Transport of Gases in Blood Oxygen Transport Carbon

Transport of Gases in Blood Oxygen Transport Carbon Dioxide Transport 1/9/2022 Transport of Gases 1

Oxygen Transport n In order for O 2 to cross the RM – It must first dissolve in tissue fluid – Then diffuse in plasma n Henry’s law defines the volume of gas “x” that will dissolve in any liquid as – Cx = ( x * Px) where § Cx = volume of gas “x” § x = solubility coefficient § Px = partial pressure of gas 1/9/2022 Transport of Gases 2

Oxygen Transport n Amount of gas dissolved in a liquid – Increases as a function of partial pressure n Partial pressure of gas represent – The pressure it would exert in gas phase § Unit is mm Hg 1/9/2022 Transport of Gases 3

Oxygen Transport n Gas content represent – The volume of gas per unit vol of liquid that is present § Unit of gas content – Is ml gas/ml(or ml/L) of solvent 1/9/2022 Transport of Gases 4

Dissolved O 2 in Plasma n At 37 0 C and PO 2 of 95 mm. Hg – Only 0. 30 ml O 2/ 100 ml of blood n Thus total quantity of O 2 transported – Would be equal to 5000 * (0. 3/100) = 15. 0 ml O 2/min § This amount is not sufficient for the metabolic processes 1/9/2022 Transport of Gases 5
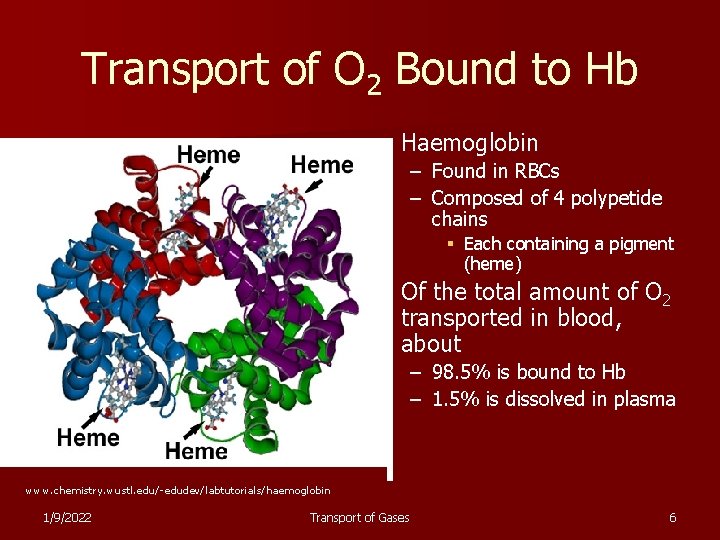
Transport of O 2 Bound to Hb n Haemoglobin – Found in RBCs – Composed of 4 polypetide chains § Each containing a pigment (heme) n Of the total amount of O 2 transported in blood, about – 98. 5% is bound to Hb – 1. 5% is dissolved in plasma www. chemistry. wustl. edu/-edudev/labtutorials/haemoglobin 1/9/2022 Transport of Gases 6

The Pigment Component n Pyrrole group Four identical protoporphyrins with central Fe++ – Fe++ is bound to 2 ionic & 2 dative bonds – Valence 1, 2, 3, 4 § bond to N atom of pyrolle group Pyrrole group 1/9/2022 Pyrrole group Transport of Gases – 5 linkages to globin – 6 valence available for combination § O 2 or CO 7
Haemoglobin Heme Fe 1/9/2022 Transport of Gases 11
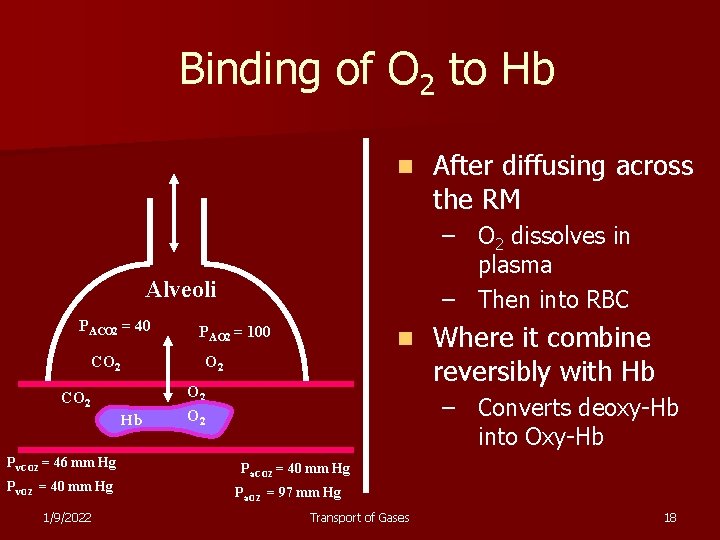
Binding of O 2 to Hb n – O 2 dissolves in plasma – Then into RBC Alveoli PACO 2 = 40 PAO 2 = 100 CO 2 Hb Pv. CO 2 = 46 mm Hg Pv. O 2 = 40 mm Hg 1/9/2022 n O 2 CO 2 After diffusing across the RM O 2 Where it combine reversibly with Hb – Converts deoxy-Hb into Oxy-Hb Pa. CO 2 = 40 mm Hg Pa. O 2 = 97 mm Hg Transport of Gases 18
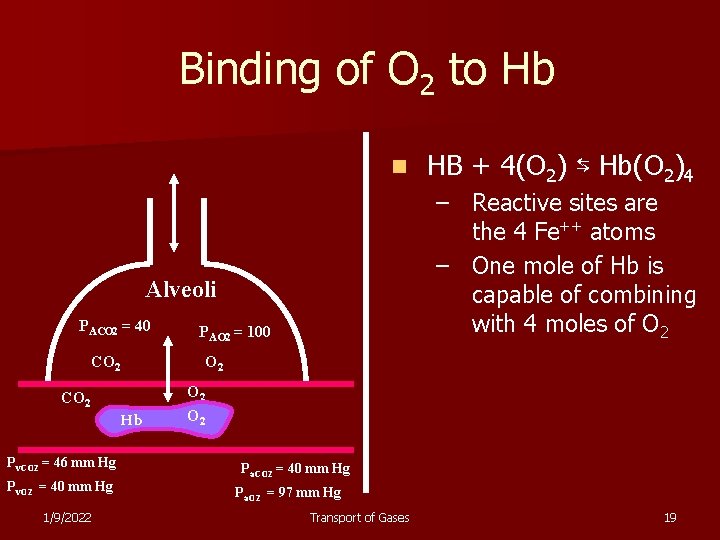
Binding of O 2 to Hb n – Reactive sites are the 4 Fe++ atoms – One mole of Hb is capable of combining with 4 moles of O 2 Alveoli PACO 2 = 40 PAO 2 = 100 CO 2 Hb Pv. CO 2 = 46 mm Hg Pv. O 2 = 40 mm Hg 1/9/2022 HB + 4(O 2) ⇆ Hb(O 2)4 O 2 Pa. CO 2 = 40 mm Hg Pa. O 2 = 97 mm Hg Transport of Gases 19

Binding of O 2 to Hb n Under ideal conditions – 1 gm of Hb § Combine with 1. 39 ml O 2 n In the normal human body – 1 gm of Hb combine with 1. 34 to 1. 36 ml of O 2 (Huffner’s number) § Small fraction of Hb is in an inactive form 1/9/2022 Transport of Gases 20

