad


PHYSICA A 327 (2003) 44 -48
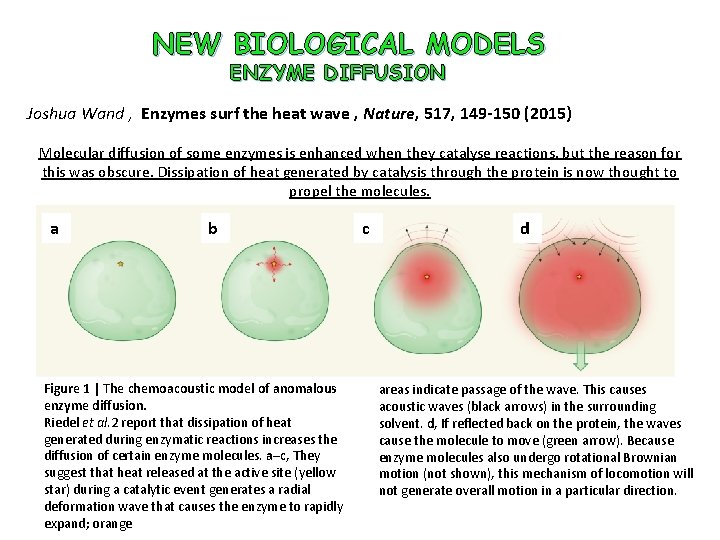
NEW BIOLOGICAL MODELS ENZYME DIFFUSION Joshua Wand , Enzymes surf the heat wave , Nature, 517, 149 -150 (2015) Molecular diffusion of some enzymes is enhanced when they catalyse reactions, but the reason for this was obscure. Dissipation of heat generated by catalysis through the protein is now thought to propel the molecules. a b Figure 1 | The chemoacoustic model of anomalous enzyme diffusion. Riedel et al. 2 report that dissipation of heat generated during enzymatic reactions increases the diffusion of certain enzyme molecules. a–c, They suggest that heat released at the active site (yellow star) during a catalytic event generates a radial deformation wave that causes the enzyme to rapidly expand; orange c d areas indicate passage of the wave. This causes acoustic waves (black arrows) in the surrounding solvent. d, If reflected back on the protein, the waves cause the molecule to move (green arrow). Because enzyme molecules also undergo rotational Brownian motion (not shown), this mechanism of locomotion will not generate overall motion in a particular direction.

NEW BIOLOGICAL MODELS ENZYME DIFFUSION Nature, 517, 227 -230 (2015) The heat released during catalytic turnover enhances the diffusion of an enzyme Clement Riedel 1 , Ronen Gabizon , Christian A. M. Wilson, Kambiz Hamadani 1, Konstantinos Tsekouras , Susan Marqusee, Steve Presse & Carlos Bustamante A stochastic model (detailed in the Supplementary Information and Extended Data Figs 6, 7 and 8) describes the enhanced diffusion upon catalysis in terms of the heat released by the chemical reaction. In this model, we assume that enzymes transiently diffuse more quickly—with diffusion coefficient D 1 —for some short period of time, dt, following a chemical reaction. Otherwise, the enzyme displays its diffusion coefficient in the absence of substrate, D 0. The net diffusion coefficient in the presence of substrate, D, is therefore the ensemble average over both subpopulations with the probability of observing an enhanced diffusion proportional to V, the reaction rate. We then relate the enhanced diffusion coefficient, D 1, to the amount of heat, Q, evolved by an enzymatic reaction. To do so, we assume that the kinetic energy of the enzyme’s centre-of-mass immediately following a reaction is proportional to some fraction c of Q. From this simple model, we obtain the following expression which shows a diffusion coefficient enhancement linear in V and Q, wheremis the mass of the enzyme, dt 5 m/f is the relaxation timescale associated with the enzyme displacement following an enzymatic turnover, and f is an effective friction coefficient for the enzyme (see Supplementary Materials)

CONTINUUM FORMULATION OF DISTURBED (DELAYED) DIFFUSION PROCESSES

A NEW ANALYTICAL FORMULATION OF DISTURBING EFFECTS ON PARTICLE DIFFUSION PROCESSES INTRODUCTION OF A BIMODAL SPEED DISTRIBUTION TO MASS TRANSFER PROBLEMS

FUNDAMENTAL HYPOTHESIS I. The system is self-contained in the sense that all possible internal reactions preserve the total mass in the diffusion process. II. There are neither sinks nor sources in the system. All particles are under a continuous state of excitation, that is, moving continuously. III. There are two distinct energy states E 1(p 1, L 1) and E 2(p 2, L 2), corresponding to the two subsets of particles, β and (1− β) respectively, where p represents the linear momentum and L the angular momentum. IV. Particles in the system are either in the energy state I or in the energy state II
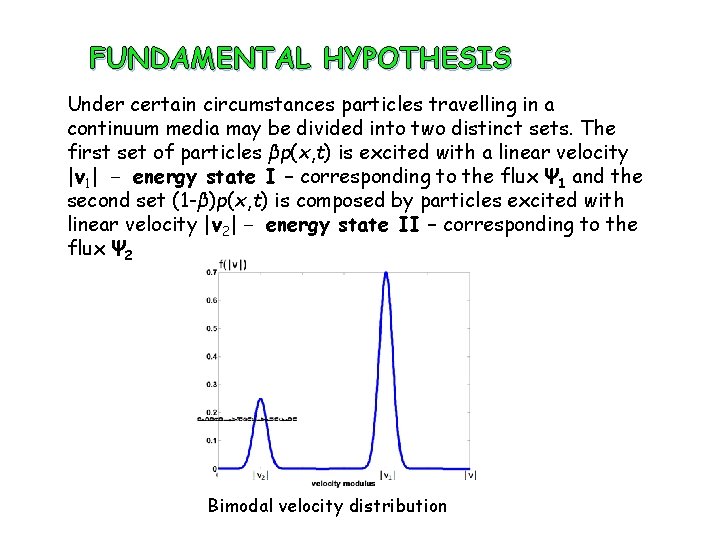
FUNDAMENTAL HYPOTHESIS Under certain circumstances particles travelling in a continuum media may be divided into two distinct sets. The first set of particles βp(x, t) is excited with a linear velocity |v 1| − energy state I – corresponding to the flux Ψ 1 and the second set (1 -β)p(x, t) is composed by particles excited with linear velocity |v 2| − energy state II – corresponding to the flux Ψ 2 Bimodal velocity distribution

DERIVATION OF THE FUNDAMENTAL EQUATION THE DISCRETE APPROACH
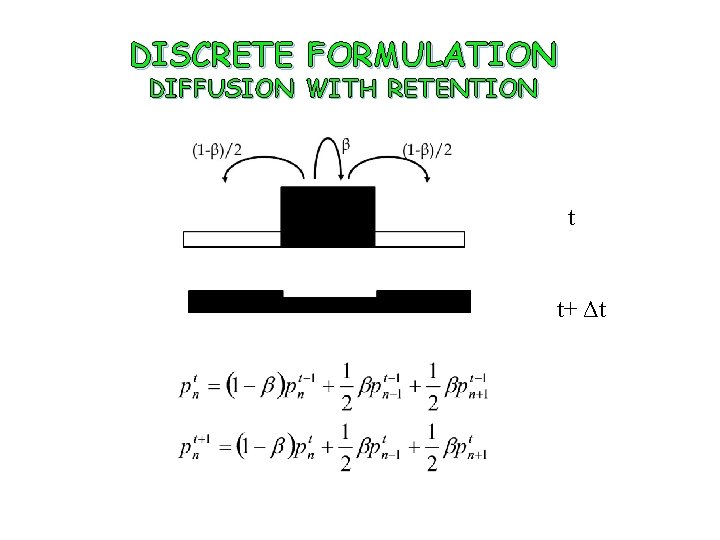
DISCRETE FORMULATION DIFFUSION WITH RETENTION t t+ Δt

