Transport Across Membranes Transport across the cell membrane
Transport Across Membranes
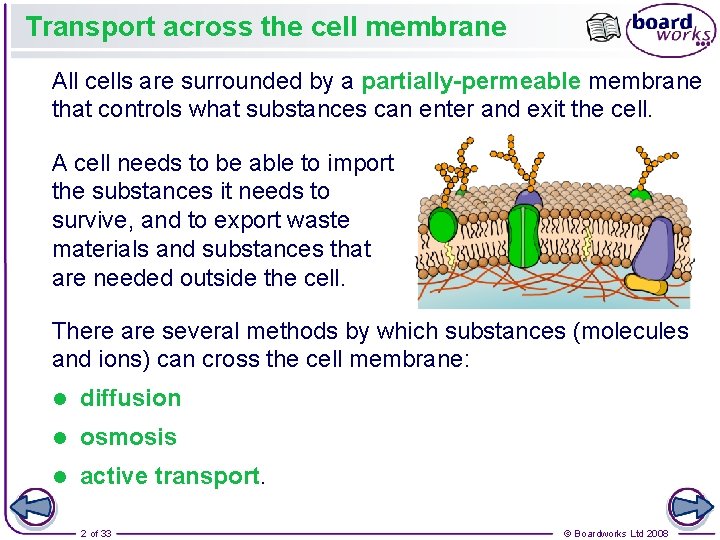
Transport across the cell membrane All cells are surrounded by a partially-permeable membrane that controls what substances can enter and exit the cell. A cell needs to be able to import the substances it needs to survive, and to export waste materials and substances that are needed outside the cell. There are several methods by which substances (molecules and ions) can cross the cell membrane: l diffusion l osmosis l active transport. 2 of 33 © Boardworks Ltd 2008
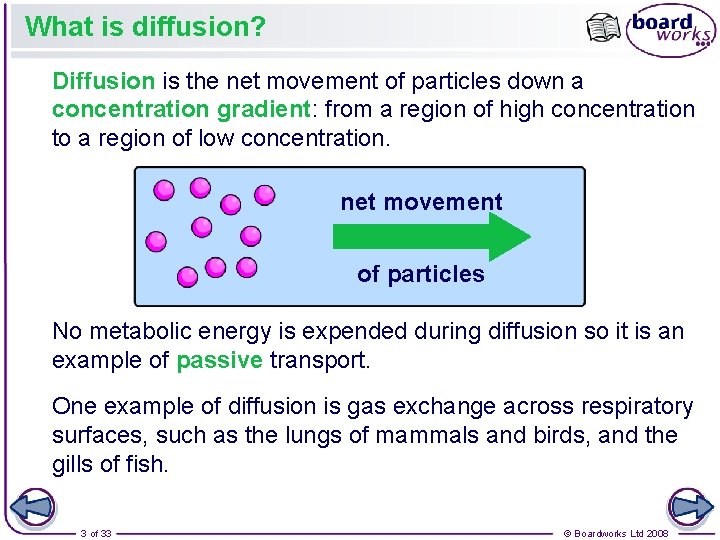
What is diffusion? Diffusion is the net movement of particles down a concentration gradient: from a region of high concentration to a region of low concentration. net movement of particles No metabolic energy is expended during diffusion so it is an example of passive transport. One example of diffusion is gas exchange across respiratory surfaces, such as the lungs of mammals and birds, and the gills of fish. 3 of 33 © Boardworks Ltd 2008

The rate of diffusion in a given direction across an exchange surface can be summarized by Fick’s law, which states that: rate of diffusion is proportional to: surface area × difference in conc. length of diffusion path (membrane thickness) Increasing the surface area across which the particles diffuse, or increasing the size of the concentration gradient will increase the rate of diffusion. Increasing the distance (or thickness of the membrane) over which diffusion takes place will decrease the rate. 4 of 33 © Boardworks Ltd 2008

What factors affect diffusion? 5 of 33 © Boardworks Ltd 2008

Cell membranes, polarity and diffusion The non-polar, hydrophobic tails of phospholipid molecules in a cell membrane act as a barrier to most substances. Generally, the smaller and less polar a molecule, the easier and faster it will diffuse across a cell membrane. l Small, non-polar molecules such as oxygen and carbon dioxide rapidly diffuse across a membrane. l Small, polar molecules, such as water and urea, also diffuse across, but much more slowly. l Charged particles (ions) cannot diffuse across a membrane, even if they are very small. 6 of 33 © Boardworks Ltd 2008