Binding of O 2 to Hb n When the Fe++ is oxidized to Fe+++ – The haemoglobin is known as § Methaemoglobin § Its ability to combine with O 2 is lost n Carbon monoxide – Combine with Fe++ – Displaces the O 2 from it 1/9/2022 Transport of Gases 21

Binding of O 2 to Hb n Cyanide combine and inactivate Hb – It combines with Fe+++ of the methaemoglobin – Hb (Fe++) ⇆ Hb (Fe+++) + CN Cyano – methaemoglobin § Normally methaemoglobin is <1% § Cyano-methaemoglobin – Is stable – Reaction is one way 1/9/2022 Transport of Gases 22

Binding of O 2 to Hb n Other respiratory pigments include – Myoglobin § Has one Fe++ atom § Functions as for 1 atom of Fe++ in Hb – Cytochrome § Change from O 2 poor to O 2 rich – Associated with oxidation of Fe++ to Fe+++ 1/9/2022 Transport of Gases 23

Oxygen Capacity of Blood n Maximum by the Hb amount of O 2 that can be bound – 1 gm of Hb combine with 1. 34 ml O 2 – Amount of Hb available = 15 gm Hb/100 ml of blood § 15*1. 34 = 20. 1 ml O 2 / 100 ml blood § This is the O 2 carrying capacity of blood 1/9/2022 Transport of Gases 24

Oxygen Flux n Total amount of O 2 transported by blood per minute – O 2 flux = C. O*(arterial O 2 content) = 5000*(20/100) = 1000 ml O 2 /min n Under normal condition – 250 ml of O 2 is utilized (25% of O 2 flux) § Thus circulating blood releases 25% of its Oxygen § Venous blood is approx 75% saturated with O 2 § The 75% of the un-extracted O 2 forms “reserve” 1/9/2022 Transport of Gases 25

Oxygen Flux n Determined by – Cardiac output – Hb concentration – Saturation with O 2 1/9/2022 Transport of Gases 26
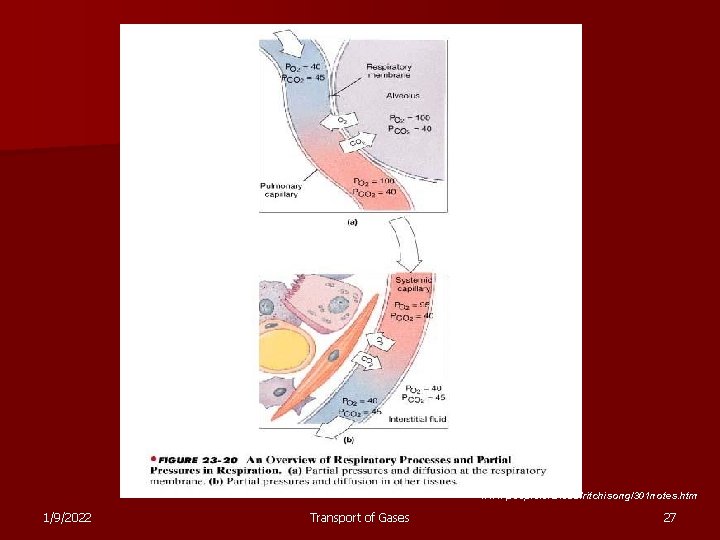
www. people. eku. edu/ritchisong/301 notes. htm 1/9/2022 Transport of Gases 27

O 2 -Hb Dissociation (Association) Curve n The property of Hb – Ability to change from deoxy-Hb to Oxy-Hb at extremely rapid rate at PO 2 in lungs – Hb + O 2 ⇌ Hb(O 2) n The ability to give up O 2 in tissue – Hb(O 2) ⇌ Hb + O 2 – This ability varies & depends on § Metabolic activities & requirements 1/9/2022 Transport of Gases 28