DISCRETE APPROACH TO DIFFUSION PHENOMENA Reduce the right hand side terms to the reference time t-1 Subtract to get

DISCRETE APPROACH TO DIFFUSION PHENOMENA Taking the limit when Δx → 0 and Δt → 0 and assuming p(x, t) sufficiently smooth the continuum equivalent to the discrete formulation is obtained:

GENERALIZED BI-FLUX DIFFUSION EQUATION From the discrete approach to the mass conservation principle Equation for the bi-flux diffusion Primary Flux (Fick´s law) D: diffusion coefficient Secondary Flux. Subsidiary to the principal flux R: reactivity coefficient
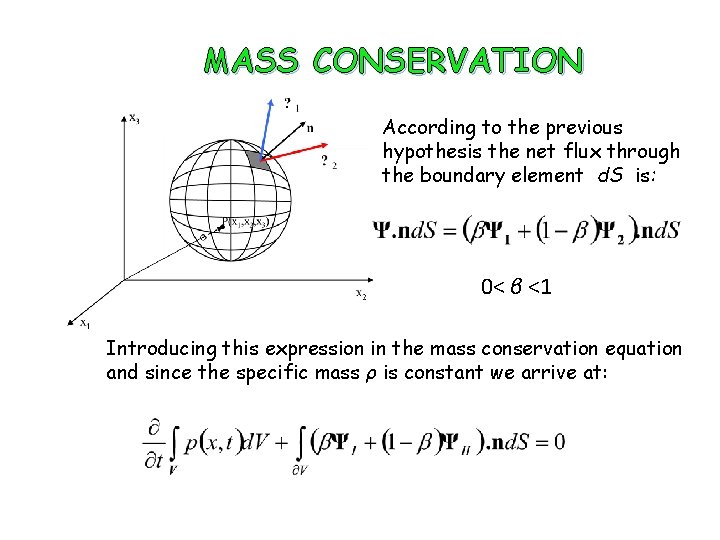
MASS CONSERVATION According to the previous hypothesis the net flux through the boundary element d. S is: 0< β <1 Introducing this expression in the mass conservation equation and since the specific mass ρ is constant we arrive at:
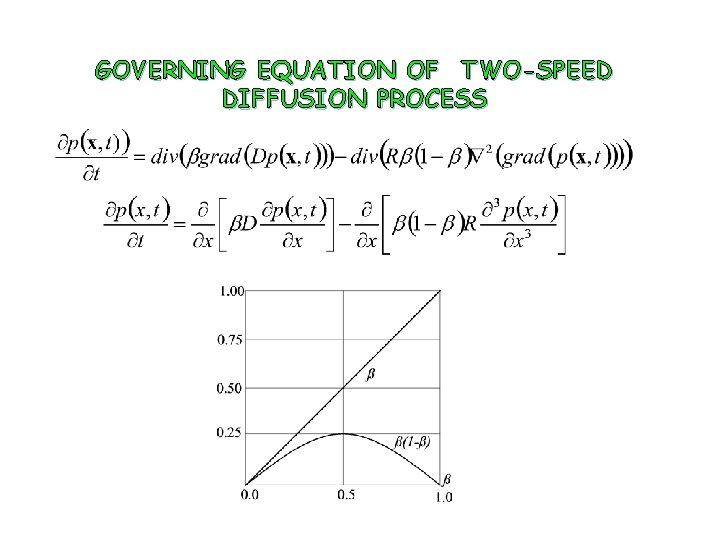
GOVERNING EQUATION OF TWO-SPEED DIFFUSION PROCESS

PHYSICAL MEANING OF THE BOUNDARY CONDITIONS ARE DETERMINED ONCE WE KNOW AT THE BOUNDARY: The concentration : The gradient of the concentration : Ψ 1 The time variation of the concentration: The gradient of the time variation of the concentration : Ψ 2

