Transfemoral Access Devices Tips for Closures Devices James





- Slides: 15

Transfemoral Access Devices & Tips for Closures Devices James P. Zidar, M. D. , F. A. C. C. , F. S. C. A. I Clinical Professor of Medicine, UNC Health Systems UNC Health System Physician-in-Chief, Heart & Vascular Corporate Chief of Cardiology, Rex Healthcare Raleigh, North Carolina

Disclosures • In the past year, I have been on a scientific advisory board, worked as a consultant for, or conducted clinical research for: – • Abbott Vascular, Medtronic, and Amgen All of these companies produce cardiovascular and endovascular products

Closure Devices

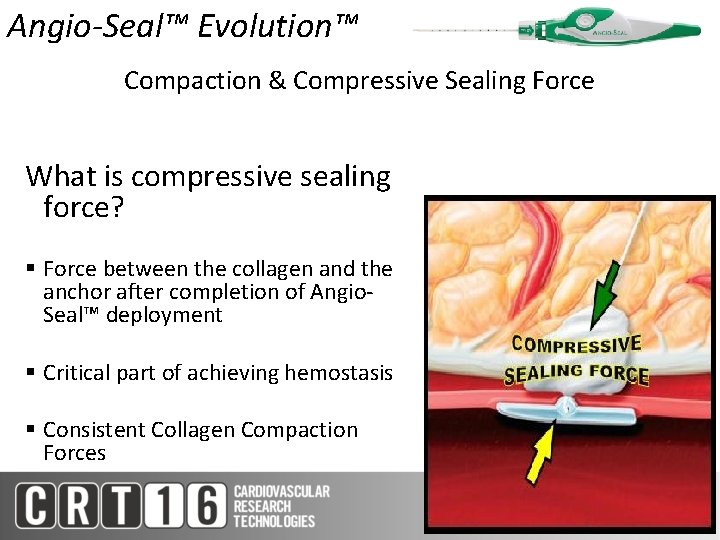
Angio-Seal™ Evolution™ Compaction & Compressive Sealing Force What is compressive sealing force? § Force between the collagen and the anchor after completion of Angio. Seal™ deployment § Critical part of achieving hemostasis § Consistent Collagen Compaction Forces

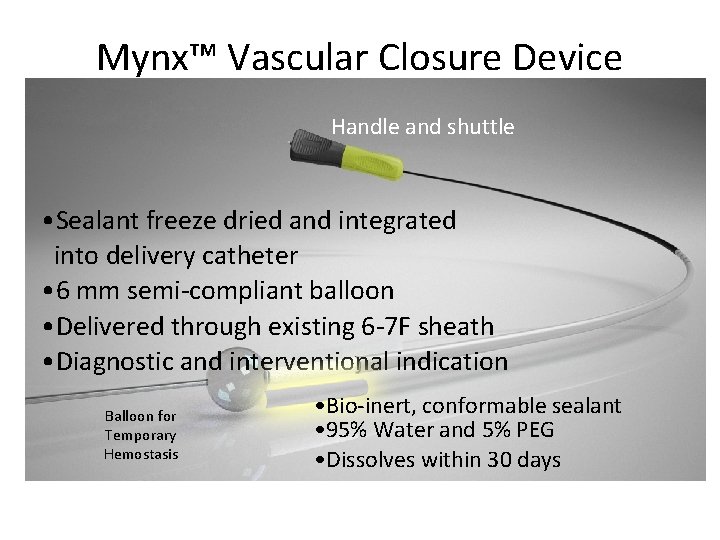
Mynx™ Vascular Closure Device Handle and shuttle • Sealant freeze dried and integrated into delivery catheter • 6 mm semi-compliant balloon • Delivered through existing 6 -7 F sheath • Diagnostic and interventional indication Balloon for Temporary Hemostasis • Bio-inert, conformable sealant • 95% Water and 5% PEG • Dissolves within 30 days

Why Big Holes? 26 Fr 23 Fr 18 Fr 14 Fr 12 Fr • Introduction of lower profile Endovascular thoracic and abdominal devices have expanded the market • Closure devices have facilitated the adoption of transcatheter aortic valves

Most physicians currently use Abbott’s closure devices for large access closure 1, 2 • Suture-based Prostar XL and Perclose Pro. Glide are used to “pre-close” EVAR access sites 1, 2 – Perclose is the preferred device due to its easy of use 1