O 2 -Hb Dissociation (Association) Curve n The effective release of O 2 at tissue capillaries occurs because – Amount of O 2 combined to Hb varies with § Po 2 § Acidity § Temperature 1/9/2022 Transport of Gases 29
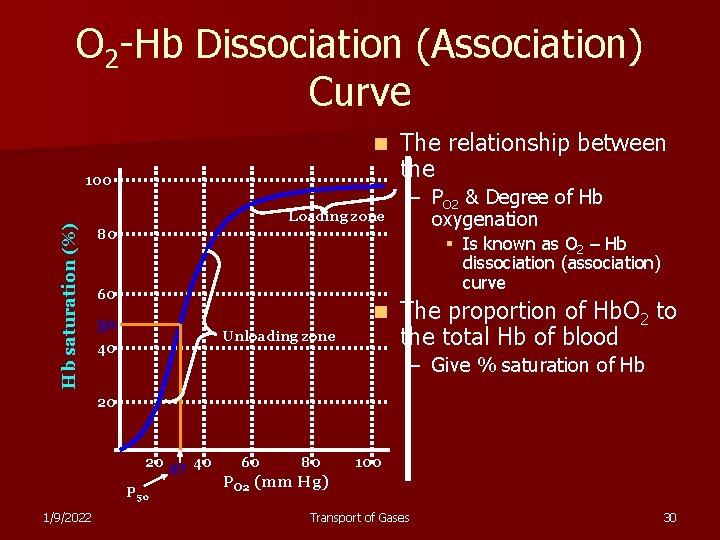
O 2 -Hb Dissociation (Association) Curve n Hb saturation (%) 100 Loading zone 80 The relationship between the – PO 2 & Degree of Hb oxygenation § Is known as O 2 – Hb dissociation (association) curve 60 n 50 Unloading zone 40 The proportion of Hb. O 2 to the total Hb of blood – Give % saturation of Hb 20 20 27 40 P 50 1/9/2022 60 80 100 PO 2 (mm Hg) Transport of Gases 30
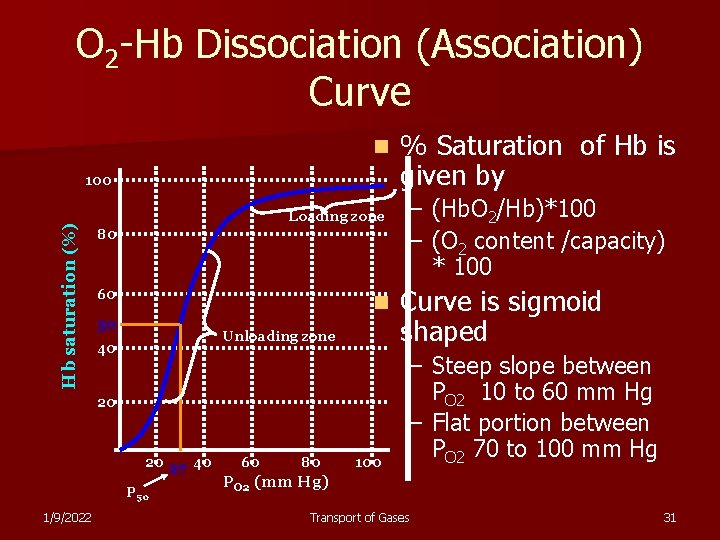
O 2 -Hb Dissociation (Association) Curve n Hb saturation (%) 100 Loading zone 80 60 n 50 Unloading zone 40 20 20 27 40 P 50 1/9/2022 60 80 100 % Saturation of Hb is given by – (Hb. O 2/Hb)*100 – (O 2 content /capacity) * 100 Curve is sigmoid shaped – Steep slope between PO 2 10 to 60 mm Hg – Flat portion between PO 2 70 to 100 mm Hg PO 2 (mm Hg) Transport of Gases 31
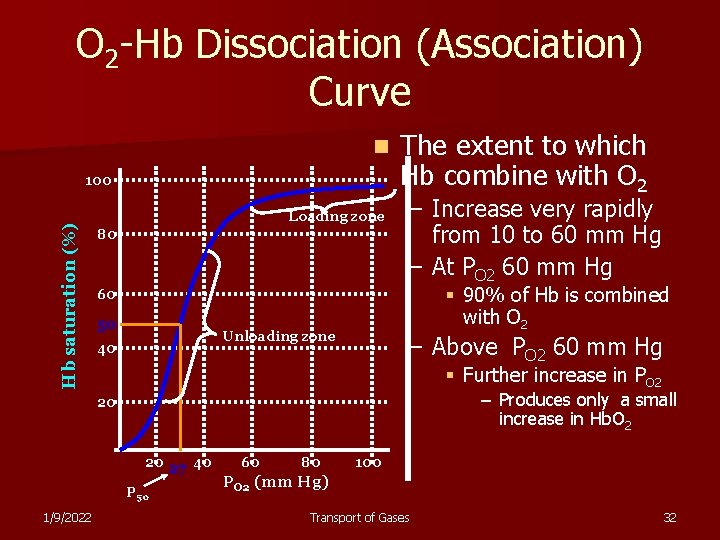
O 2 -Hb Dissociation (Association) Curve n Hb saturation (%) 100 Loading zone 80 The extent to which Hb combine with O 2 – Increase very rapidly from 10 to 60 mm Hg – At PO 2 60 mm Hg § 90% of Hb is combined with O 2 60 50 Unloading zone 40 – Above PO 2 60 mm Hg § Further increase in PO 2 – Produces only a small increase in Hb. O 2 20 20 27 40 P 50 1/9/2022 60 80 100 PO 2 (mm Hg) Transport of Gases 32
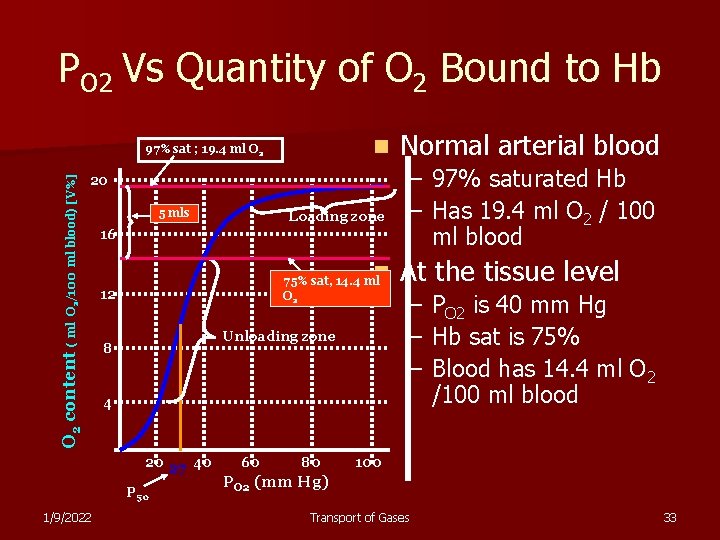
PO 2 Vs Quantity of O 2 Bound to Hb n 20 O 2 content ( ml O 2/100 ml blood) [V%] 97% sat ; 19. 4 ml O 2 5 mls 16 n 75% sat, 14. 4 ml O 2 12 Unloading zone 8 4 20 27 40 P 50 1/9/2022 Loading zone 60 80 Normal arterial blood – 97% saturated Hb – Has 19. 4 ml O 2 / 100 ml blood At the tissue level – PO 2 is 40 mm Hg – Hb sat is 75% – Blood has 14. 4 ml O 2 /100 ml blood 100 PO 2 (mm Hg) Transport of Gases 33