Facilitated diffusion 7 of 33 © Boardworks Ltd 2008
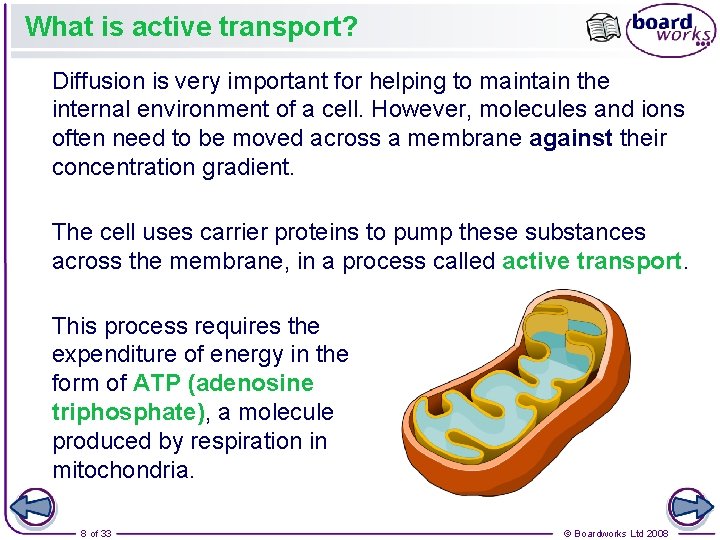
What is active transport? Diffusion is very important for helping to maintain the internal environment of a cell. However, molecules and ions often need to be moved across a membrane against their concentration gradient. The cell uses carrier proteins to pump these substances across the membrane, in a process called active transport. This process requires the expenditure of energy in the form of ATP (adenosine triphosphate), a molecule produced by respiration in mitochondria. 8 of 33 © Boardworks Ltd 2008
Examples of active transport Active transport is used in many processes, such as the: l uptake of glucose and amino acids in the small intestine l absorption of mineral ions by plant roots l excretion of hydrogen ions and urea by kidneys l exchange of sodium and potassium ions in neurons and muscle cells. Cells that undertake active transport on a large scale have many mitochondria. 9 of 33 © Boardworks Ltd 2008
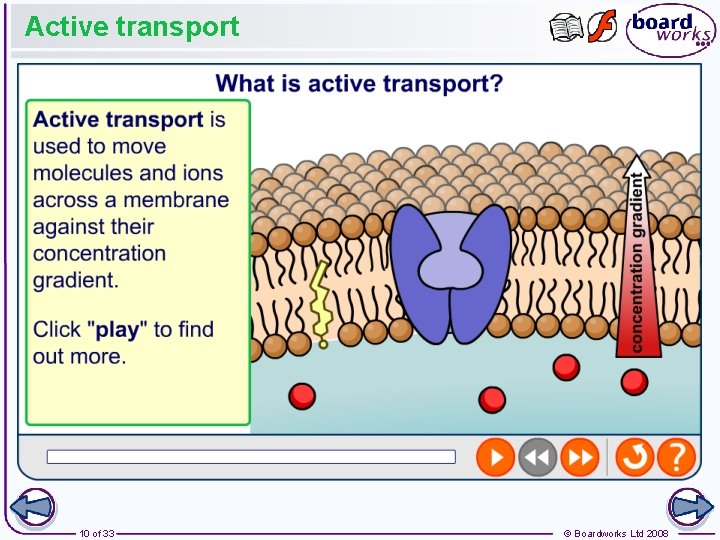
Active transport 10 of 33 © Boardworks Ltd 2008
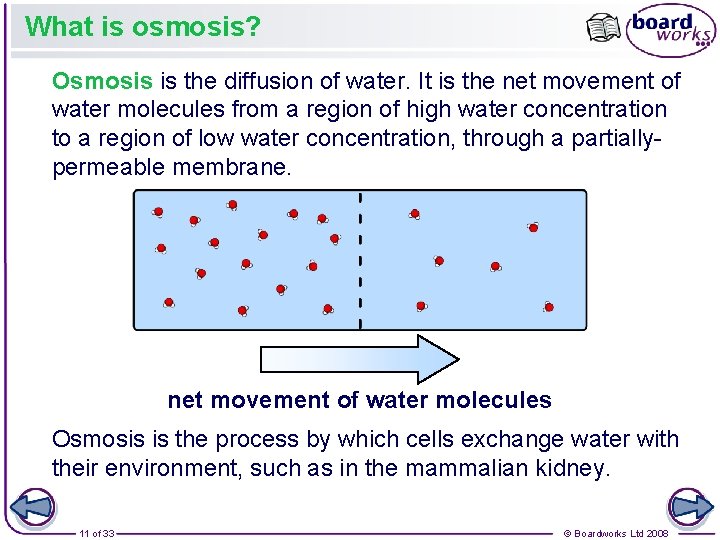
What is osmosis? Osmosis is the diffusion of water. It is the net movement of water molecules from a region of high water concentration to a region of low water concentration, through a partiallypermeable membrane. net movement of water molecules Osmosis is the process by which cells exchange water with their environment, such as in the mammalian kidney. 11 of 33 © Boardworks Ltd 2008