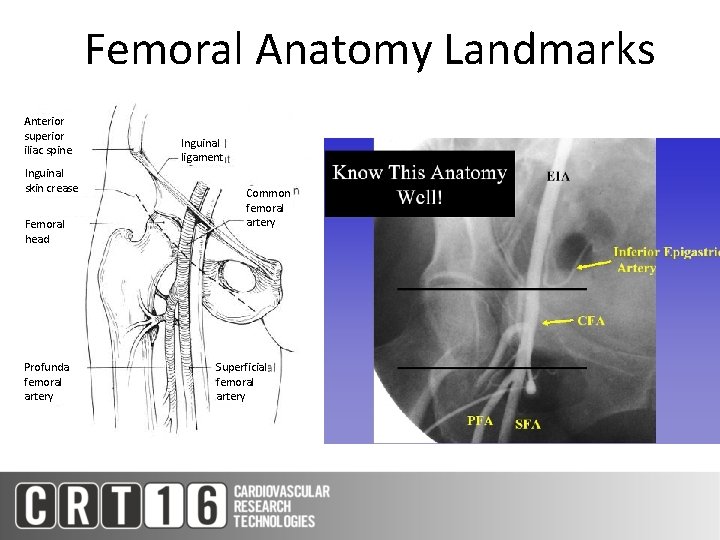
Femoral Anatomy Landmarks Anterior superior iliac spine Inguinal skin crease Femoral head Profunda femoral artery Inguinal ligament Common femoral artery Superficial femoral artery X CFA PFA SFA

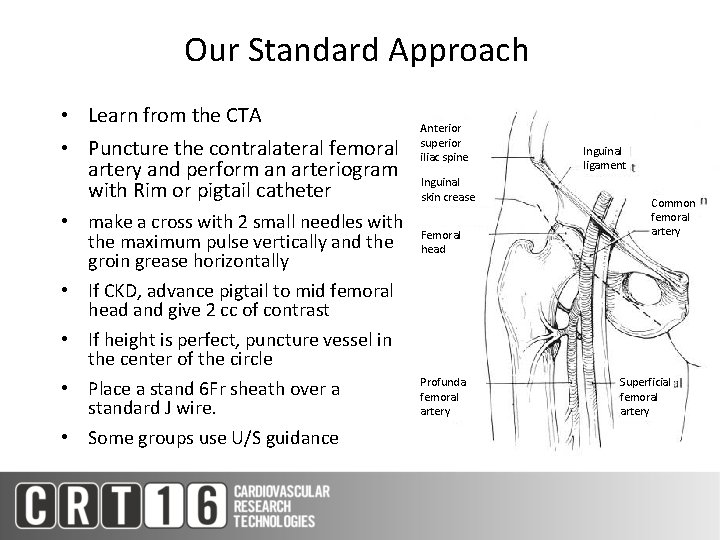
Our Standard Approach • Learn from the CTA • Puncture the contralateral femoral artery and perform an arteriogram with Rim or pigtail catheter • make a cross with 2 small needles with the maximum pulse vertically and the groin grease horizontally • If CKD, advance pigtail to mid femoral head and give 2 cc of contrast • If height is perfect, puncture vessel in the center of the circle • Place a stand 6 Fr sheath over a standard J wire. • Some groups use U/S guidance Anterior superior iliac spine Inguinal skin crease Femoral head Profunda femoral artery Inguinal ligament Common femoral artery Superficial femoral artery

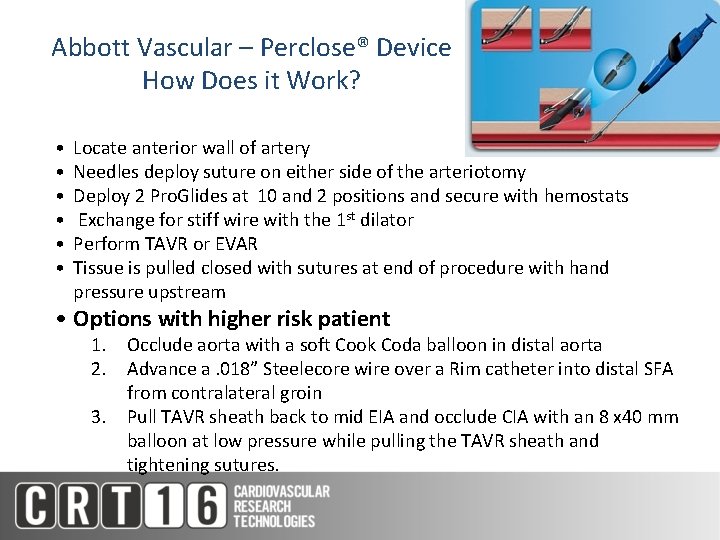
Abbott Vascular – Perclose® Device How Does it Work? • • • Locate anterior wall of artery Needles deploy suture on either side of the arteriotomy Deploy 2 Pro. Glides at 10 and 2 positions and secure with hemostats Exchange for stiff wire with the 1 st dilator Perform TAVR or EVAR Tissue is pulled closed with sutures at end of procedure with hand pressure upstream • Options with higher risk patient 1. Occlude aorta with a soft Cook Coda balloon in distal aorta 2. Advance a. 018” Steelecore wire over a Rim catheter into distal SFA from contralateral groin 3. Pull TAVR sheath back to mid EIA and occlude CIA with an 8 x 40 mm balloon at low pressure while pulling the TAVR sheath and tightening sutures.