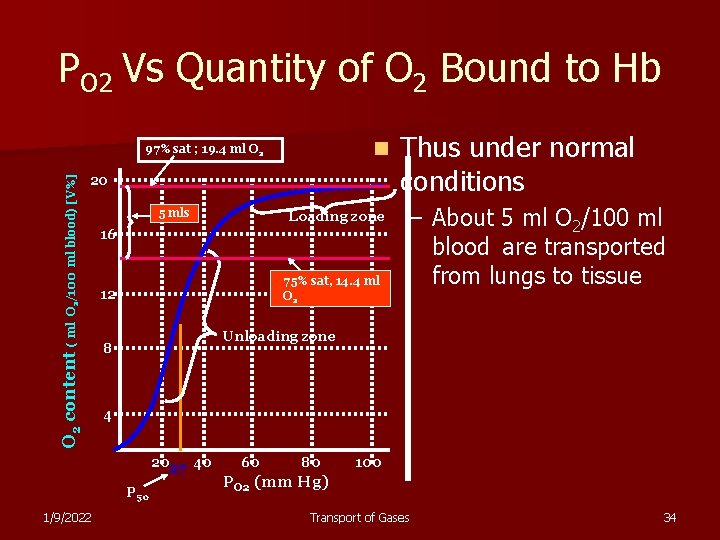
PO 2 Vs Quantity of O 2 Bound to Hb n 20 O 2 content ( ml O 2/100 ml blood) [V%] 97% sat ; 19. 4 ml O 2 5 mls 16 75% sat, 14. 4 ml O 2 12 – About 5 ml O 2/100 ml blood are transported from lungs to tissue Unloading zone 8 4 2027 40 P 50 1/9/2022 Loading zone Thus under normal conditions 60 80 100 PO 2 (mm Hg) Transport of Gases 34
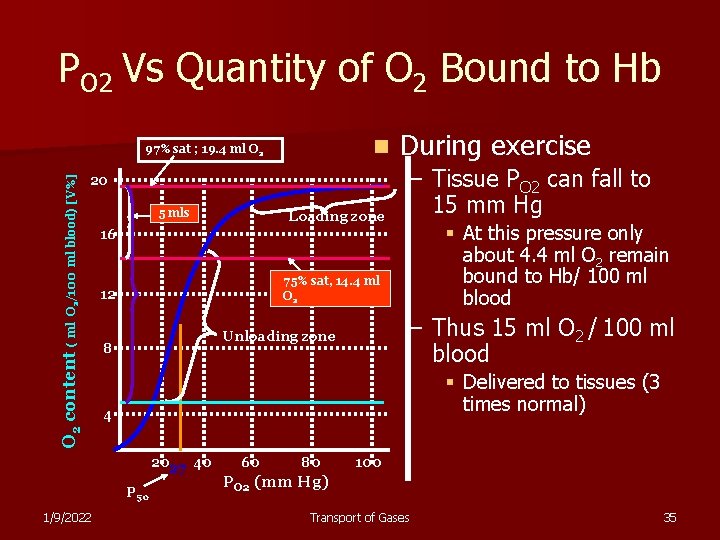
PO 2 Vs Quantity of O 2 Bound to Hb n 20 O 2 content ( ml O 2/100 ml blood) [V%] 97% sat ; 19. 4 ml O 2 5 mls – Tissue PO 2 can fall to 15 mm Hg § At this pressure only about 4. 4 ml O 2 remain bound to Hb/ 100 ml blood 16 75% sat, 14. 4 ml O 2 12 – Thus 15 ml O 2 / 100 ml blood Unloading zone 8 § Delivered to tissues (3 times normal) 4 2027 40 P 50 1/9/2022 Loading zone During exercise 60 80 100 PO 2 (mm Hg) Transport of Gases 35
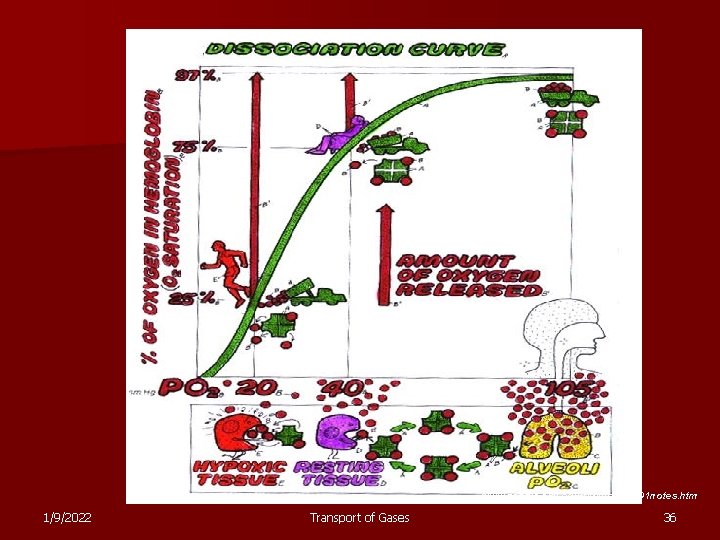
www. people. eku. edu/ritchisong/301 notes. htm 1/9/2022 Transport of Gases 36
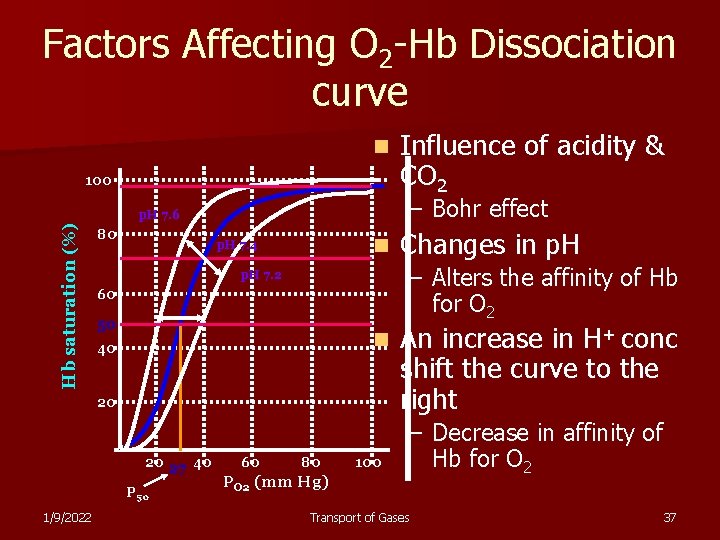
Factors Affecting O 2 -Hb Dissociation curve n 100 – Bohr effect Hb saturation (%) p. H 7. 6 80 n p. H 7. 4 60 50 n 40 20 P 50 1/9/2022 60 Changes in p. H – Alters the affinity of Hb for O 2 p. H 7. 2 20 27 40 Influence of acidity & CO 2 80 PO 2 (mm Hg) 100 An increase in H+ conc shift the curve to the right – Decrease in affinity of Hb for O 2 Transport of Gases 37
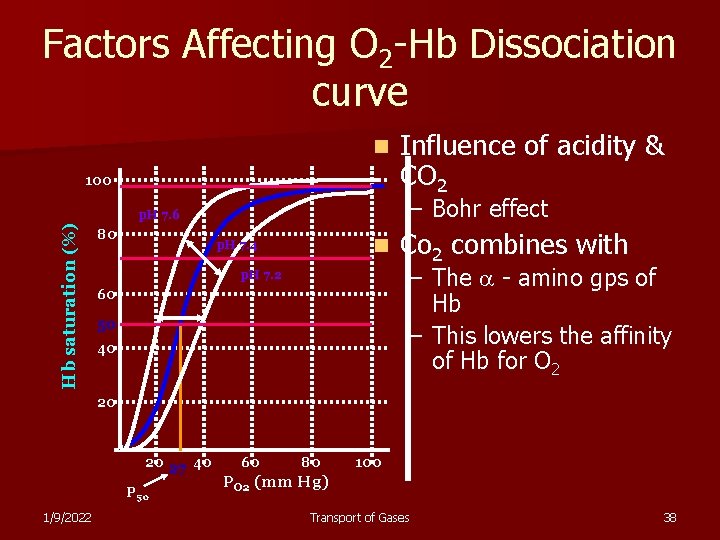
Factors Affecting O 2 -Hb Dissociation curve n 100 – Bohr effect Hb saturation (%) p. H 7. 6 80 Influence of acidity & CO 2 n p. H 7. 4 Co 2 combines with – The - amino gps of Hb – This lowers the affinity of Hb for O 2 p. H 7. 2 60 50 40 20 20 27 40 P 50 1/9/2022 60 80 100 PO 2 (mm Hg) Transport of Gases 38