What is water potential? The net movement of water by osmosis is determined by differences in water potential between two solutions connected by a partially-permeable membrane. Water potential is the tendency of water molecules in a system to move. It is denoted by the symbol Ψ and is measured in kilo. Pascals (k. Pa). Pure water has the highest water potential, and has a value of 0 k. Pa. Solutions have a lower water potential than pure water, and have a negative water potential. Water molecules always move from a region of high water potential to a region of low (more negative) water potential. 12 of 33 © Boardworks Ltd 2008
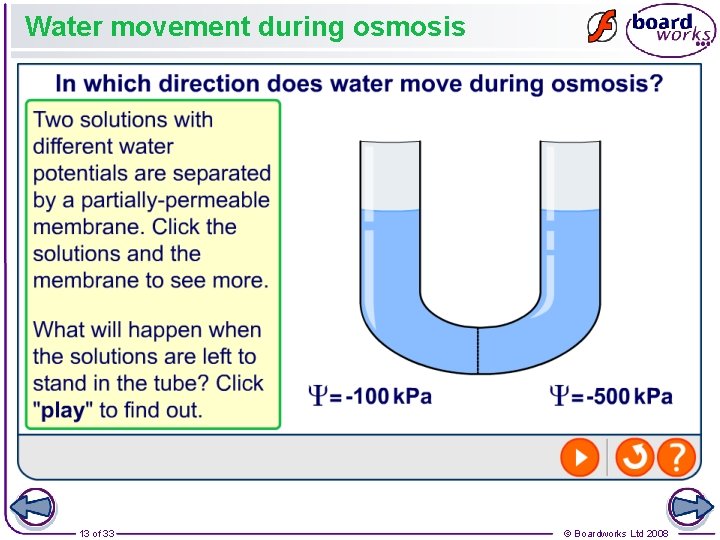
Water movement during osmosis 13 of 33 © Boardworks Ltd 2008

What is solute potential? The water potential of a solution is affected by the amount of solute it contains. The greater the amount of solute, the lower the water potential. This is because water molecules bind to the solute molecules, reducing the number of water molecules that are free to diffuse. free water molecule solute molecule The contribution that solutes make to the water potential of a solution is the solute potential (ΨS), and is a negative value. 14 of 33 © Boardworks Ltd 2008

