Transfusion and Sickle Cell Anemia Jed B Gorlin

Transfusion and Sickle Cell Anemia Jed B. Gorlin MD, MBA Mn. ABB INNOVATIVEBLOODRESOURCES. ORG | NYBC. ORG | SAVEALIFENOW. ORG

Expanding our organization to meet current clinical, cellular and transfusion product and service needs for patients. Now providing almost 1 million blood products, over 450, 000 laboratory and infectious disease tests and 12, 500 specialty clinical procedures annually to hospitals nationwide.
Transfusion and Sickle Cell Anemia

SEPTEMBER IS SICKLE CELL ANEMIA AWARENESS MONTH http: //www. health. state. m n. us/divs/phl/newborn/fa milies/minnestories. html http: //www. health. state. m n. us/divs/phl/newborn/fa milies/fierceadvocate. pdf MN Newborn screening has great audio clips from family members describing how they learned about their child’s disorder

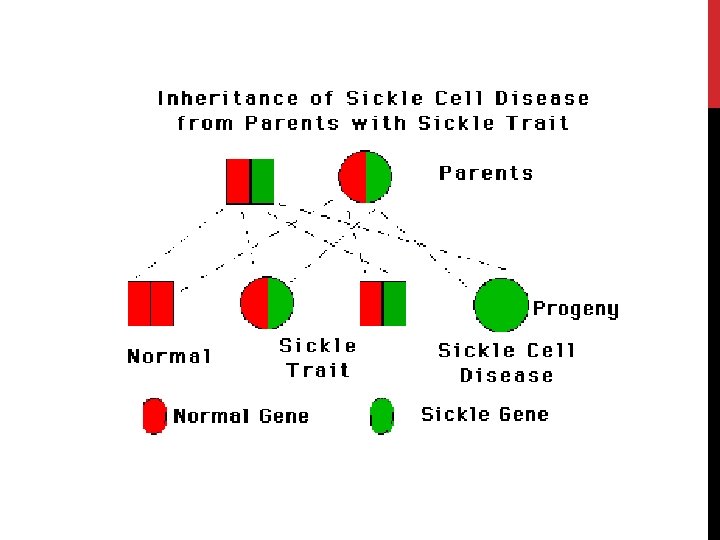
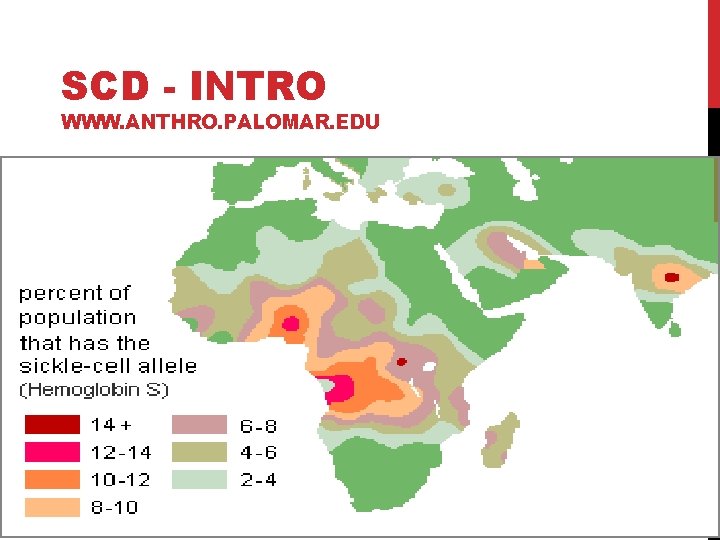
WHAT IS SICKLE CELL ANEMIA? Inherited blood disease affecting primarily African-Americans in the US ~1/600 African-Americans (AA) has sickle cell anemia 1/12 AA carries sickle cell trait Single amino acid change in B-hemoglobin • Valine for a glutamic acid in position 6 Causes hemoglobin to clump in red cells