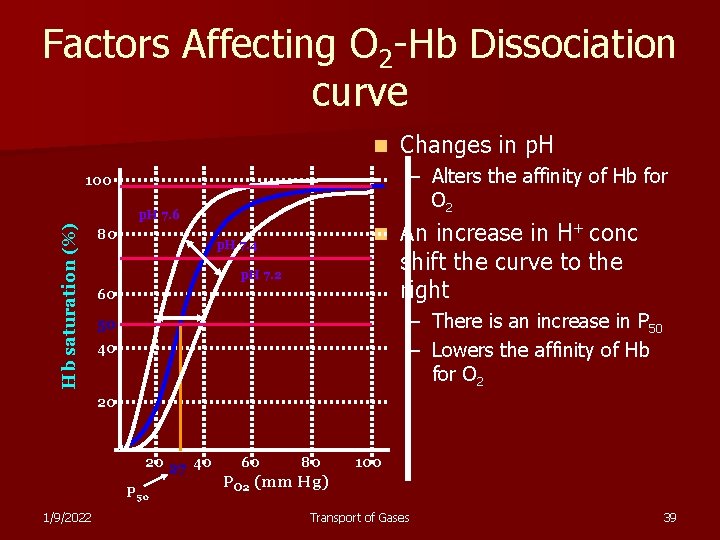
Factors Affecting O 2 -Hb Dissociation curve n – Alters the affinity of Hb for O 2 100 Hb saturation (%) p. H 7. 6 80 Changes in p. H 7. 4 p. H 7. 2 60 An increase in H+ conc shift the curve to the right – There is an increase in P 50 – Lowers the affinity of Hb for O 2 50 40 20 20 27 40 P 50 1/9/2022 60 80 100 PO 2 (mm Hg) Transport of Gases 39
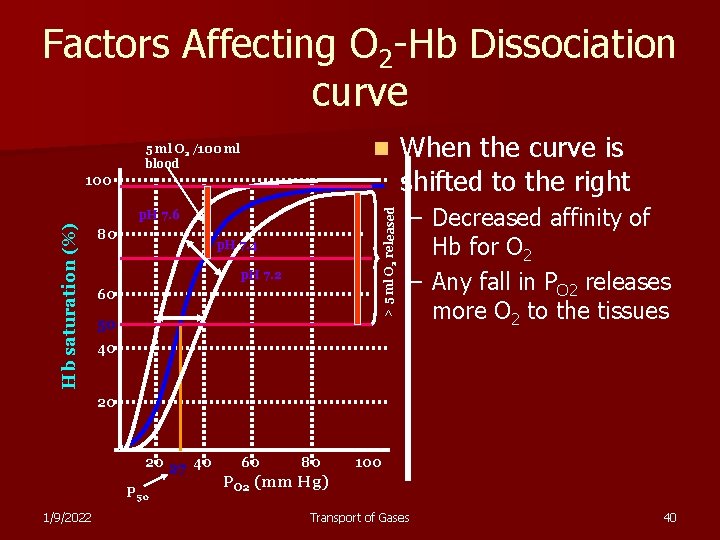
Factors Affecting O 2 -Hb Dissociation curve n 5 ml O 2 /100 ml blood > 5 ml O 2 released 100 Hb saturation (%) p. H 7. 6 80 p. H 7. 4 p. H 7. 2 60 50 When the curve is shifted to the right – Decreased affinity of Hb for O 2 – Any fall in PO 2 releases more O 2 to the tissues 40 20 20 27 40 P 50 1/9/2022 60 80 100 PO 2 (mm Hg) Transport of Gases 40
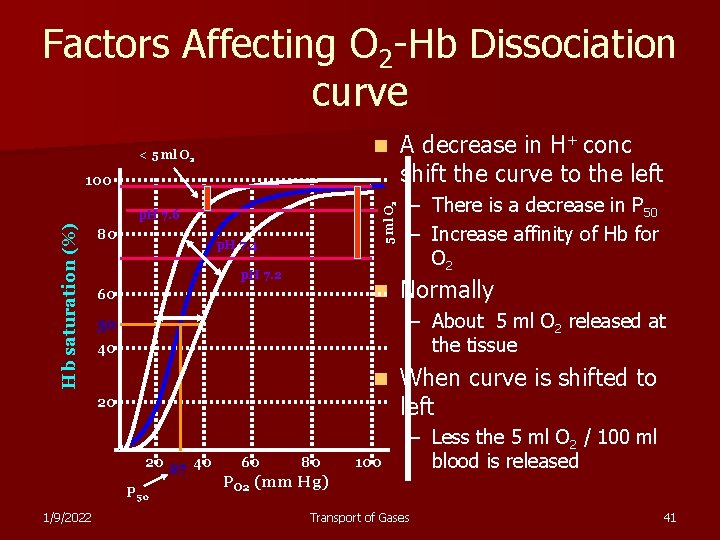
Factors Affecting O 2 -Hb Dissociation curve n < 5 ml O 2 100 Hb saturation (%) p. H 7. 6 80 p. H 7. 4 p. H 7. 2 n 60 – There is a decrease in P 50 – Increase affinity of Hb for O 2 Normally – About 5 ml O 2 released at the tissue 50 40 n 20 20 27 40 P 50 1/9/2022 A decrease in H+ conc shift the curve to the left 60 80 PO 2 (mm Hg) 100 When curve is shifted to left – Less the 5 ml O 2 / 100 ml blood is released Transport of Gases 41
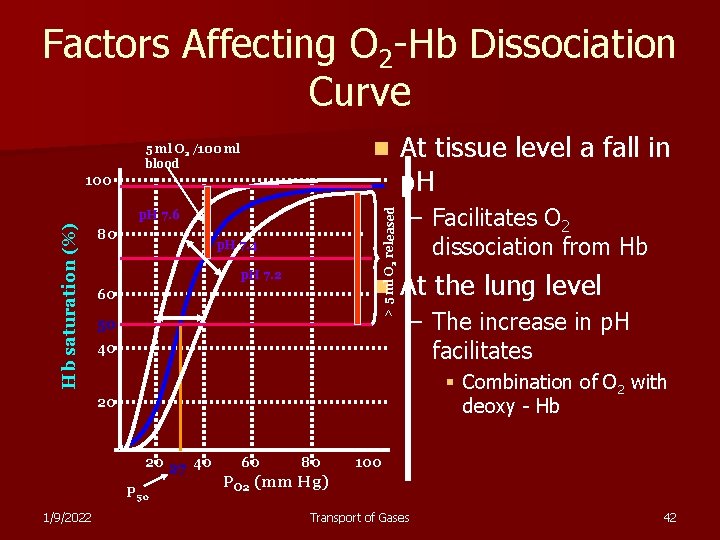
Factors Affecting O 2 -Hb Dissociation Curve n 5 ml O 2 /100 ml blood > 5 ml O 2 released 100 Hb saturation (%) p. H 7. 6 80 p. H 7. 4 p. H 7. 2 n 60 50 40 At tissue level a fall in p. H – Facilitates O 2 dissociation from Hb At the lung level – The increase in p. H facilitates § Combination of O 2 with deoxy - Hb 20 20 27 40 P 50 1/9/2022 60 80 100 PO 2 (mm Hg) Transport of Gases 42
