Transcatheter Aortic Valve Replacement Update Eberhard Grube MD

Transcatheter Aortic Valve Replacement Update Eberhard Grube MD HELIOS Klinikum Siegburg, Germany Instituto Dante Pazzanese de Cardiologia, São Paulo, Brazil Stanford University, Palo Alto, California, USA Siegburg
70’s - Today Standard therapy for critical AS is/was Surgical Aortic Valve Replacement 30 day Mortality 3% Options for s. AVR: Mechanical Tissue Stentless Siegburg
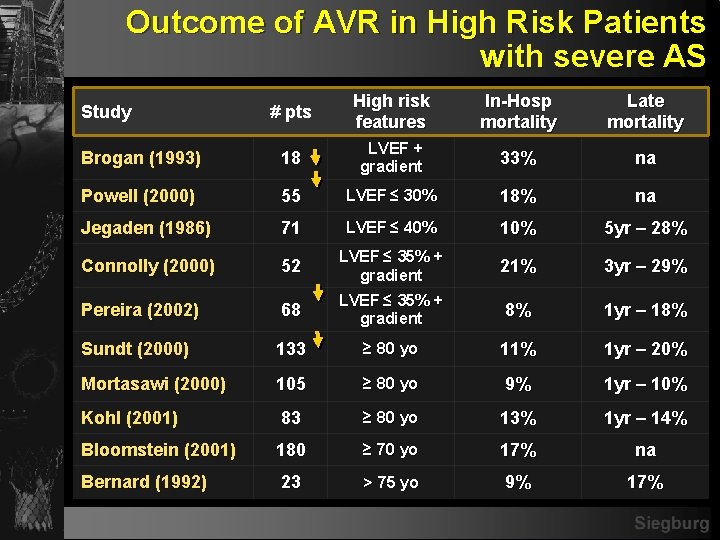
Outcome of AVR in High Risk Patients with severe AS Study # pts High risk features In-Hosp mortality Late mortality LVEF + 33% gradient na LVEF ≤ 30% 18% na LVEF ≤ 40% 10% 5 yr – 28% LVEF ≤ 35% + gradient 21% 3 yr – 29% LVEF ≤ 35% + gradient 8% 1 yr – 18% ≥ 80 yo 11% 1 yr – 20% 105 ≥ 80 yo 9% 1 yr – 10% Kohl (2001) 83 ≥ 80 yo 13% 1 yr – 14% Bloomstein (2001) 180 ≥ 70 yo 17% na Bernard (1992) 23 > 75 yo 9% 17% Brogan (1993) 18 Powell (2000) 55 Jegaden (1986) 71 Connolly (2000) 52 Pereira (2002) 68 Sundt (2000) 133 Mortasawi (2000)