THE DIFFERENT TYPES OF SOLUTIONS FOR THE FOURTH ORDER EQUATION Two cases of closed solutions will be considered
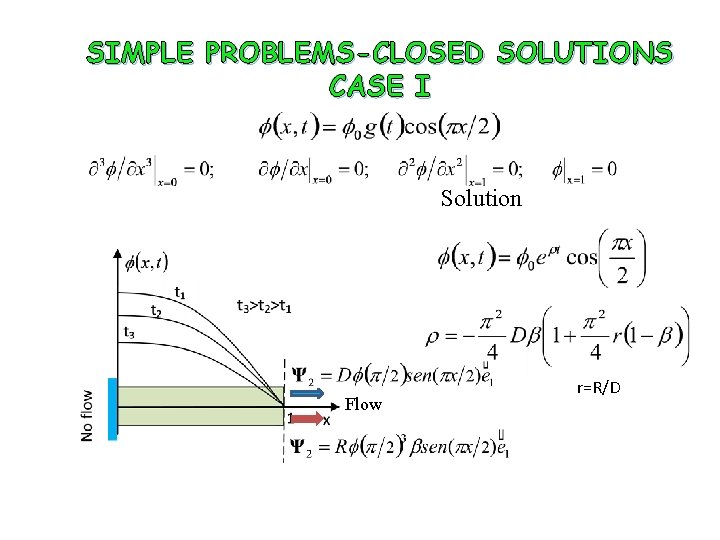
SIMPLE PROBLEMS-CLOSED SOLUTIONS CASE I Solution Flow r=R/D
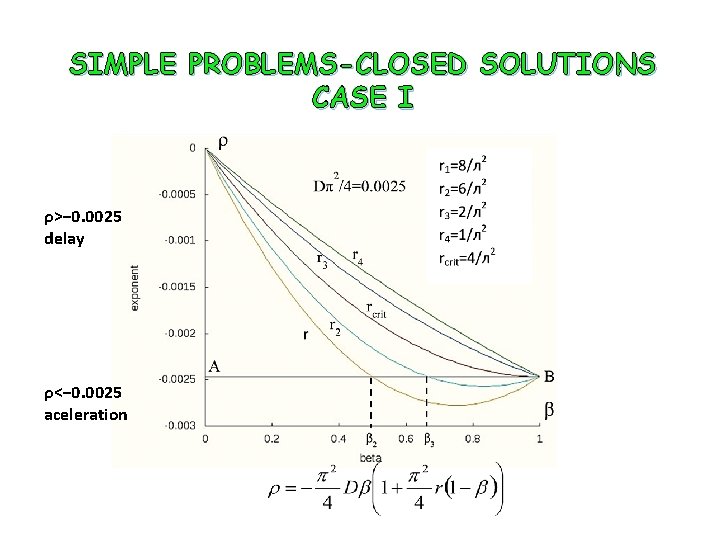
SIMPLE PROBLEMS-CLOSED SOLUTIONS CASE I ρ>− 0. 0025 delay ρ<− 0. 0025 aceleration
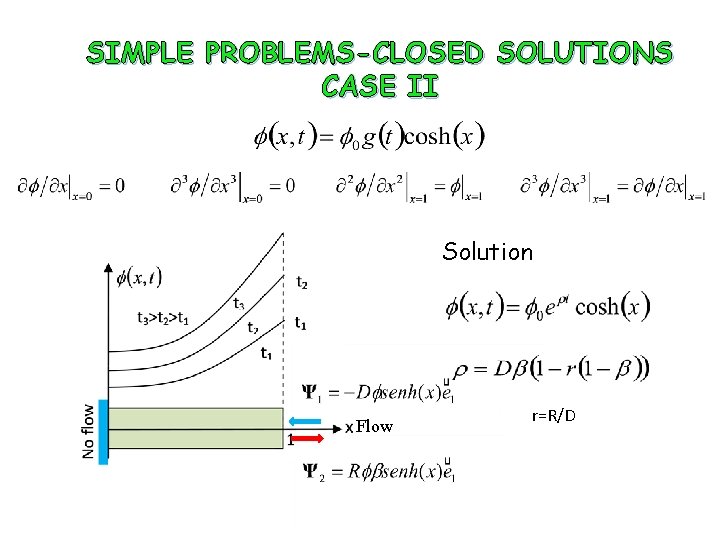
SIMPLE PROBLEMS-CLOSED SOLUTIONS CASE II Solution Flow r=R/D
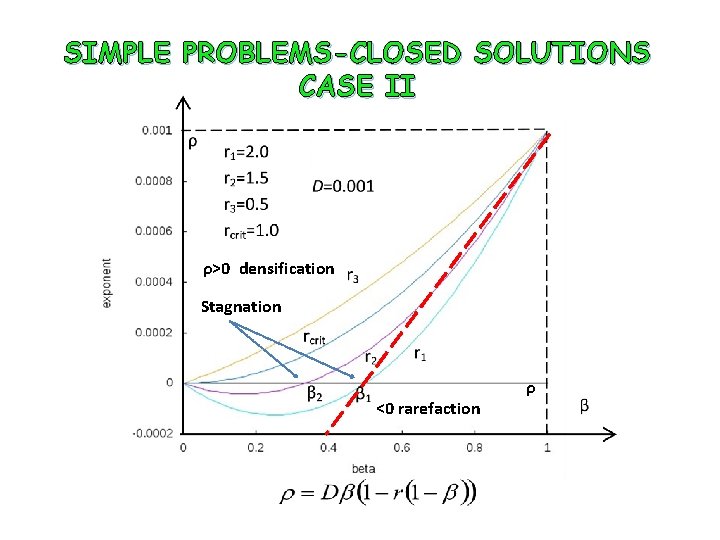
SIMPLE PROBLEMS-CLOSED SOLUTIONS CASE II ρ>0 densification Stagnation ρ <0 rarefaction
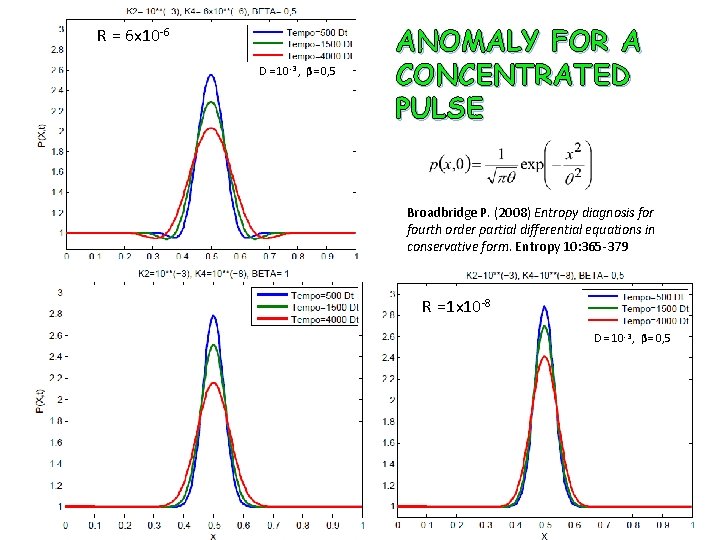
R = 6 x 10 -6 D = 10 -3 , β= 0, 5 ANOMALY FOR A CONCENTRATED PULSE Broadbridge P. (2008) Entropy diagnosis for fourth order partial differential equations in conservative form. Entropy 10: 365 -379 R =1 x 10 -8 D = 10 -3 , β= 0, 5
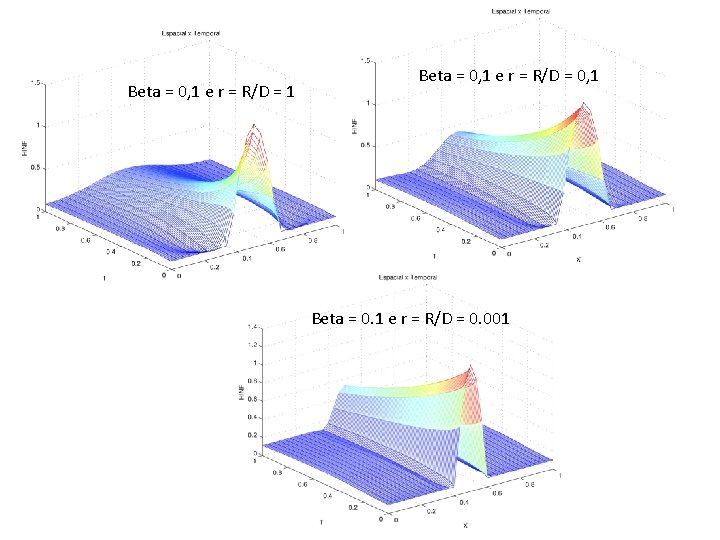
Beta = 0, 1 e r = R/D = 1 Beta = 0, 1 e r = R/D = 0, 1 Beta = 0. 1 e r = R/D = 0. 001
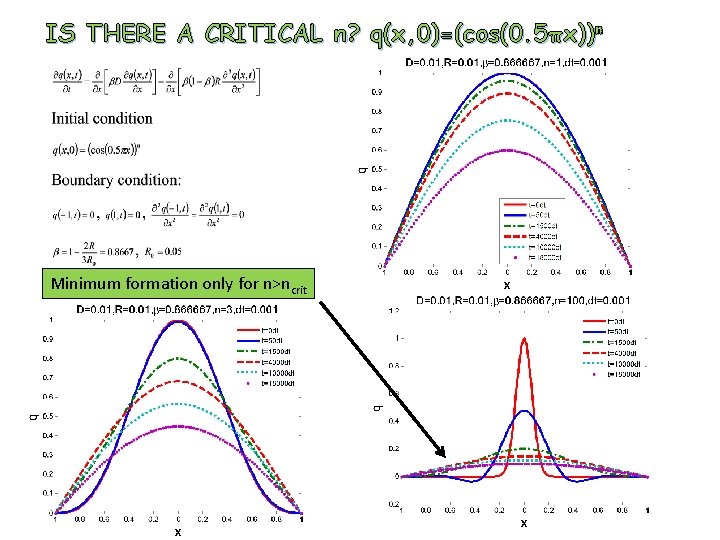
IS THERE A CRITICAL n? q(x, 0)=(cos(0. 5πx))n Minimum formation only for n>ncrit
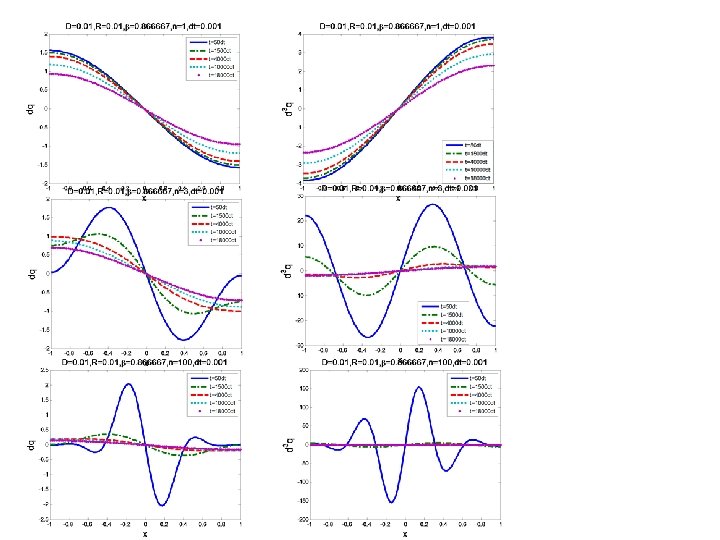

ANISOTROPIC DIFFUSION A key question for the anisotropic case is the definition of the secondary flux. We will test the following laws of formation: There are other possible laws for the secondary flux. The definition of the functional dependence of the secondary flux on β, R and p(x, t) is of fundamental importance for the concentration evolution. For the case the concentration initially tends to grow very fast for regions with high values of R. This behavior may simulate the population concentration around places with high nutrients density, or money flux towards places with high revenues.