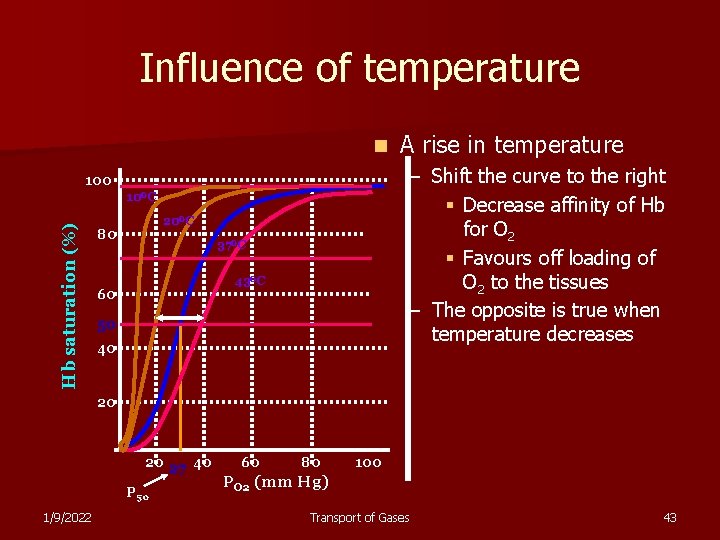
Influence of temperature n – Shift the curve to the right § Decrease affinity of Hb for O 2 § Favours off loading of O 2 to the tissues – The opposite is true when temperature decreases 100 Hb saturation (%) 10 OC 20 OC 80 37 OC 43 OC 60 A rise in temperature 50 40 20 20 27 40 P 50 1/9/2022 60 80 100 PO 2 (mm Hg) Transport of Gases 43
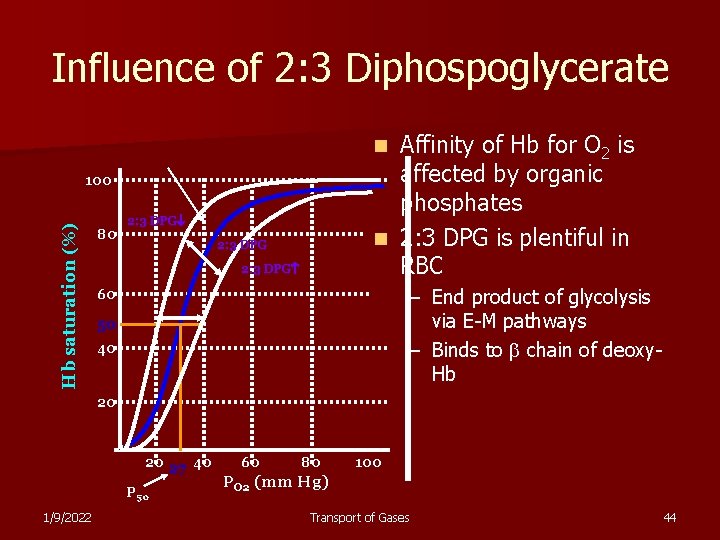
Influence of 2: 3 Diphospoglycerate Affinity of Hb for O 2 is affected by organic phosphates n 2: 3 DPG is plentiful in RBC n Hb saturation (%) 100 80 2: 3 DPG – End product of glycolysis via E-M pathways – Binds to chain of deoxy. Hb 60 50 40 20 20 27 40 P 50 1/9/2022 60 80 100 PO 2 (mm Hg) Transport of Gases 44
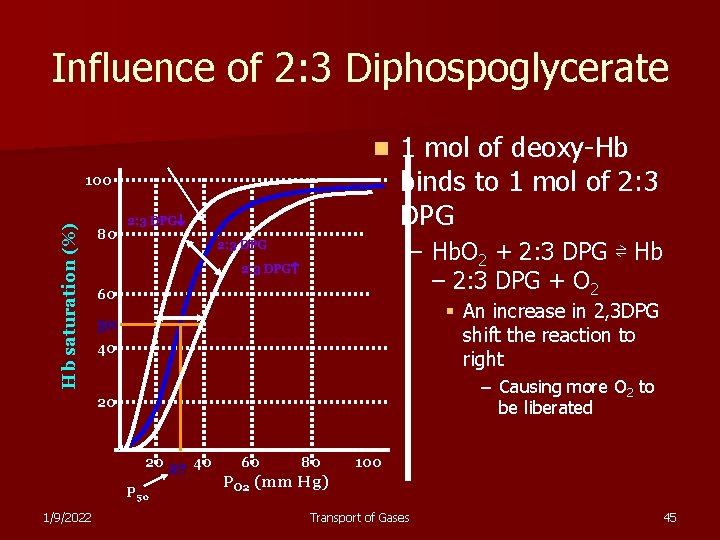
Influence of 2: 3 Diphospoglycerate n Hb saturation (%) 100 80 2: 3 DPG 1 mol of deoxy-Hb binds to 1 mol of 2: 3 DPG – Hb. O 2 + 2: 3 DPG ⇌ Hb – 2: 3 DPG + O 2 2: 3 DPG 60 § An increase in 2, 3 DPG shift the reaction to right 50 40 – Causing more O 2 to be liberated 20 20 27 40 P 50 1/9/2022 60 80 100 PO 2 (mm Hg) Transport of Gases 45
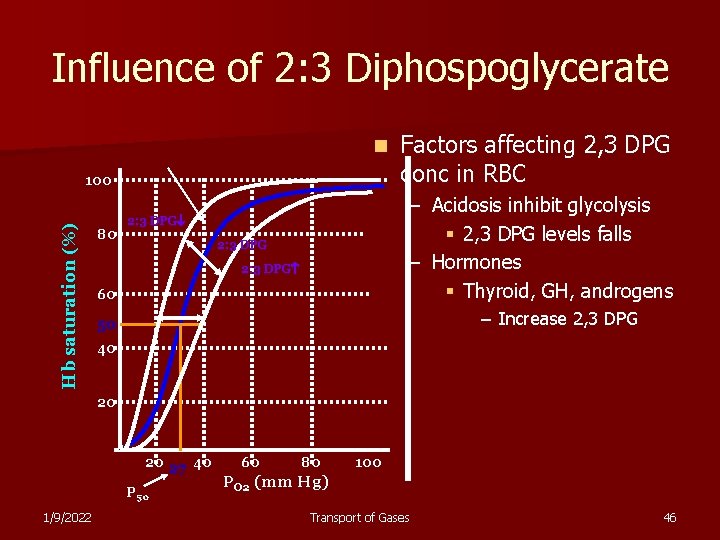
Influence of 2: 3 Diphospoglycerate n Hb saturation (%) 100 80 Factors affecting 2, 3 DPG conc in RBC – Acidosis inhibit glycolysis § 2, 3 DPG levels falls – Hormones § Thyroid, GH, androgens 2: 3 DPG 60 – Increase 2, 3 DPG 50 40 20 20 27 40 P 50 1/9/2022 60 80 100 PO 2 (mm Hg) Transport of Gases 46
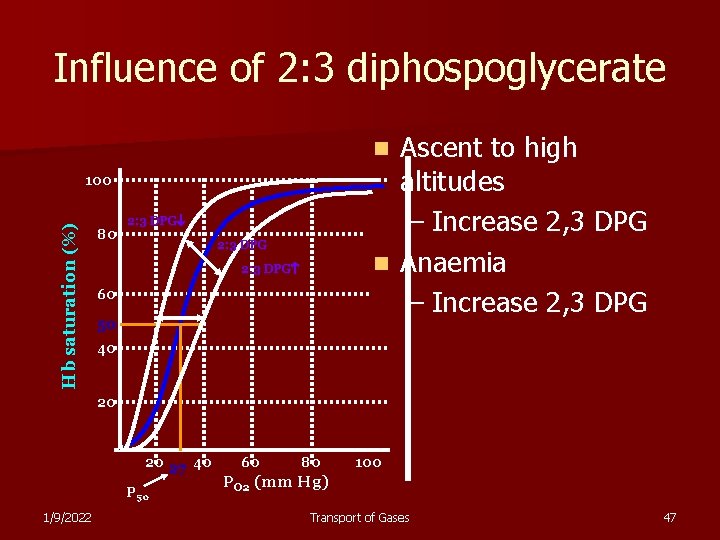
Influence of 2: 3 diphospoglycerate Ascent to high altitudes – Increase 2, 3 DPG n Anaemia – Increase 2, 3 DPG n Hb saturation (%) 100 80 2: 3 DPG 60 50 40 20 20 27 40 P 50 1/9/2022 60 80 100 PO 2 (mm Hg) Transport of Gases 47