High Risk AVR Patients with Poor Outcomes • • Radiation chest wall/heart disease Octogenarians with multiple co-morbidities Cirrhosis with portal hypertension Porcelain aorta COPD Degenerative neurocognitive dysfunction Previous Cardiac Surgery ESRD (esp. on dialysis)
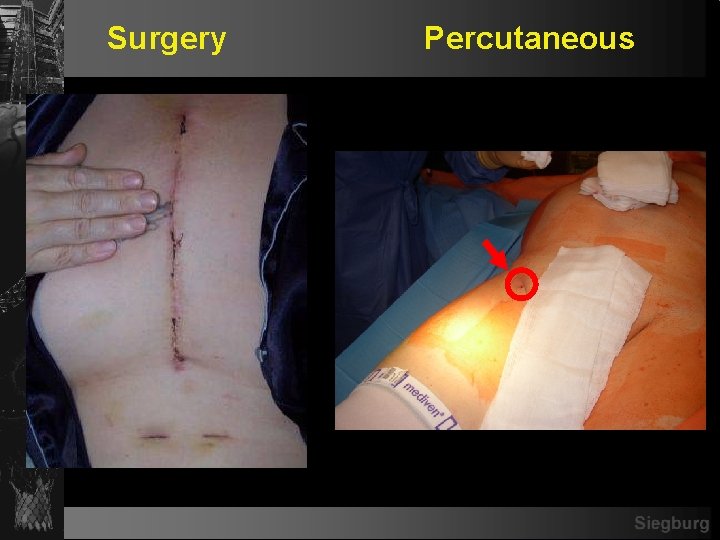
Surgery Percutaneous

Transcatheter AVR Current Generation Devices Edwards ~4, 000 patients Core. Valve ~4, 000 patients
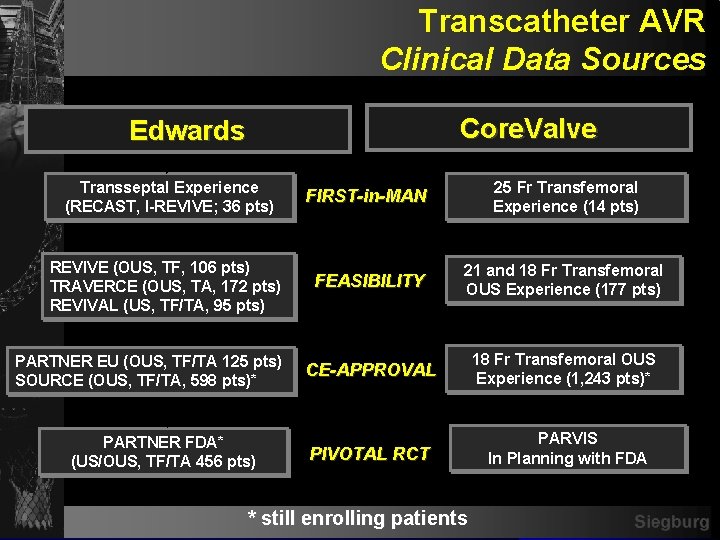
Transcatheter AVR Clinical Data Sources Core. Valve Edwards Transseptal Experience (RECAST, I-REVIVE; 36 pts) FIRST-in-MAN 25 Fr Transfemoral Experience (14 pts) REVIVE (OUS, TF, 106 pts) TRAVERCE (OUS, TA, 172 pts) REVIVAL (US, TF/TA, 95 pts) FEASIBILITY 21 and 18 Fr Transfemoral OUS Experience (177 pts) CE-APPROVAL 18 Fr Transfemoral OUS Experience (1, 243 pts)* PIVOTAL RCT PARVIS In Planning with FDA PARTNER EU (OUS, TF/TA 125 pts) SOURCE (OUS, TF/TA, 598 pts)* PARTNER FDA* (US/OUS, TF/TA 456 pts) * still enrolling patients Siegburg
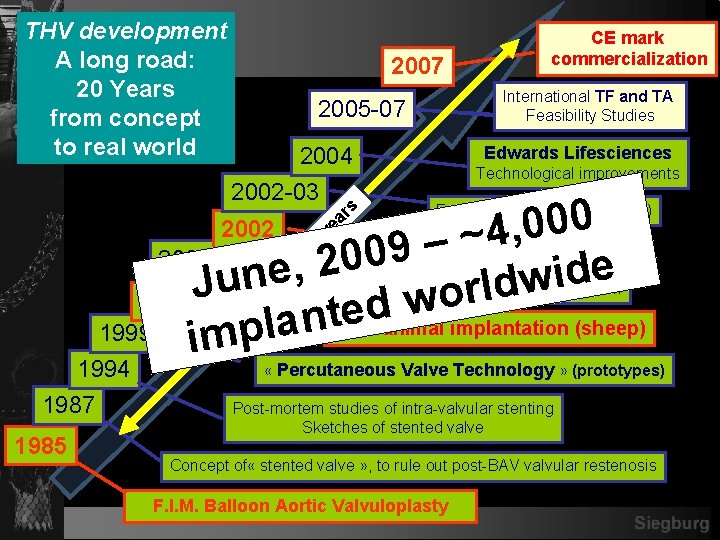
THV development A long road: 20 Years from concept to real world 2007 International TF and TA Feasibility Studies 2005 -07 2004 2002 -03 CE mark commercialization Edwards Lifesciences