What is pressure potential? The water potential of a solution is also affected by the pressure applied to it. The greater the pressure, the higher the water potential. This is called the pressure potential (ΨP) and is always a positive value. In plant cells, the pressure potential is a result of the cell wall exerting pressure on the cytoplasm. Water potential is calculated using the following equation: water potential = solute potential + pressure potential Ψ = ΨS + ΨP 15 of 33 © Boardworks Ltd 2008
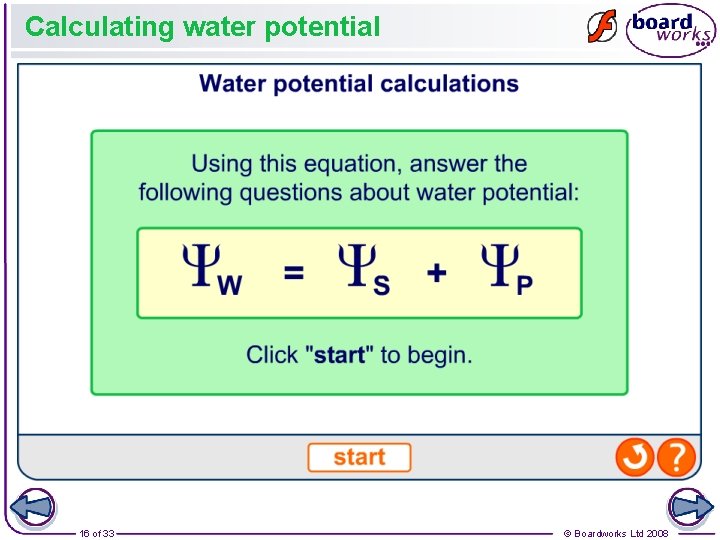
Calculating water potential 16 of 33 © Boardworks Ltd 2008
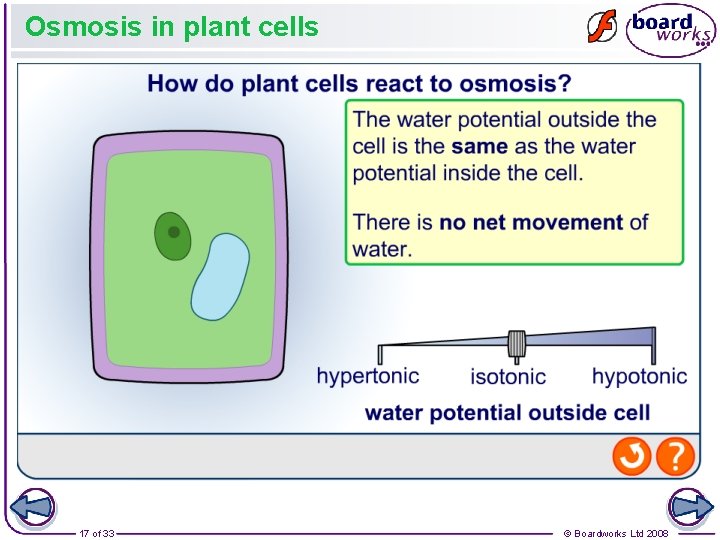
Osmosis in plant cells 17 of 33 © Boardworks Ltd 2008
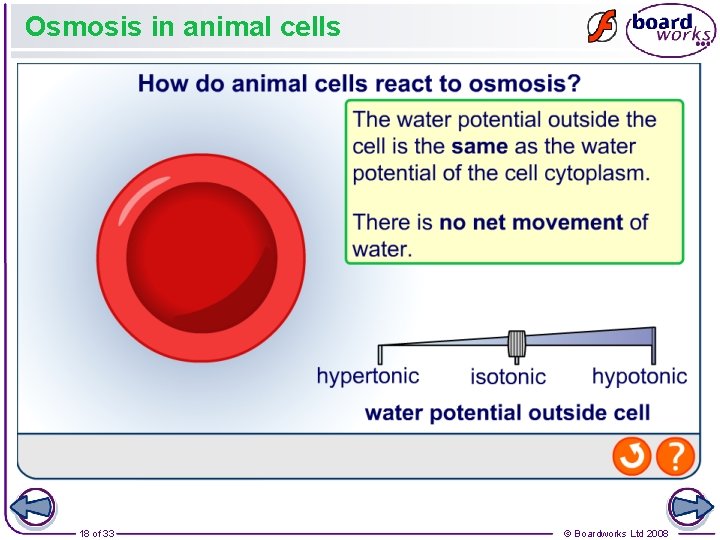
Osmosis in animal cells 18 of 33 © Boardworks Ltd 2008
Water potential of potatoes 19 of 33 © Boardworks Ltd 2008