Transport of CO 2 1/9/2022 Transport of Gases 48

CO 2 Transport n CO 2 is produce in the cells as end product of oxidative metabolism n Diffuses from the cells into – Interstitial fluid – Plasma where it § Raises the PCO 2 of capillary and venous blood 1/9/2022 Transport of Gases 49

CO 2 Transport n CO 2 is transported in three forms – Physically dissolved in plasma (10%) – As bicarbonate ions in plasma (60%) – As carbamino compounds (30%) 1/9/2022 Transport of Gases 50

CO 2 Transport in Plasma n Physically dissolved depend on the – Partial pressure – Solubility coefficient n Hydration of CO 2 – CO 2+ H 2 O ⇌ H 2 CO 3 ⇌ H+ + HCO 3– In plasma there is no carbonic anhydrase § Equilibrium is far to the left § Accumulation of H 2 CO 3 stops the reaction § H+ are buffered by plasma buffers 1/9/2022 Transport of Gases 51

CO 2 Transport in Plasma n Formation of carbamino compounds – Amino groups have ability to combine with CO 2 § To form carbamino compounds – R-N(H 2) + CO 2 ⇌ R-NH-COO- + H+ § This reaction restricted to terminal AA or § Side chains AA (lysine, arginine) 1/9/2022 Transport of Gases 52

CO 2 Transport by RBC n Small amount of CO 2 dissolved in fluid in RBC n Hydration of CO 2 – CO 2+ H 2 O ⇌ H 2 CO 3 ⇌ H+ + HCO 3 - n In RBC reaction proceed to right because – Presence of carbonic anhydrase – H+ and HCO 3 - do not accumulate 1/9/2022 Transport of Gases 53

CO 2 Transport by RBC n H+ cannot readily move out of RBC Cl. HCO 3 CO 2 Cl- CA CO 2 + H 2 O ⇌ H 2 CO 3⇌ H+ + HCO 3 - n H++ Hb. O 2 HHb + CO 2 HHb + O 2 Carbamino-Hb O 2 1/9/2022 – It is buffered by Hb buffer – H+ + Hb. O 2 ⇌ H+. Hb + O 2 As H+ binds to Hb – Reduces Hb affinity for O 2 § Cause unloading of O 2 to tissues Transport of Gases 54

CO 2 Transport by RBC – Presence of deoxy-Hb in blood Cl. HCO 3 - § Oxygenation in pulmonary capillaries CO 2 Cl- CA – Increase Hb ability to unload CO 2 + H 2 O ⇌ H 2 CO 3⇌ H+ + HCO 3 - § This is the Haldane effect H++ Hb. O 2 HHb + CO 2 – Increase ability to carry CO 2 from the tissues HHb + O 2 Carbamino-Hb O 2 1/9/2022 Transport of Gases 55

CO 2 Transport by RBC n HCO 3 - leaves the RBC Cl. HCO 3 - – Concentration gradient – Chloride shift § Chloride/bicarbonate carrier protein CO 2 Cl- CA – Cl- move into RBC – HCO 3 - move into plasma CO 2 + H 2 O ⇌ H 2 CO 3⇌ H+ + HCO 3 H++ Hb. O 2 HHb + CO 2 HHb + O 2 Carbamino-Hb O 2 1/9/2022 Transport of Gases 56
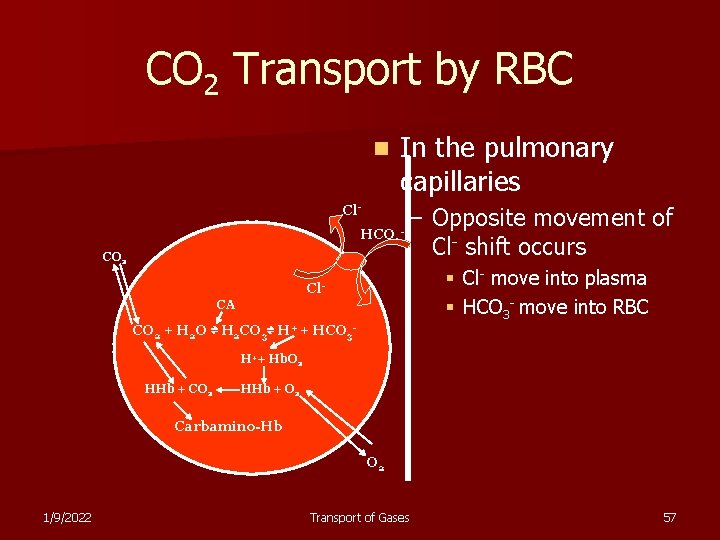
CO 2 Transport by RBC n In the pulmonary capillaries Cl. HCO 3 CO 2 – Opposite movement of Cl- shift occurs § Cl- move into plasma § HCO 3 - move into RBC Cl. CA CO 2 + H 2 O ⇌ H 2 CO 3⇌ H+ + HCO 3 H++ Hb. O 2 HHb + CO 2 HHb + O 2 Carbamino-Hb O 2 1/9/2022 Transport of Gases 57
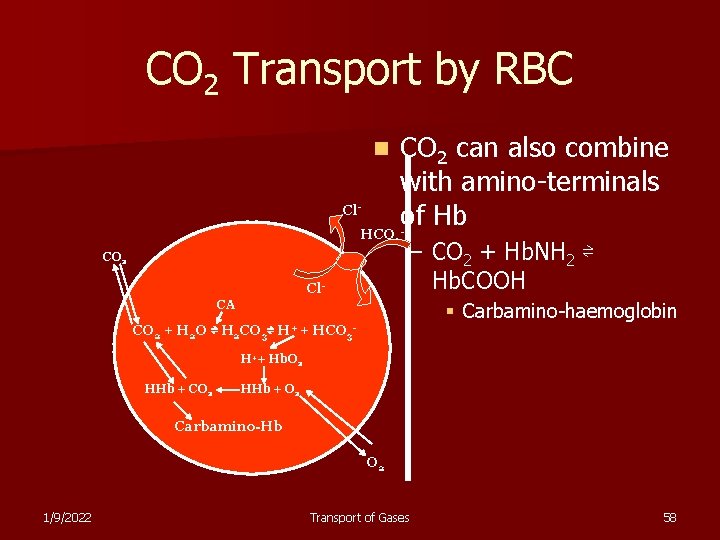
CO 2 Transport by RBC CO 2 can also combine with amino-terminals Cl of Hb HCO n - 3 CO 2 Cl- – CO 2 + Hb. NH 2 ⇌ Hb. COOH CA CO 2 + H 2 O ⇌ H 2 CO 3⇌ H+ + § Carbamino-haemoglobin HCO 3 - H++ Hb. O 2 HHb + CO 2 HHb + O 2 Carbamino-Hb O 2 1/9/2022 Transport of Gases 58