Technological improvements 20 y ea rs 0 0 0 , 4 ~ F. I. M. PHV implantation – 9 0 0 2 , e e d i n w u d l r o 2000 J w d e t. First animal implantation (sheep) n a l 1999 p im 2002 2000 -02 Feasibility Studies (antegrade) Large series of animal implantation 1994 1987 1985 « Percutaneous Valve Technology » (prototypes) Post-mortem studies of intra-valvular stenting Sketches of stented valve Concept of « stented valve » , to rule out post-BAV valvular restenosis F. I. M. Balloon Aortic Valvuloplasty Siegburg

TAVI – Core. Valve Number of Countries & Centers Siegburg

TAVI – Core. Valve Number of Cases 9 Siegburg
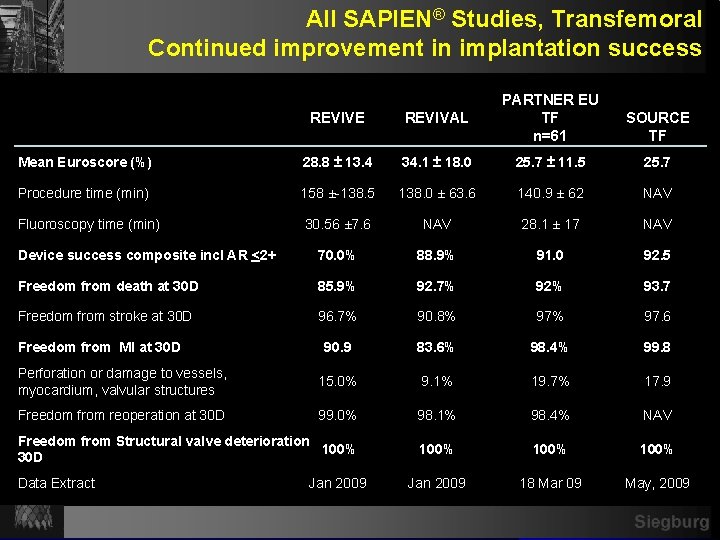
All SAPIEN® Studies, Transfemoral Continued improvement in implantation success REVIVE REVIVAL PARTNER EU TF n=61 Mean Euroscore (%) 28. 8 ± 13. 4 34. 1 ± 18. 0 25. 7 ± 11. 5 25. 7 Procedure time (min) 158 ±-138. 5 138. 0 ± 63. 6 140. 9 ± 62 NAV 30. 56 ± 7. 6 NAV 28. 1 ± 17 NAV Device success composite incl AR <2+ 70. 0% 88. 9% 91. 0 92. 5 Freedom from death at 30 D 85. 9% 92. 7% 92% 93. 7 Freedom from stroke at 30 D 96. 7% 90. 8% 97. 6 90. 9 83. 6% 98. 4% 99. 