Transition Elements Transition elements are d block elements
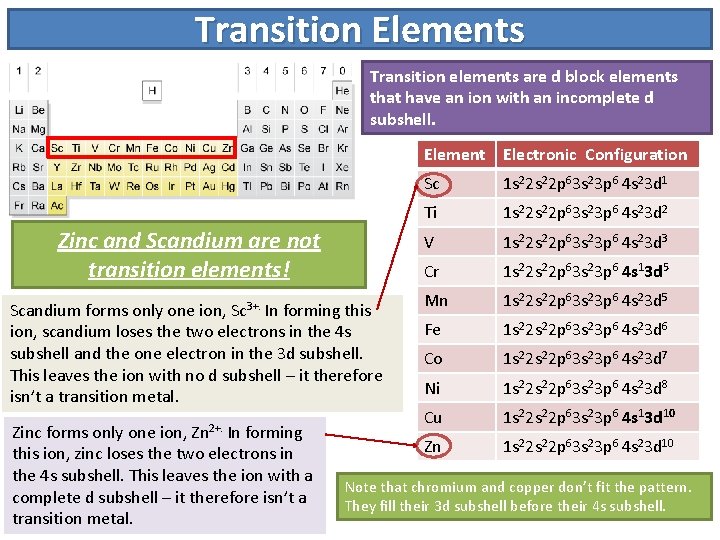
Transition Elements Transition elements are d block elements that have an ion with an incomplete d subshell. Element Electronic Configuration Zinc and Scandium are not transition elements! Scandium forms only one ion, Sc 3+. In forming this ion, scandium loses the two electrons in the 4 s subshell and the one electron in the 3 d subshell. This leaves the ion with no d subshell – it therefore isn’t a transition metal. Zinc forms only one ion, Zn 2+. In forming this ion, zinc loses the two electrons in the 4 s subshell. This leaves the ion with a complete d subshell – it therefore isn’t a transition metal. Sc 1 s 22 p 63 s 23 p 6 4 s 23 d 1 Ti 1 s 22 p 63 s 23 p 6 4 s 23 d 2 V 1 s 22 p 63 s 23 p 6 4 s 23 d 3 Cr 1 s 22 p 63 s 23 p 6 4 s 13 d 5 Mn 1 s 22 p 63 s 23 p 6 4 s 23 d 5 Fe 1 s 22 p 63 s 23 p 6 4 s 23 d 6 Co 1 s 22 p 63 s 23 p 6 4 s 23 d 7 Ni 1 s 22 p 63 s 23 p 6 4 s 23 d 8 Cu 1 s 22 p 63 s 23 p 6 4 s 13 d 10 Zn 1 s 22 p 63 s 23 p 6 4 s 23 d 10 Note that chromium and copper don’t fit the pattern. They fill their 3 d subshell before their 4 s subshell.
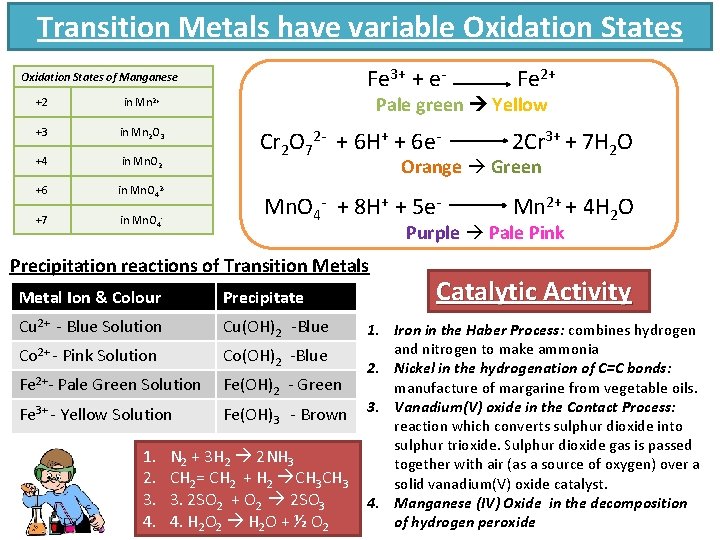
Transition Metals have variable Oxidation States Fe 3+ + e- Oxidation States of Manganese +2 in Mn 2+ +3 in Mn 2 O 3 +4 in Mn. O 2 +6 in Mn. O 42 - +7 in Mn. O 4 - Pale green Yellow Cr 2 O 72 - + 6 H+ + 6 e- 2 Cr 3+ + 7 H 2 O Mn. O 4 - + 8 H+ + 5 e- Mn 2+ + 4 H 2 O Orange Green Purple Pale Pink Precipitation reactions of Transition Metals Metal Ion & Colour Precipitate Cu 2+ - Blue Solution Cu(OH)2 -Blue Co 2+ - Pink Solution Co(OH)2 -Blue Fe 2+- Pale Green Solution Fe(OH)2 - Green Fe 3+ - Yellow Solution Fe(OH)3 - Brown 1. 2. 3. 4. Fe 2+ N 2 + 3 H 2 2 NH 3 CH 2= CH 2 + H 2 CH 3 3. 2 SO 2 + O 2 2 SO 3 4. H 2 O 2 H 2 O + ½ O 2 Catalytic Activity 1. Iron in the Haber Process: combines hydrogen and nitrogen to make ammonia 2. Nickel in the hydrogenation of C=C bonds: manufacture of margarine from vegetable oils. 3. Vanadium(V) oxide in the Contact Process: reaction which converts sulphur dioxide into sulphur trioxide. Sulphur dioxide gas is passed together with air (as a source of oxygen) over a solid vanadium(V) oxide catalyst. 4. Manganese (IV) Oxide in the decomposition of hydrogen peroxide
- Slides: 2