Digestion

Digestion is when large molecules are broken down into smaller molecules. By the time the digested food reaches the small intestine it is like a soup The small molecules have been mixed with digestive juices and with sodium and chloride ions from the epithelial cells lining the first part of the small intestine
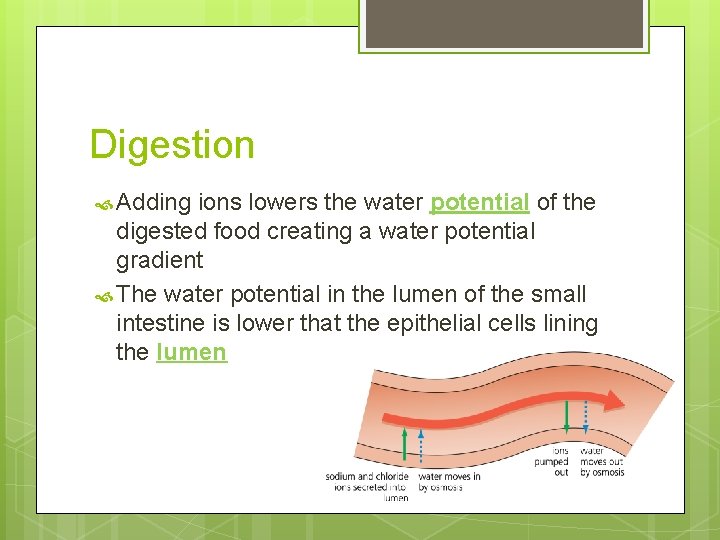
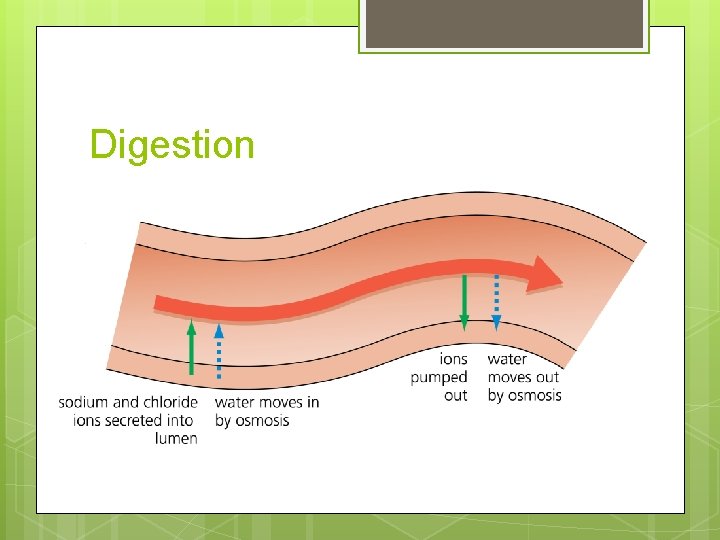
Digestion Adding ions lowers the water potential of the digested food creating a water potential gradient The water potential in the lumen of the small intestine is lower that the epithelial cells lining the lumen
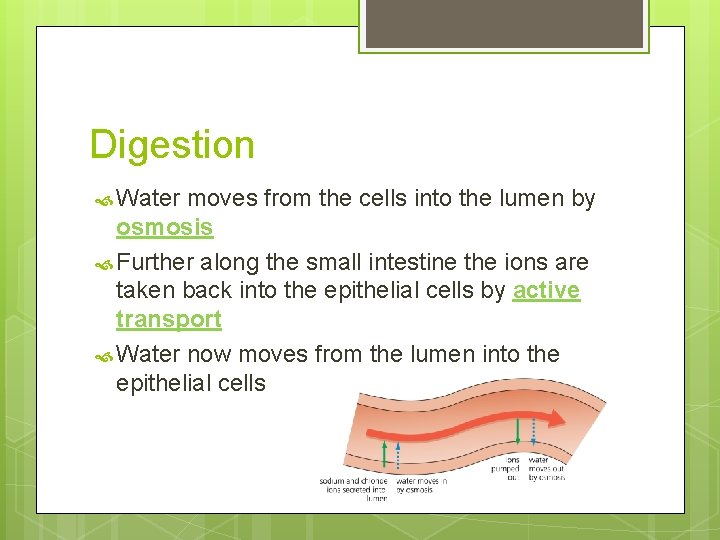
Digestion Water moves from the cells into the lumen by osmosis Further along the small intestine the ions are taken back into the epithelial cells by active transport Water now moves from the lumen into the epithelial cells
Digestion

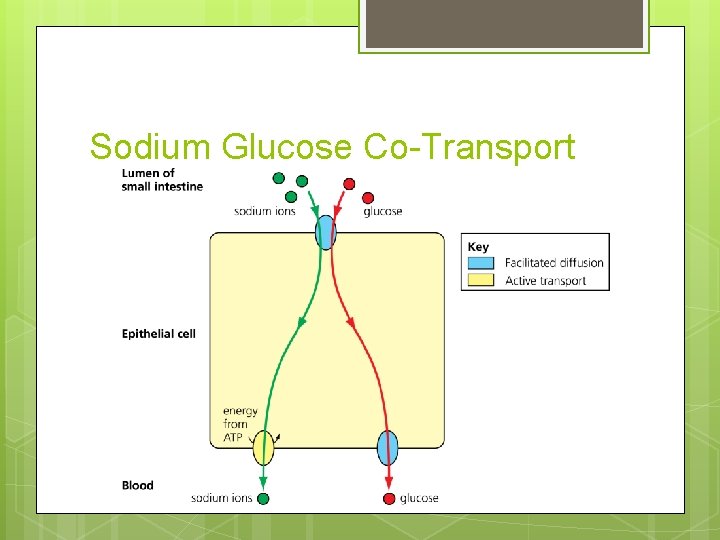
Sodium Glucose Co-Transport One of the most important products of carbohydrate digestion is glucose It is absorbed from the small intestine by facilitated diffusion and active transport It uses carrier molecules called co-transport proteins in the cell surface membrane of the epithelial cells