SCD - INTRO Types of SCD • SS - most common and most severe type • SC - milder form • Sß Thal - in combo with Thalassemia • AS – “Sickle Trait” carrier state • protective natural selection in malaria endemic regions • Other miscellaneous types
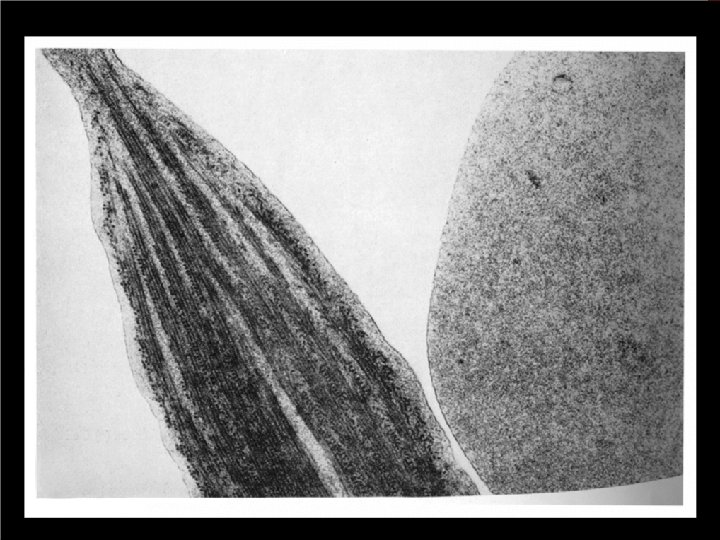
SCD - INTRO WWW. ANTHRO. PALOMAR. EDU

EARLY OBSERVATIONS, PRE 1910 Chwechwe “perpetual chewing” Ahututuo “Aoo, I’m dying” Nwiiwii “I will die, eeh” Nuidudui “body chewing” Adep “beaten up” Hem. Kom “body biting”
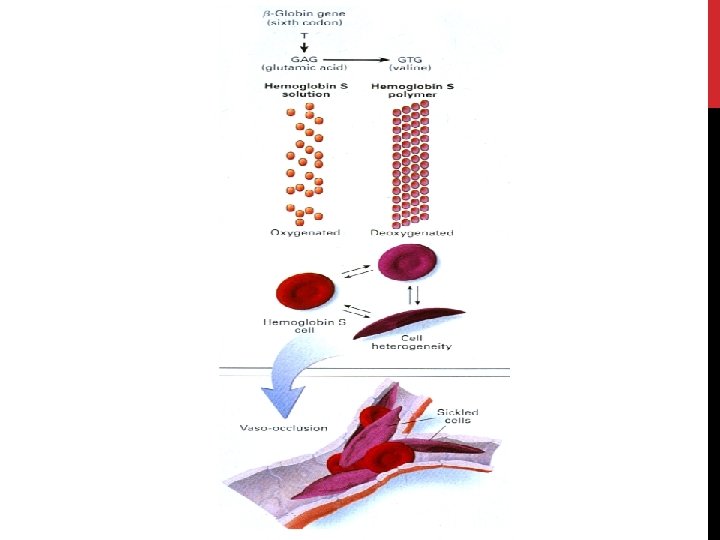
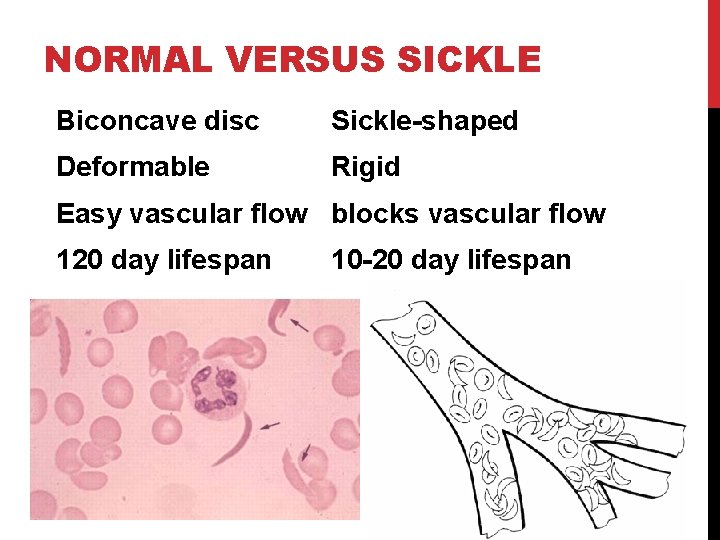
NORMAL VERSUS SICKLE Biconcave disc Sickle-shaped Deformable Rigid Easy vascular flow blocks vascular flow 120 day lifespan 10 -20 day lifespan

COMPLICATIONS OF SICKLE CELL ANEMIA Hemolysis: Red cells don’t last long • Normally have a lower hematocrit ~22% • Aplastic crisis Vessel Occlusion • Splenic sequestration crisis • Painful crises • Stroke • Acute Chest syndrome

TRANSFUSION: WHICH PATIENTS? Mild disease in some patients Others require episodic transfusion Minority require chronic transfusion: • STOP trial was a study using a test called ultrasound to determine who was at highest risk for stroke. Now caregivers are able to start chronic transfusions BEFORE someone has a stroke, thereby preventing brain damage to those at greatest risk.