ANISOTROPIC DIFFUSION Fundamental equations Case 1 Case 2

ANISOTROPIC AND COUPLED DIFFUSION R(x, t) could be nutrient or pheromones for population dynamics R p

ARE R AND β CORRELATED? After the research developed at IPRJ and recently at UFPB there are convincing arguments to assume a functional relation β = F(R) Possible relations: β= 1– 2 R/3 R 0 β = exp(–k. R) [30] L. G. Silva, D. C. Knupp, L. Bevilacqua, A. C. R. Galeão, A. J. Silva Neto, Inverse. Problem in Anomalous Diffusion with Uncertainty Propagation, Computer Assisted Methods in Mechanics and Science, 21: 245– 255, 2014 ISSN 2299 -3649 [31] Jairo R. de Faria, Ana P. P. Wyse, Antonio J. B. dos Santos, Luiz Bevilacqua, Flavio P. Costa, Second Order Topological Derivative for the Inverse Problem in Diffusion with Retention, CILAMCE 2015, PUC-RJ, 22 -25 Nov 2015

SOME EXAMPLES 1 -D ANISOTROPIC MEDIUM Case 1: R=R(x) see figure Case 2: R - Diffusing Case 3: R – decrease exponentially exp(-0. 2 t)
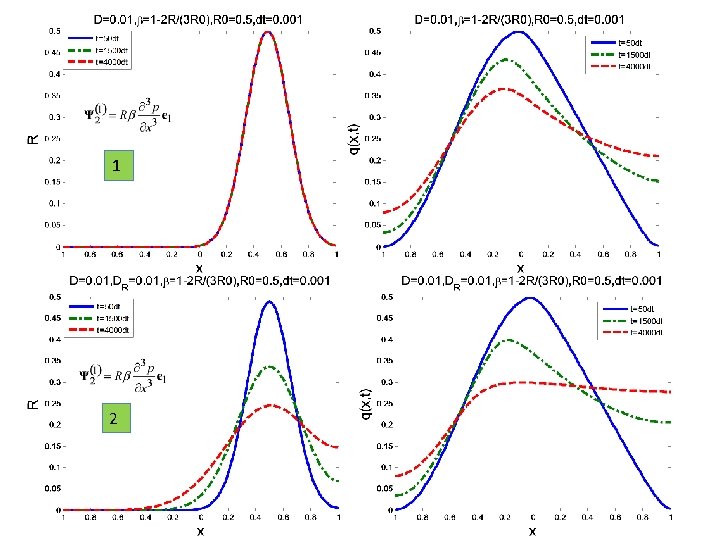
1 2
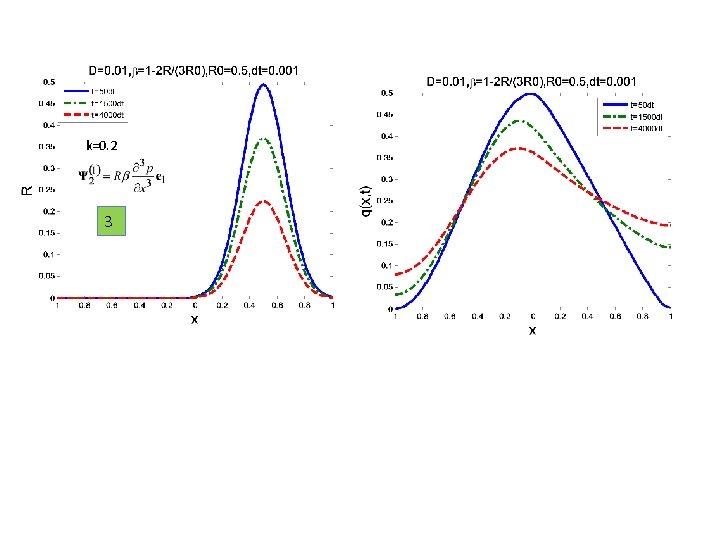
k=0. 2 3
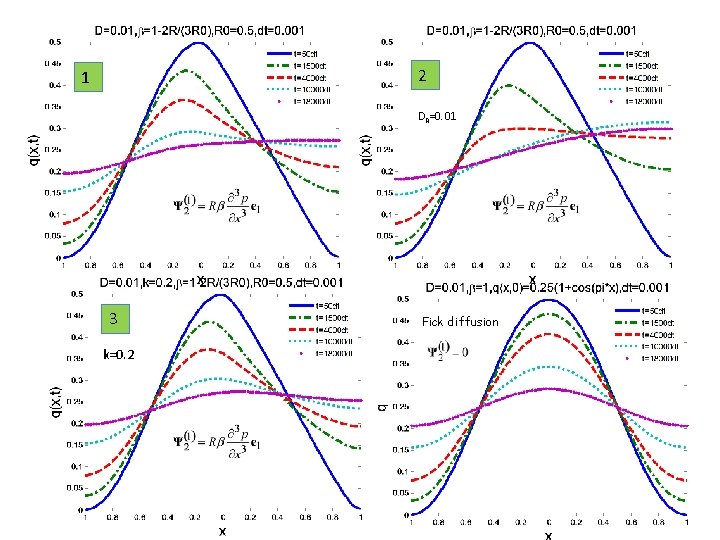
2 1 DR=0. 01 3 k=0. 2 Fick diffusion
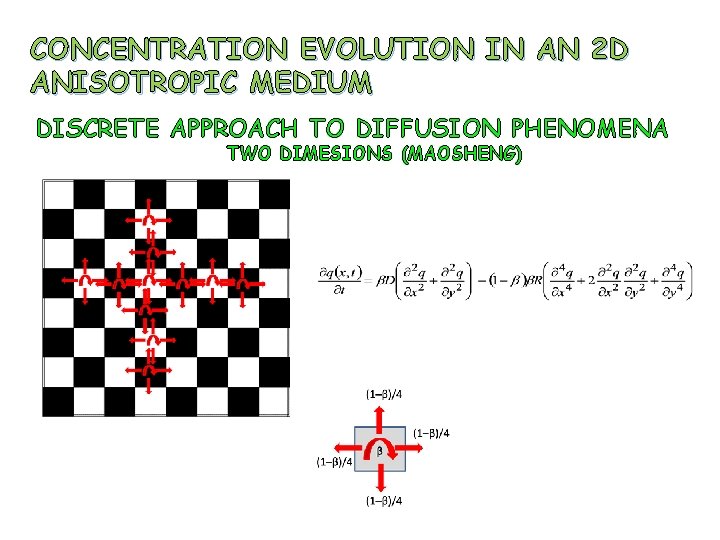
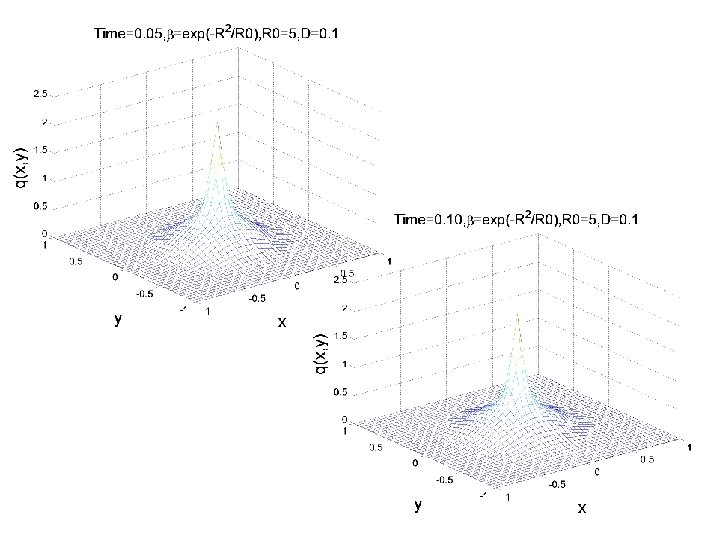
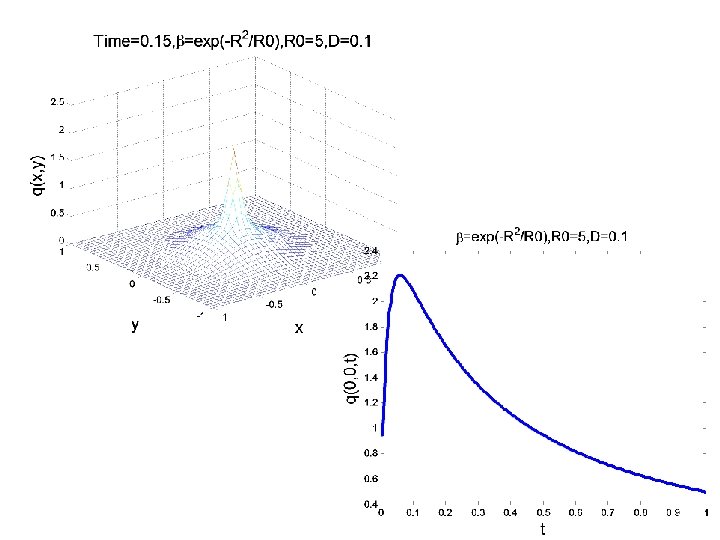
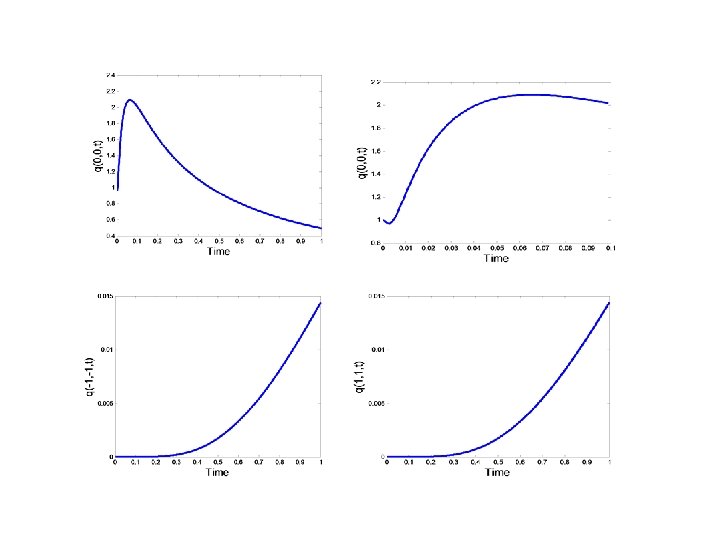
CONCENTRATION EVOLUTION IN AN 2 D ANISOTROPIC MEDIUM DISCRETE APPROACH TO DIFFUSION PHENOMENA TWO DIMESIONS (MAOSHENG)