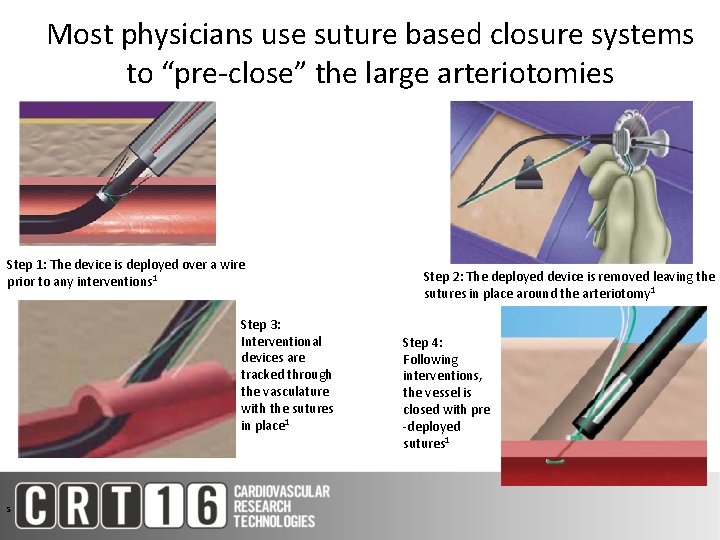
Most physicians use suture based closure systems to “pre-close” the large arteriotomies Step 1: The device is deployed over a wire prior to any interventions 1 Step 3: Interventional devices are tracked through the vasculature with the sutures in place 1 S Step 2: The deployed device is removed leaving the sutures in place around the arteriotomy 1 Step 4: Following interventions, the vessel is closed with pre -deployed sutures 1

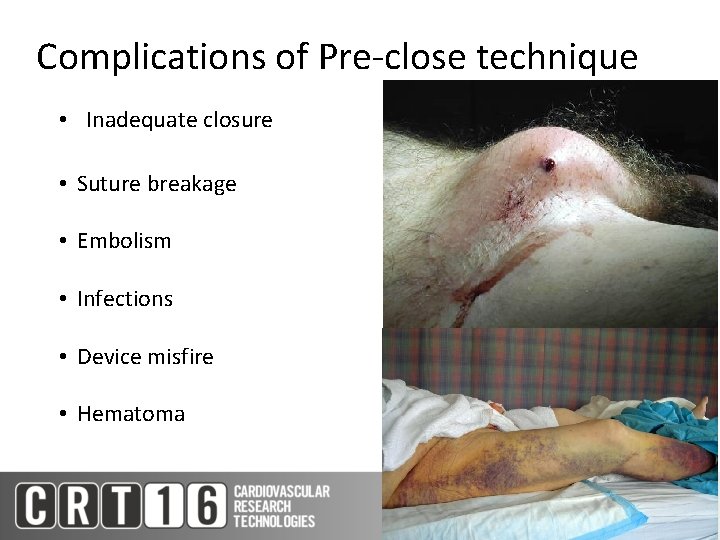
Complications of Pre-close technique • Inadequate closure • Suture breakage • Embolism • Infections • Device misfire • Hematoma

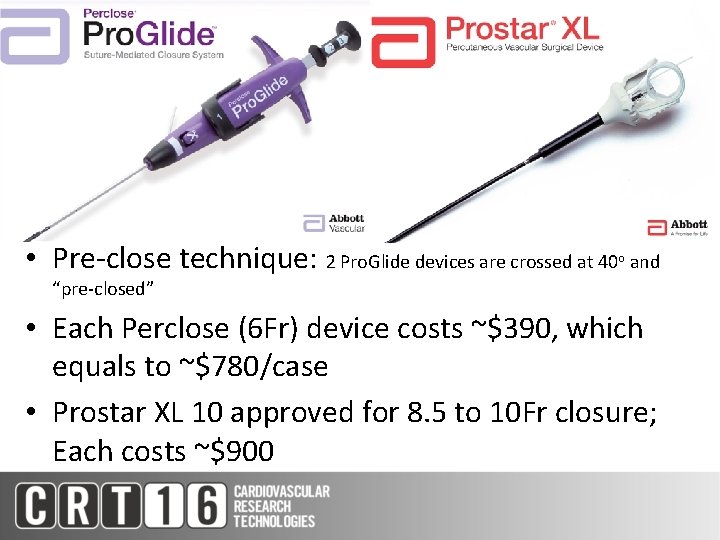
• Pre-close technique: 2 Pro. Glide devices are crossed at 40 o and “pre-closed” • Each Perclose (6 Fr) device costs ~$390, which equals to ~$780/case • Prostar XL 10 approved for 8. 5 to 10 Fr closure; Each costs ~$900

Vascular Closure Devices Have Patient Limitations § Peripheral vascular disease § Bifurcation sticks § Small femoral vessels § Obesity/Low BMI

Caveats • Initial Access Matters • Learning Curve • Spend the extra minute to make an adequate tissue track • We still do not have the ideal device