NEW DEVELOPMENTS, 1990 -TODAY Newborn Screening Hemoglobin F therapy Cellular modulation • Mg Oxide • Clotrimazole Extra-cellular modulation • Nitric Oxide • Adhesion alteration • Anticoagulation • Arginine

NEWBORN SCREENING All 50 US states screen Vital to diagnose sickle cell anemia early to prevent: • Overwhelming pneumococcal infection • Vaccination and penicillin prophylaxis • Splenic sequestration syndrome-alert parents

NEW DEVELOPMENTS, 1990 -TODAY II Transcranial Doppler screening Erythrocytapheresis Oral iron chelation Bone marrow transplantation Gene therapy/Pre-implantation embryo selection Pulmonary hypertension

HYDROXYUREA Is one of several agents known to increase hemoglobin F Hydroxyurea is simple, inexpensive and less likely to promote carcinogenesis Results in 50% lower rates of crises, improved carotid flow and superior outcome (40% lower mortality)

HYDROXYUREA UTILIZATION IN NIGERIA, A LESSON IN PUBLIC HEALTH AUTHORS: ZAKARI Y. ALIYU, ALIYU BABADOKO, AISHA MAMMAN ASH (American Society of Hematology) Survey of 10 hematologists and ~1000 sickle cell patients • No patient (0%) were on hydroxyurea • Only 5% of patients aware of this therapy • Barriers to adoption included hematologists concerns for toxicity, availability, compliance, ability to follow blood counts, affordability

TRANSFUSION OF SCD PTS Used to accomplish two goals • To increase O 2 carrying capacity • To replace abnormal cells Controversial trigger point • guidelines based on pt’s baseline Hgb • 20% or greater reduction from baseline Simple vs. Exchange transfusion
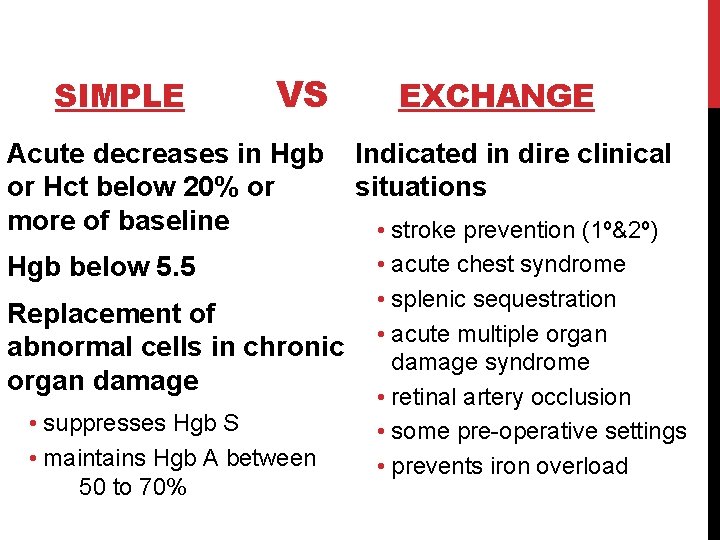
SIMPLE VS Acute decreases in Hgb or Hct below 20% or more of baseline Hgb below 5. 5 Replacement of abnormal cells in chronic organ damage • suppresses Hgb S • maintains Hgb A between 50 to 70% EXCHANGE Indicated in dire clinical situations • stroke prevention (1º&2º) • acute chest syndrome • splenic sequestration • acute multiple organ damage syndrome • retinal artery occlusion • some pre-operative settings • prevents iron overload

LESS PROVEN REASONS TO TRANSFUSE ? Prophylactic transfusion perioperatively • assoc. with fewer sickle-related post-op complications • no accepted standard regimen ? Pregnant pts with SCD ? Leg ulcers ? Priapism ? Acute painful episode

COMPLICATIONS OF RBC TRANSFUSION OF PARTICULAR CONCERN IN SCD PATIENTS Transfusion-related circulatory overload Hyperviscosity syndrome Alloimmunization Hyperhemolytic syndrome Iron overload

TRANSFUSION-RELATED CIRCULATORY OVERLOAD (TACO) Chronic anemia, increased plasma volume places patients at risk of TACO In some situations may need to limit transfusion volumes to 1 -2 m. L/kg/hr • if necessary use aliquots with/without diuretics • may need to use exchange rather than simple transfusion

HYPERVISCOSITY REACTIONS S Viscosity = resistance to flow S RBC transfusion (simple transfusion) increases viscosity S Potential effects of hyperviscosity S Decreases in oxygen delivery S SCA Hbmax = 10 -11 g/d. L S Normal Hbmax = 14 -16 g/d. L S RBC sickling in small blood vessels S RBC adhesion to endothelium