CONCENTRATION EVOLUTION IN 2 D ANISOTROPIC MEDIUM Initial condition as shown Boundary condition: The parameter R is function of x, y
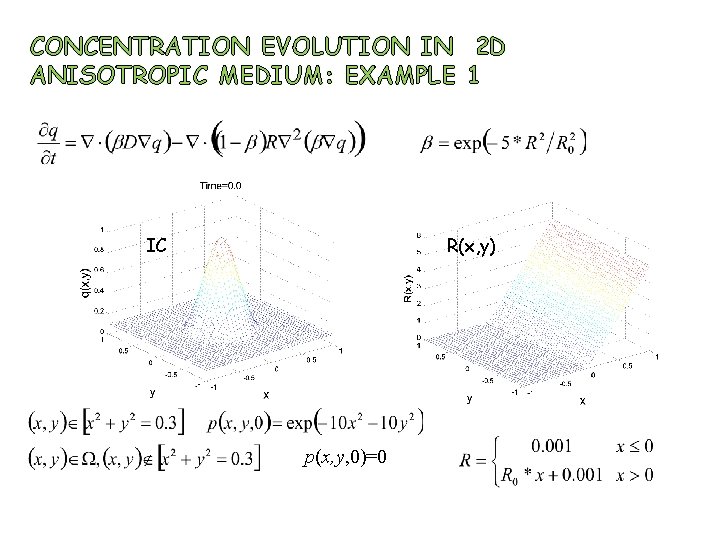
CONCENTRATION EVOLUTION IN 2 D ANISOTROPIC MEDIUM: EXAMPLE 1 IC R(x, y) p(x, y, 0)=0
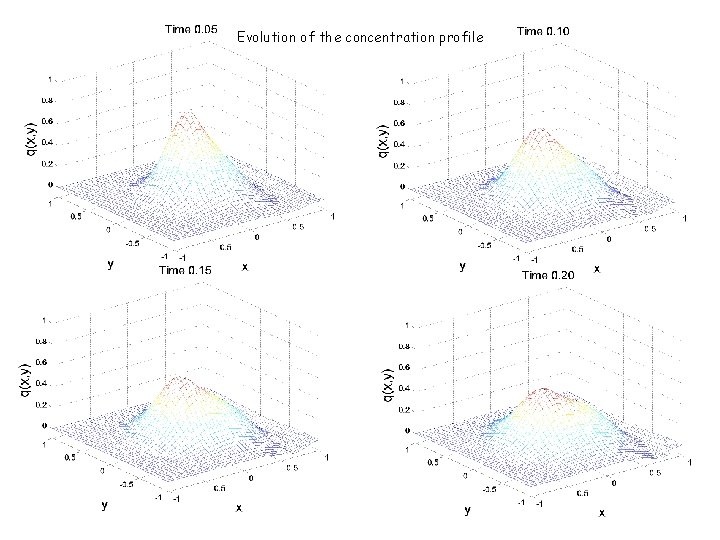
Evolution of the concentration profile
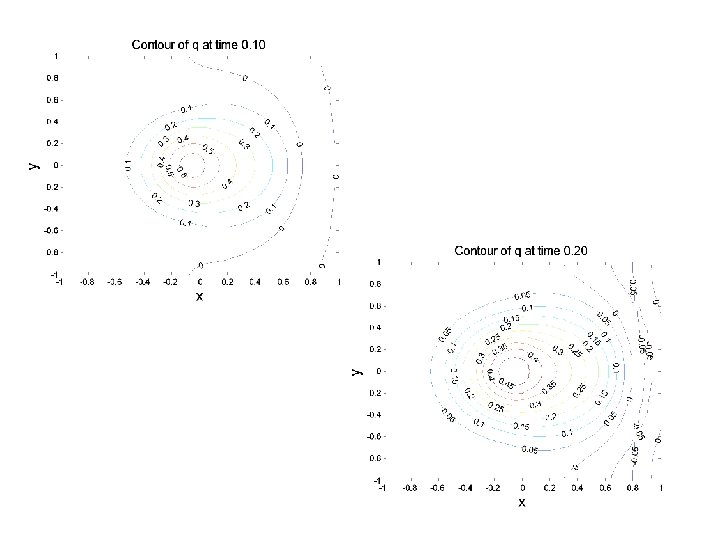
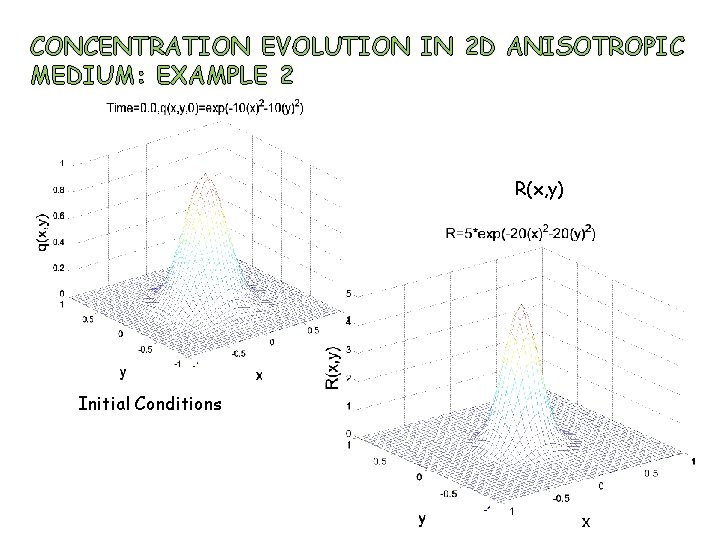
CONCENTRATION EVOLUTION IN 2 D ANISOTROPIC MEDIUM: EXAMPLE 2 R(x, y) Initial Conditions
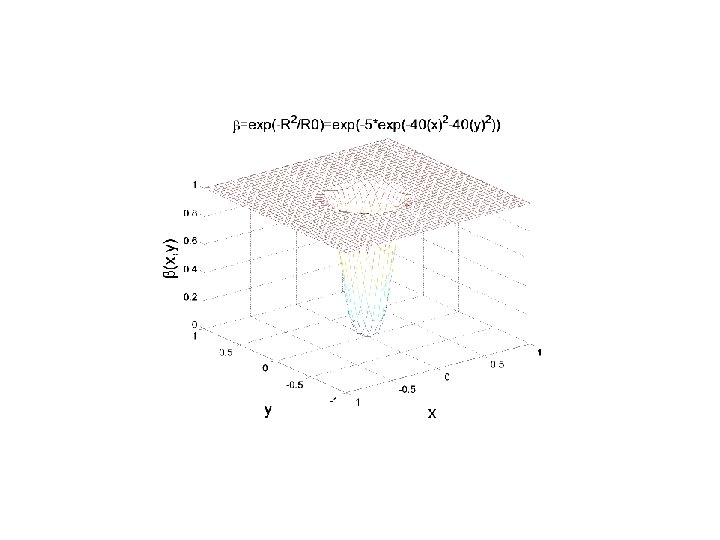