8 Perforation or damage to vessels, myocardium, valvular structures 15. 0% 9. 1% 19. 7% 17. 9 Freedom from reoperation at 30 D 99. 0% 98. 1% 98. 4% NAV Freedom from Structural valve deterioration 100% 30 D 100% Jan 2009 18 Mar 09 May, 2009 Fluoroscopy time (min) Freedom from MI at 30 D Data Extract Jan 2009 SOURCE TF Siegburg
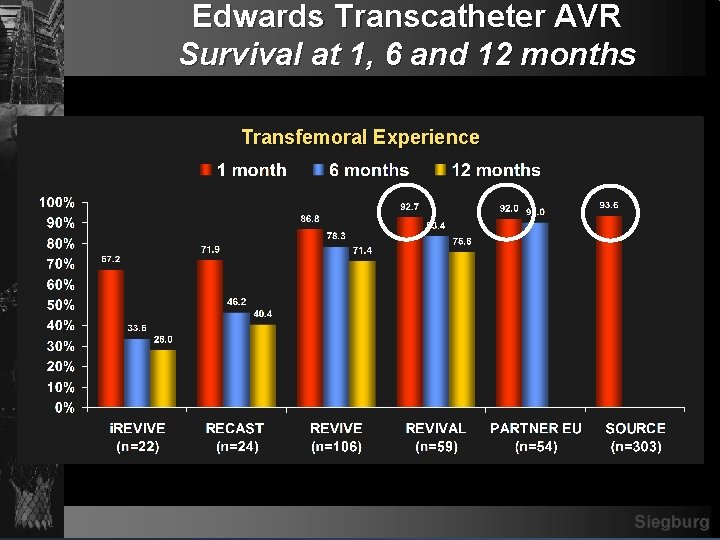
Edwards Transcatheter AVR Survival at 1, 6 and 12 months Transfemoral Experience
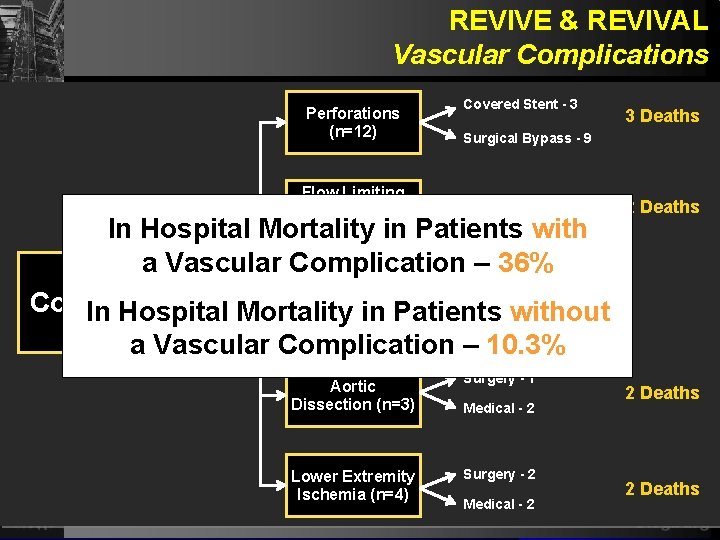
REVIVE & REVIVAL Vascular Complications Perforations (n=12) Flow Limiting Iliac Dissection (n=4) Covered Stent - 3 3 Deaths Surgical Bypass - 9 Surgical Repair - 4 In Hospital Mortality in Patients with a Vascular Complication – 36% Vascular Avulsed Iliac Surgical Bypass - 3 Complications Artery (n=3) In Hospital Mortality in Patients without (n=25) a Vascular Complication – 10. 3% Aortic Dissection (n=3) Lower Extremity Ischemia (n=4) Surgery - 1 Medical - 2 Surgery - 2 Medical - 2 2 Deaths Siegburg