Sodium Glucose Co-Transport These molecules only transport glucose in the presence of sodium ions Each time a sodium ion is transported into the cell so is a glucose molecule via facilitated diffusion Since facilitated diffusion requires a concentration gradient, sodium ions must be actively transported out of the cell into the blood The glucose molecules pass from the cells into the blood by facilitated diffusion
Sodium Glucose Co-Transport

Sodium Glucose Co-Transport These molecules only transport glucose in the presence of sodium ions Each time a glucose molecule is transported into the cell so is a sodium ion via facilitated diffusion Since facilitated diffusion requires a concentration gradient, sodium ions must be actively transported out of the cell into the blood The glucose molecules pass from the cells into the blood by facilitated diffusion

Sodium Glucose Co-Transport Different factors can affect the rate of absorption The concentration of glucose Temperature


Cholera

Cholera is a disease that was once common in the UK causing 2400 deaths in Manchester in 1831. It is now very common is countries with poor sanitation leading to contaminated water It’s symptoms are muscle cramps, nausea, vomiting and severe diarrhoea A victim can lose up to 20 dm 3 of liquid a day and without treatment a person will usually die.
![Bacteria isolated as cause of cholera disease in 1854 by Fillipo Pacini [Italian Anatomist]. Bacteria isolated as cause of cholera disease in 1854 by Fillipo Pacini [Italian Anatomist].](http://slidetodoc.com/presentation_image/4e46fbc8b0dfa0952c1b3c4e541e89c2/image-33.jpg)
Bacteria isolated as cause of cholera disease in 1854 by Fillipo Pacini [Italian Anatomist]. Findings popularised by Rober Koch 30 years later
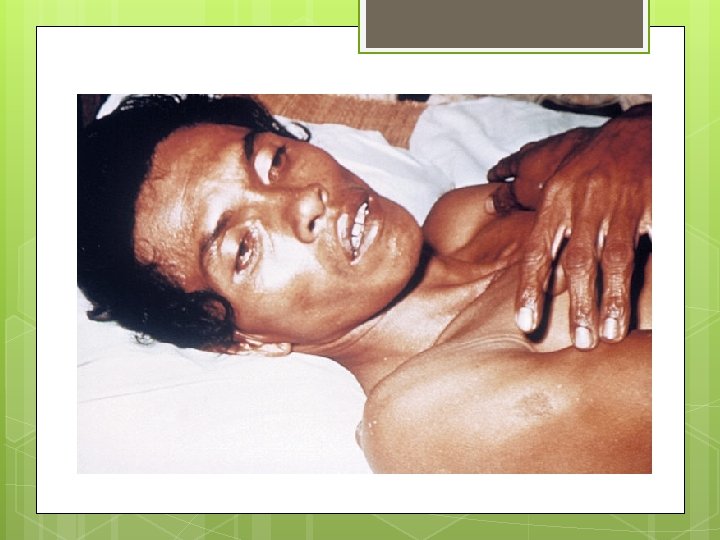

Cholera The disease is caused by the cholera bacteria Vibrio cholerae that releases a toxin called choleragen. Molecules of choleragen bind to the CSM of epithelial cells lining the small intestine and open chloride ion channels which causes chloride ions to flood into the lumen of the small intestine. The increase in chloride ions in the lumen lowers its water potential so water flows from the cells into the lumen resulting in the massive diarrhoea in sufferers.


Oral Rehydration Solutions

Oral Rehydration Solutions Cholera affects the transport of ions from epithelial cells but has very little affect on the cotransport proteins that reabsorb ions in the lower part of the small intestine. If we can get these co-transporters to work better then we could make sure that adequate amounts of glucose and sodium are taken from the lumen into the cells and thus result in much less water loss. This is the basis of O. R. S.

Oral Rehydration Solutions A sachet of glucose and mineral salts are mixed with a set volume of water. The patient drinks the solution and this stimulates the sodium and glucose to be taken up by the co-transporter. This means that water is now absorbed from the lumen of the small intestine and diarrhoea is brought under control.

- Slides: 40