CAPITAL FLUX ( Black&Scholes )
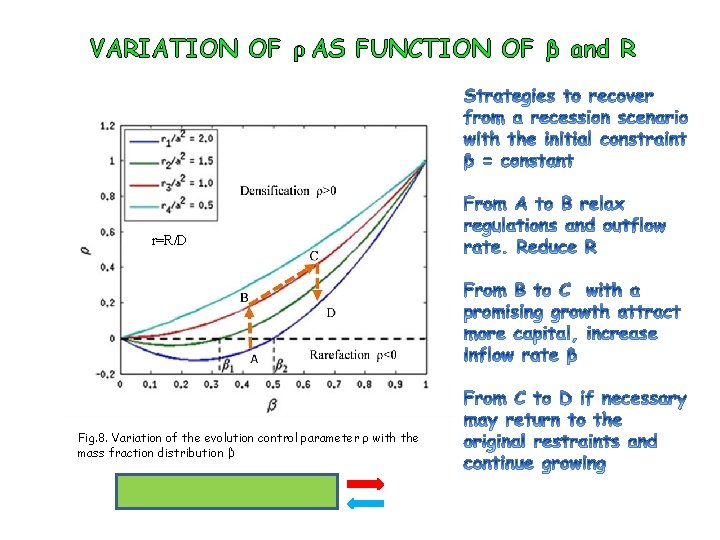
VARIATION OF ρ AS FUNCTION OF β and R r=R/D Fig. 8. Variation of the evolution control parameter ρ with the mass fraction distribution β

VARIATION OF Ρ AS FUNCTION OF β , R AND THE SOURCE INTENSITY α THE SOURCE TERM COMPENSATE THE OUTFLOW VOLUME PROPORTIONAL TO (1 -β) α =A/D r =R/D Fig. 9. Influence of the parameter r on the evolution of an economic chain for a given source α=2. 0. r 1 = 10, r 2 =5. 8, r 3 =3. 5. Parameter controlling the evolution of the process Fig 10. Influence of the source intensity on the evolution of an economic chain for (a) r=4; α 1=5, α 2=2, α 3=1, α 4=0. 1;
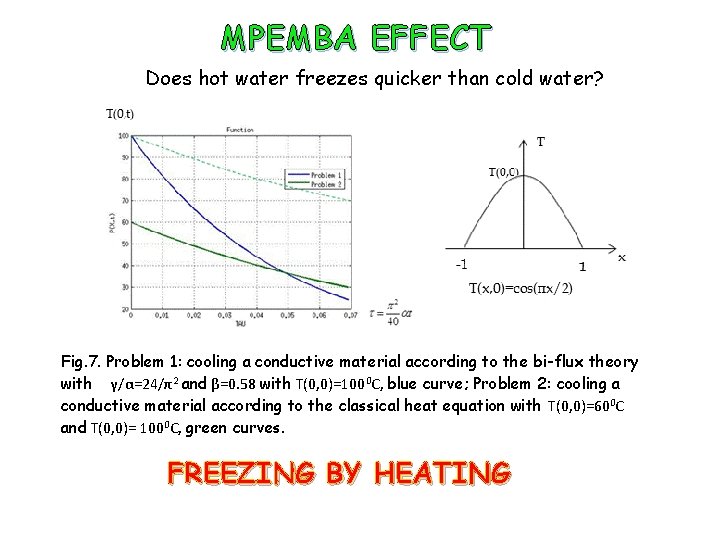
MPEMBA EFFECT Does hot water freezes quicker than cold water? Fig. 7. Problem 1: cooling a conductive material according to the bi-flux theory with γ/α=24/π2 and β=0. 58 with T(0, 0)=1000 C, blue curve; Problem 2: cooling a conductive material according to the classical heat equation with T(0, 0)=600 C and T(0, 0)= 1000 C, green curves. FREEZING BY HEATING