HYPERVISCOSITY REACTIONS S Reactions in SCD patients consisting of headaches, increased blood pressure, altered level of consciousness, seizures thought to be due to hyperviscosity S Prevention S avoid overly-rapid RBC transfusion S avoid transfusing to Hb levels higher than 10 g/d. L S in some cases may need to use exchange rather than simple transfusion to accomplish this

COMPLICATIONS OF TRANSFUSION Immunologic Complications • Alloimmunization • Increased DHTR • Autoimmunization Non-immunologic complications • Hyperviscosity • Iron overload • Transmission of infectious disease (now rare)

ALLOIMMUNIZATION – A PARTICULAR PROBLEM FOR SCD PATIENTS S RBC transfusions are matched for ABO blood group and Rh (D) positive/negative S Other RBC antigen differences between the blood donor and transfusion recipient may lead to alloimmunization (formation of an antibody) Ø most commonly C, E, Kell Ø also Jk a/b, Fy a/b, MNS

ALLOIMMUNIZATION – A PARTICULAR PROBLEM FOR SCD PATIENTS Historically transfused SCD patients have had reported RBC alloimmunization rates of 19 -43% Multiply transfused non-SCD patients have RBC alloimmunization rates of approximately 5% Reason for this? • in North America – different RBC antigen phenotypes of donors vs recipients • also something unique about SCD? ?

PROBLEMS ARISING FROM ALLOIMMUNIZATION S Can become very difficult to find antigen negative/compatible blood units, especially in emergency situations S In pregnancy - hemolytic disease of newborn/fetus S Can cause delayed hemolytic transfusion reactions S If severe “hyperhemolytic syndrome”

COMPLICATIONS OF TRANSFUSION Alloimmunization occurs in 8 -35% of chronically transfused SCD pts commonly Rh system (anti-C, -E), -Jkb, -Fya, -S, -K, • racial discordance: Donors and recipients may have different minor group antigens Delayed Hemolytic Transfusion Reaction (DHTR) is reported to have an incidence of 4 to 22% • Red cell measure (hematocrit) can fall even lower than pretransfusion hematocrit due to “hyperhemolysis” and/or “bystander hemolysis”

PREVENTION OF RBC ALLOIMMUNIZATION S Judicious use of transfusions S Phenotyping/Genotyping of SCD patients S facilitate availability of transfusion records S Minimal “extended” RBC matching S in addition to ABO and Rh (D) - match for C, E, Kell S now considered (by most specialized centres) to be standard care for SCD patients S More extensive matching if the patient develops a clinically significant antibody S some centers do this prophylactically

GENOTYPING: RBC • All patients should be genotyped including Rh variants • Genotyping is cheaper than performing extended phenotyping for more than 4 -6 antigens • Genotyping more accurate for Rh variants • Most centers start with Phenotypic matching of units for Rh (c, C, E) but more extended matching being studied

DELAYED HEMOLYTIC TRANSFUSION REACTION S Destruction of transfused RBCs 7 -10 days following transfusion S In SCD patients may also lead to the destruction of the patient’s own RBCs S Hb drops to lower levels after transfusion than before transfusion S may be accompanied by reticulocytopenia S In SCD, may present as a typical pain crisis
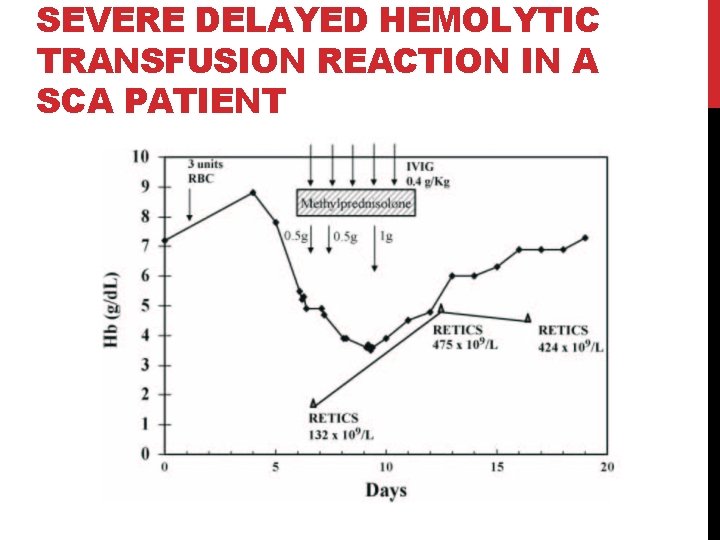
SEVERE DELAYED HEMOLYTIC TRANSFUSION REACTION IN A SCA PATIENT

TREATMENT OF A HYPERHEMOLYTIC TRANSFUSION REACTION If at all possible avoid further transfusion If transfusion is necessary, provide RBCs with the best possible phenotypic match High dose corticosteroids Intravenous immunoglobulin

RBC PRODUCT CHOICES RBC components preferable to whole blood RBC components • no advantage/disadvantage of additive vs nonadditive preservative solutions (except for possibly massive transfusion in the neonate) Blood from Hb. S trait donors • generally not used in North America • interferes with monitoring Hb. S target levels • Clogs LR filters, or fails to be leukoreduced.