CO 2 Transport n Although the major reactions regarding transport of CO 2 – Occur in the RBCs n The bulk of CO 2 is transported in plasma – In the form of HCO 3 - 1/9/2022 Transport of Gases 59
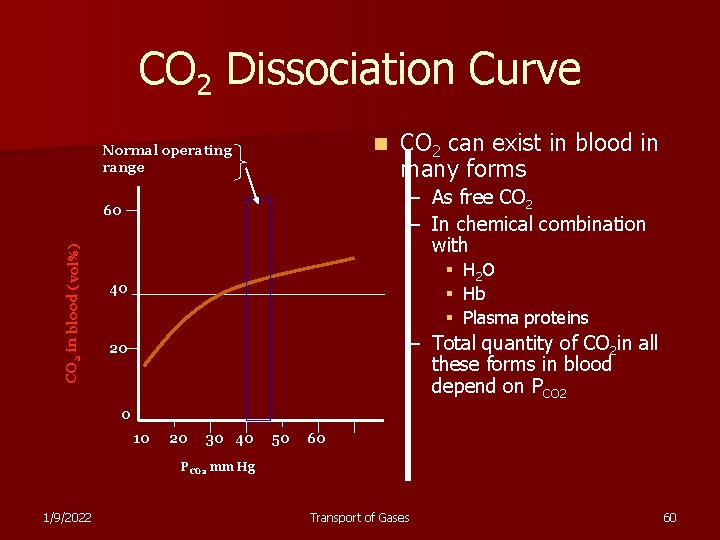
CO 2 Dissociation Curve n Normal operating range – As free CO 2 – In chemical combination with 60 CO 2 in blood (vol%) CO 2 can exist in blood in many forms § § § 40 H 2 O Hb Plasma proteins – Total quantity of CO 2 in all these forms in blood depend on PCO 2 20 0 10 20 30 40 50 60 PCO 2 mm Hg 1/9/2022 Transport of Gases 60
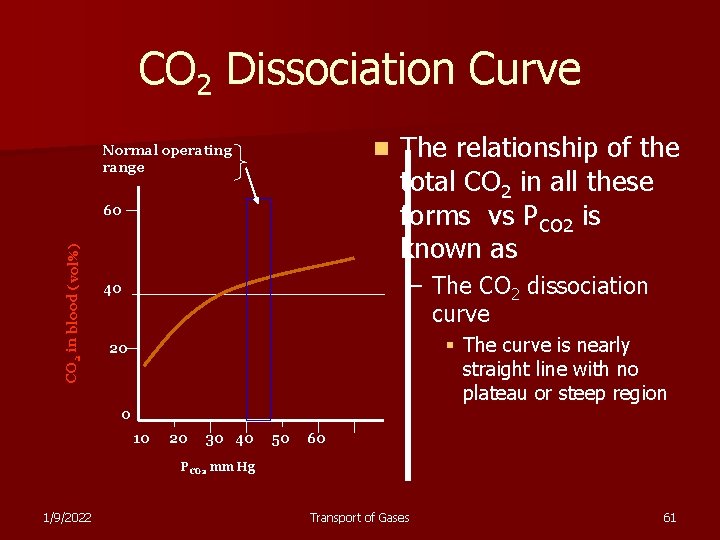
CO 2 Dissociation Curve n Normal operating range CO 2 in blood (vol%) 60 The relationship of the total CO 2 in all these forms vs PCO 2 is known as – The CO 2 dissociation curve 40 § The curve is nearly straight line with no plateau or steep region 20 0 10 20 30 40 50 60 PCO 2 mm Hg 1/9/2022 Transport of Gases 61
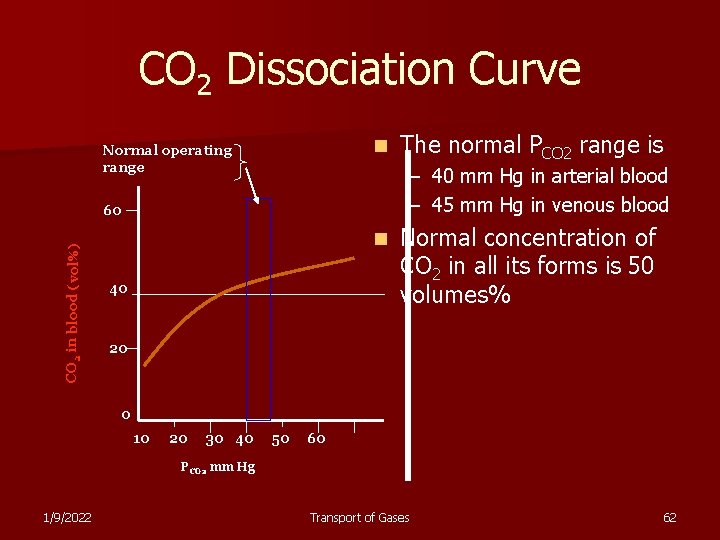
CO 2 Dissociation Curve n Normal operating range – 40 mm Hg in arterial blood – 45 mm Hg in venous blood 60 CO 2 in blood (vol%) The normal PCO 2 range is n 40 Normal concentration of CO 2 in all its forms is 50 volumes% 20 0 10 20 30 40 50 60 PCO 2 mm Hg 1/9/2022 Transport of Gases 62
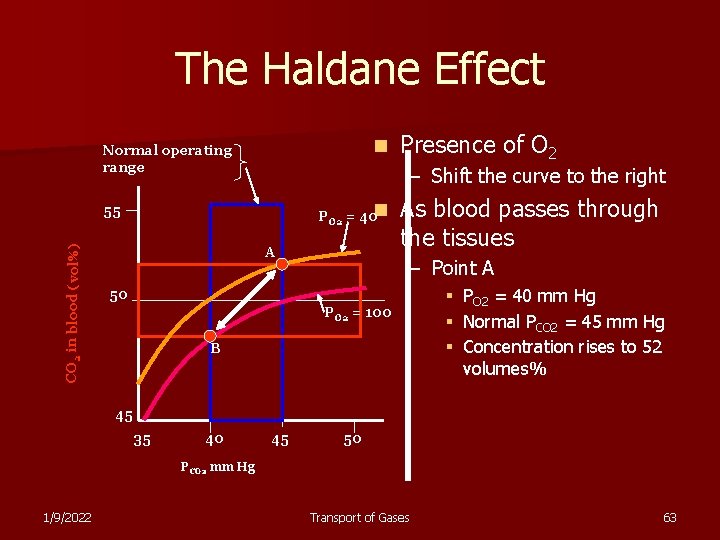
The Haldane Effect n Normal operating range – Shift the curve to the right PO 2 = 40 n 55 CO 2 in blood (vol%) Presence of O 2 A As blood passes through the tissues – Point A 50 PO 2 = 100 B § § § PO 2 = 40 mm Hg Normal PCO 2 = 45 mm Hg Concentration rises to 52 volumes% 45 35 40 45 50 PCO 2 mm Hg 1/9/2022 Transport of Gases 63
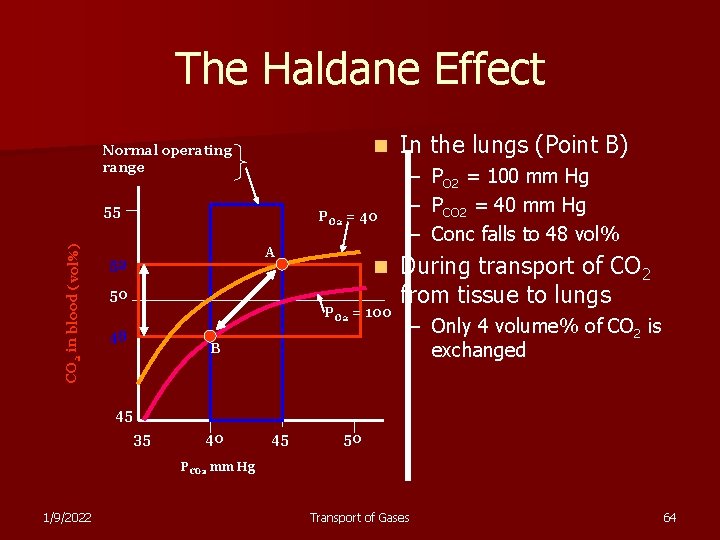
The Haldane Effect n Normal operating range CO 2 in blood (vol%) 55 PO 2 = 40 A 52 n 50 PO 2 = 100 48 B In the lungs (Point B) – – – PO 2 = 100 mm Hg PCO 2 = 40 mm Hg Conc falls to 48 vol% During transport of CO 2 from tissue to lungs – Only 4 volume% of CO 2 is exchanged 45 35 40 45 50 PCO 2 mm Hg 1/9/2022 Transport of Gases 64
- Slides: 55