REFFERENCES L. Bevilacqua, A. C. N. Galeão, and F. P. Costa, A new analytical formulation of retention effects on particle diffusion processes, Proc. Braz. Acad. of Science. , 83(4), 1443‒ 1464, 2011. L. Bevilacqua, A. C. N. R. Galeão, J. G. Simas and A. P. Rio Doce, A new theory for anomalous diffusion with a bimodal flux distribution, J Braz. Soc. Mech. Sci. Eng. , 35, 431– 440, 2013 Silva, L. G. , Knupp, D. C. , Bevilacqua, L. , Galeão, A. C. N. R. , Simas, J. G. , Vasconcellos, J. F. V. e Silva Neto, A. J. , Investigation of a New Model for Anomalous Diffusion Phenomena by Means of an Inverse Analysis, Proc. 4 th Inverse Problems, Design and Optimization Symposium (IPDO-2013), Albi, França, 2013. Silva, L. G. , Knupp, D. C. , Bevilacqua, L. , Galeão, A. C. N. R. e Silva Neto, A. J. , Inverse Problem in Anomalous Diffusion with Uncertainty Propagation, Proc. 8 th International Conference on Inverse Problems in Engineering: Theory and Practice, Cracow, Poland, 2014. [31] Jairo R. de Faria, Ana P. P. Wyse, Antonio J. B. dos Santos, Luiz Bevilacqua, Flavio P. Costa, Second Order Topological Derivative for the Inverse Problem in Diffusion with Retention, CILAMCE 2015, PUC-RJ, 22 -25 Nov 2015 L. Bevilacqua, M. Jiang, A. Silva. Neto, A. C. R. N. Galeão, An Evolutionary Model of Bi-Flux Diffusion Processes, J Braz. Soc. Mech. Sci. Eng. (2016)

FORMAÇÃO DE UM GRUPO DE PESQUISA IPRJ, UFPB, UESC, UFRJ DIFUSÃO E TRANSPORTE DE PARTÍCULAS REATIVAS (? )

Thanks for the attention UFABC 2014 61


MULTI-FLUX PHENOMENA. GENERALIZED THEORY The two sets of particles or bi-flux theory can be generalized to a multi set of particles or multi-flux diffusion phenomena. Indeed, let us assume the existence of m nested excitation states {E 1, E 2, … Em}. Let βi be the fraction of mass belonging to the excitation state Ei. Since all the particles in the system must be in some excitation state, clearly . The generalized of mass flow rate is given by: and Now, for homogeneous media the mass conservation principle leads to the generalized governing equation:

GAS DIFFUSION IN POROUS MEDIA J. M. D. Mac. Elroy, Diffusion in Homogeneous Media, in Diffusion in Polymers, Eds. P. Neogi, Marcel Dekker Inc. , 1996
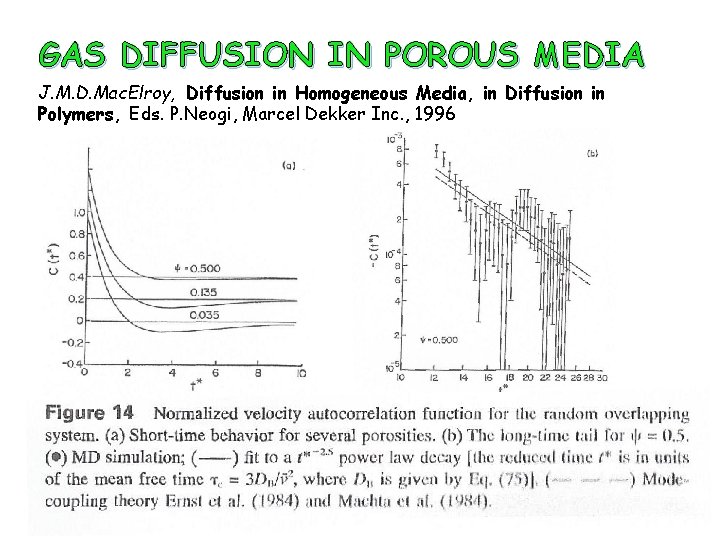
GAS DIFFUSION IN POROUS MEDIA J. M. D. Mac. Elroy, Diffusion in Homogeneous Media, in Diffusion in Polymers, Eds. P. Neogi, Marcel Dekker Inc. , 1996

GAS DIFFUSION IN POROUS MEDIA J. M. D. Mac. Elroy, Diffusion in Homogeneous Media, in Diffusion in Polymers, Eds. P. Neogi, Marcel Dekker Inc. , 1996
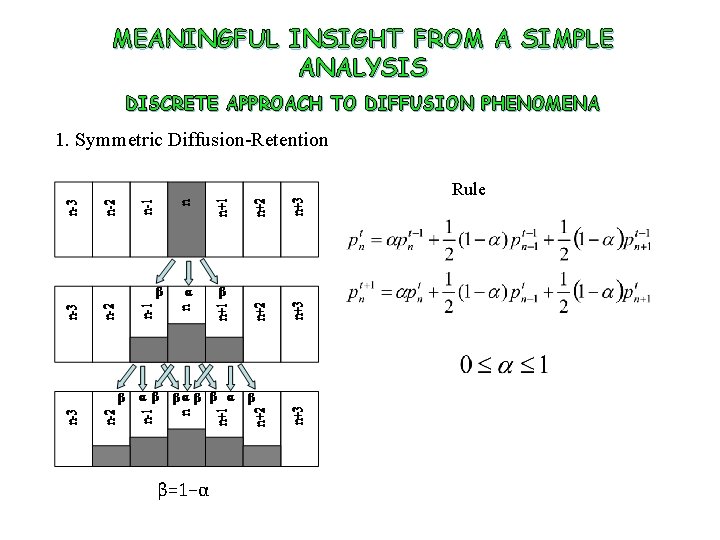
MEANINGFUL INSIGHT FROM A SIMPLE ANALYSIS DISCRETE APPROACH TO DIFFUSION PHENOMENA 1. Symmetric Diffusion-Retention Rule β=1−α

DISCRETE APPROACH TO DIFFUSION PHENOMENA Reduce the right hand side terms to time t-1 Writing the difference between these two expressions we get:

DISCRETE APPROACH TO DIFFUSION PHENOMENA After a careful separation of discrete variables Limit α→ 1 stationary regime Limit α→ 0 symmetric diffusion with no retention Recall

DISCRETE APPROACH TO DIFFUSION PHENOMENA If pn is the discrete representation of p(xn) and p(x) is sufficiently smooth it is possible to write: With β=1−α Where L 0, L 1 and T 0 are scale factors.

A SINGULAR UNIVERSE Kinetic energy is the main energy source driving the diffusion process. Then the energy states may be defined through the average linear momentum and the angular momentum composing the pair for each particle belonging to the set Si. Therefore the total kinetic energy Ei stored in the set Si is :