LR FAILURES IN UNITS COLLECTED FROM SICKLE TRAIT DONORS: 50% OCCLUDE FILTER, 50% FAIL TO ACHIEVE LR TOP = TRAIT BOTTOM = CTL

AVOIDING RBC TRANSFUSIONS IN SCD PATIENTS Judicious use of RBC transfusions • indications • dose Hydroxyurea • MSH trial (NEJM 1995) • HU group 48/152 underwent transfusion • Placebo group 73/147 underwent transfusion • P<0. 0001
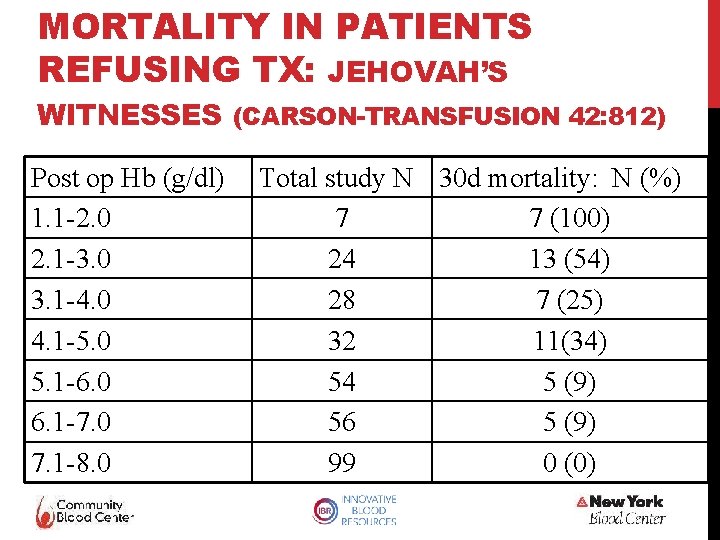
MORTALITY IN PATIENTS REFUSING TX: JEHOVAH’S WITNESSES (CARSON-TRANSFUSION 42: 812) Post op Hb (g/dl) 1. 1 -2. 0 2. 1 -3. 0 3. 1 -4. 0 4. 1 -5. 0 5. 1 -6. 0 6. 1 -7. 0 7. 1 -8. 0 Total study N 30 d mortality: N (%) 7 7 (100) 24 13 (54) 28 7 (25) 32 11(34) 54 5 (9) 56 5 (9) 99 0 (0)

CLINICAL CONSIDERATIONS Multiple antibodies- difficult to find compatible units Avoid Tx if tolerable (makes sense when hgb>4 -5) Risk of mortality increases substantially with hgb <4 Had pre-loaded with IVIG, steroids

BONE MARROW TRANSPLANTATION https: //www. nhlbi. nih. gov/news/spotlight/fa ct-sheet/sky%E 2%80%99 s-limit-womansickle-cell-disease-gets-life-changing-bonemarrow-transplant BUT • Increased rate of intracranial hemorrhage • Risk of acute transplant-related mortality • Risk of chronic graft-vs-host

OTHER OPPORTUNITIES Gene therapy: Active research but challenge is not only making normal protein but decreasing abnormal one https: //www. nature. com/news/crispr-deployed-to-combatsickle-cell-anaemia-1. 20782 Combination erythropoietin-HU-NIH study Agents to decrease adhesion, coagulation, pulmonary hypertension Prevention-Prenatal diagnosis, selective embryo implantation,

RBC TRANSFUSION IN SICKLE CELL DISEASE - CONCLUSION S Transfusions today are safer than ever before, but there does remain a chance of serious adverse events S Know local/regional adverse event rates S As for all treatments - weigh the benefits vs risks, evaluate alternatives S In many situations the benefits are substantial Ø acute situations - life-saving Ø chronic situations - prevention of organ damage, significantly improved quality of life

INNOVATIVEBLOODRESOURCES. ORG | NYBC. ORG | SAVEALIFENOW. ORG
- Slides: 46