ENERGY STATES DEFINITIONS State S 1 where no rotational energy is present, that is the total energy is related to the linear momentum: State S 2 where the kinetic energy is generated by the two fundamental quantities, the linear momentum and the angular momentum: State S 3 where only the angular momentum exists or where the linear momentum vanishes. Note that the energy of a stationary state is not zero it is stored as rotational energy. Therefore for S 3 holds:
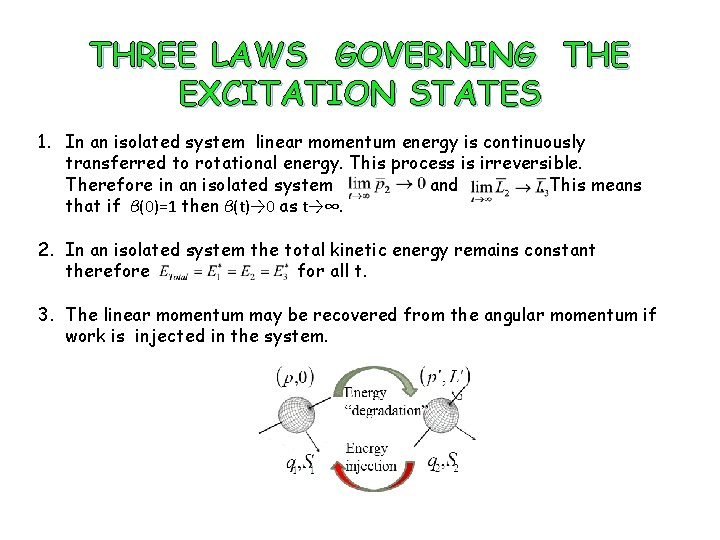
THREE LAWS GOVERNING THE EXCITATION STATES 1. In an isolated system linear momentum energy is continuously transferred to rotational energy. This process is irreversible. Therefore in an isolated system and . This means that if β(0)=1 then β(t)→ 0 as t→∞. 2. In an isolated system the total kinetic energy remains constant therefore for all t. 3. The linear momentum may be recovered from the angular momentum if work is injected in the system.
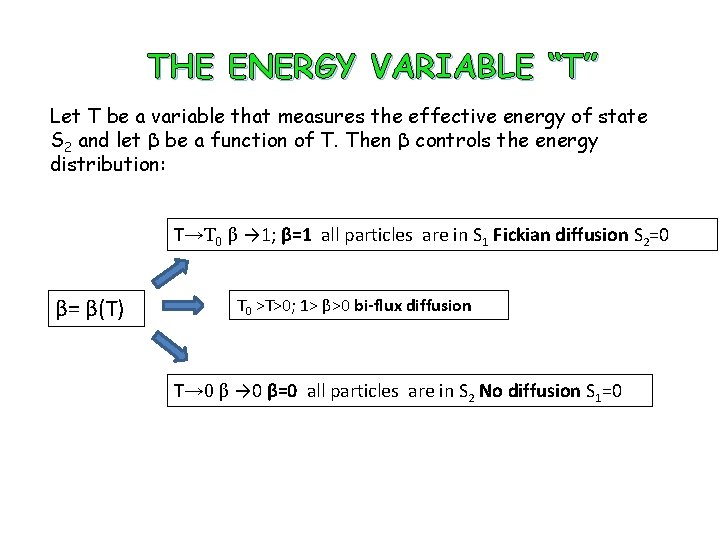
THE ENERGY VARIABLE “T” Let T be a variable that measures the effective energy of state S 2 and let β be a function of T. Then β controls the energy distribution: T→T 0 β → 1; β=1 all particles are in S 1 Fickian diffusion S 2=0 β= β(T) T 0 >T>0; 1> β>0 bi-flux diffusion T→ 0 β=0 all particles are in S 2 No diffusion S 1=0
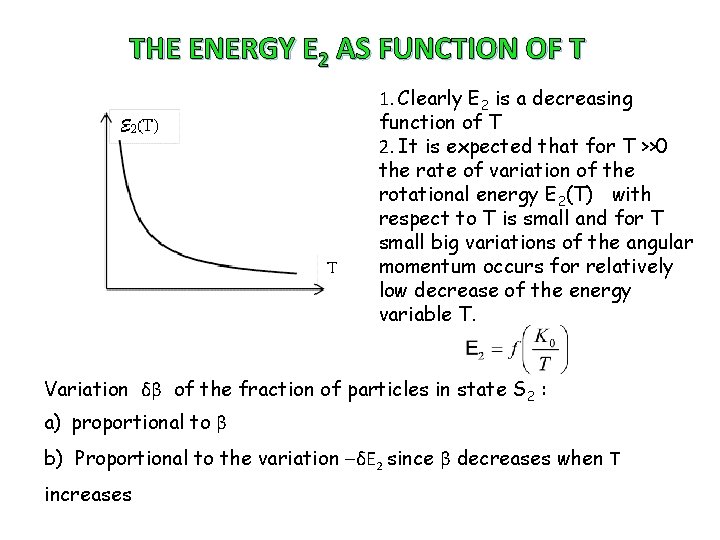
THE ENERGY E 2 AS FUNCTION OF T 1. Clearly E 2 is a decreasing function of T 2. It is expected that for T >>0 the rate of variation of the rotational energy E 2(T) with respect to T is small and for T small big variations of the angular momentum occurs for relatively low decrease of the energy variable T. Variation δβ of the fraction of particles in state S 2 : a) proportional to β b) Proportional to the variation ‒δE 2 since β decreases when T increases

THE FRACTION β AS FUNCTION OF T Variation δβ of the fraction of particles in state S 2 : a) proportional to β b) Proportional to the variation ‒δE 2 since β decreases when T increases Take Therefore f ‘ =1

ENTROPY Now define: and (S* has the dimension of entropy. ) Which suggests that β is of the form β=exp(-k. Rn)

EXAMPLE ISOLATED SYSTEM (N 1=N, T=0) N 1 → 0 as t → ∞ D=0. 1 R=0. 008 β=exp(‒t/ τ0)
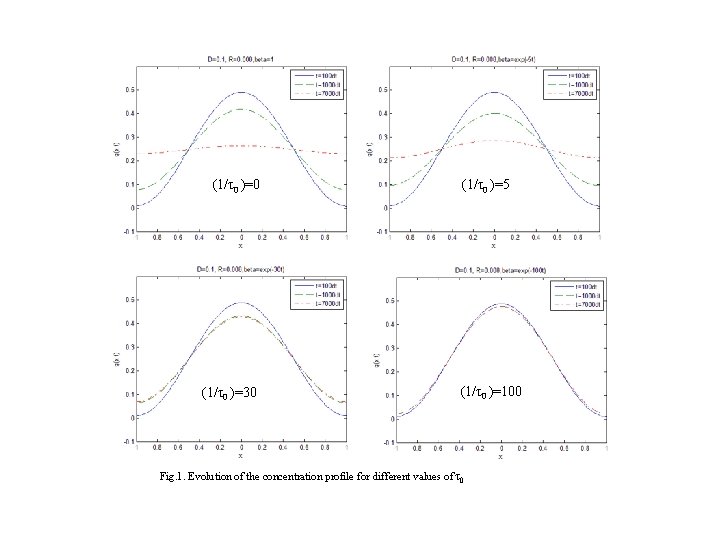
(1/τ0 )=0 (1/τ0 )=30 (1/τ0 )=5 (1/τ0 )=100 Fig. 1. Evolution of the concentration profile for different values of τ0
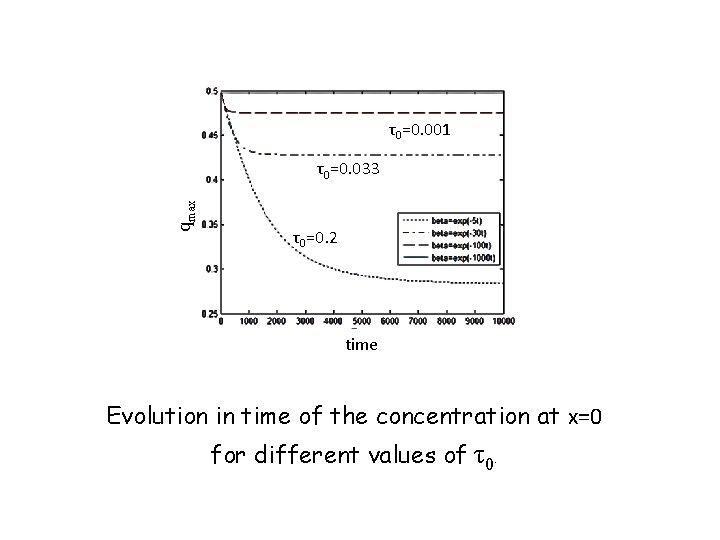
τ0=0. 001 qmax τ0=0. 033 τ0=0. 2 time Evolution in time of the concentration at x=0 for different values of τ0.
- Slides: 79