REVIVE improved screening to reduce vascular complications Vascular Access Complication Rate, by patient January 2006 – June 2006 30% Vascular Screening with Columbia University Medical Center Core Lab INSTITUTED (in coordination with DSMB) Vascular Access Complication Rate, by patient August 2006 – December 2007 5. 8% P =. 006 Siegburg
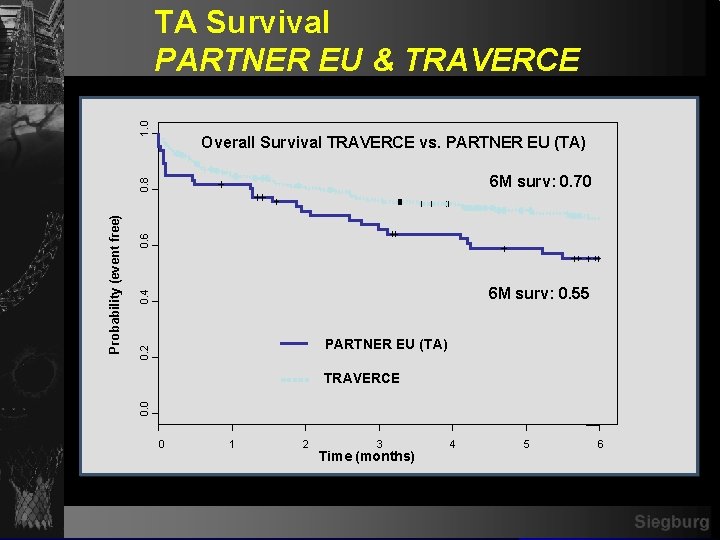
1. 0 TA Survival PARTNER EU & TRAVERCE Overall Survival TRAVERCE vs. PARTNER EU (TA) 0. 6 0. 4 6 M surv: 0. 55 0. 2 PARTNER EU (TA) TRAVERCE 0. 0 Probability (event free) 0. 8 6 M surv: 0. 70 0 1 2 3 Time (months) 4 5 6 Siegburg
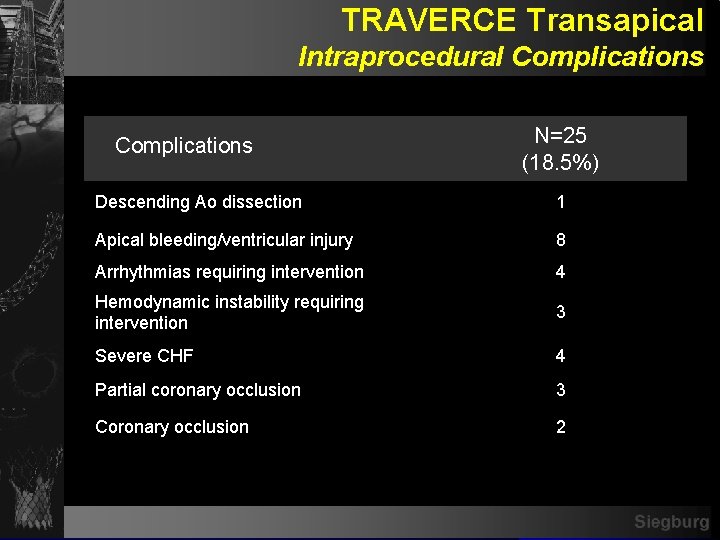
TRAVERCE Transapical Intraprocedural Complications N=25 (18. 5%) Descending Ao dissection 1 Apical bleeding/ventricular injury 8 Arrhythmias requiring intervention 4 Hemodynamic instability requiring intervention 3 Severe CHF 4 Partial coronary occlusion 3 Coronary occlusion 2 Siegburg
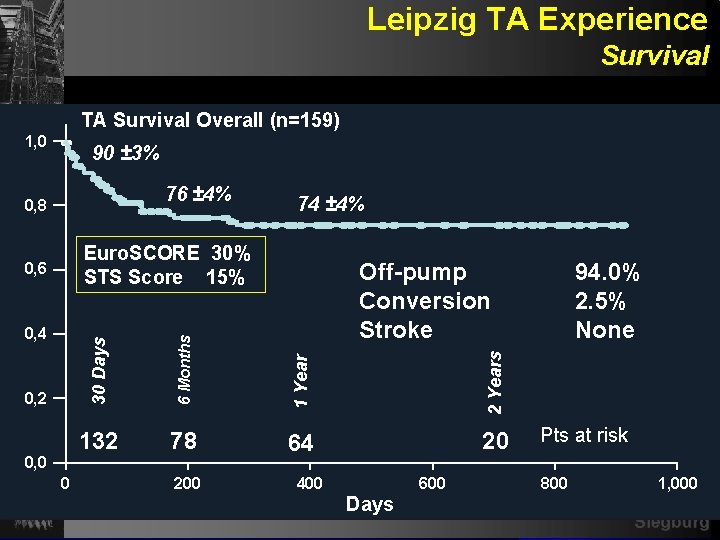
Leipzig TA Experience Survival TA Survival Overall (n=159) 1, 0 90 ± 3% 76 ± 4% 0, 8 74 ± 4% Euro. SCORE 30% STS Score 15% 0 2 Years 0, 0 1 Year 0, 2 6 Months 0, 4 Off-pump Conversion Stroke 30 Days 0, 6 132 78 64 20 200 400 Days 600 94. 0% 2. 5% None Pts at risk 800 1, 000 Siegburg
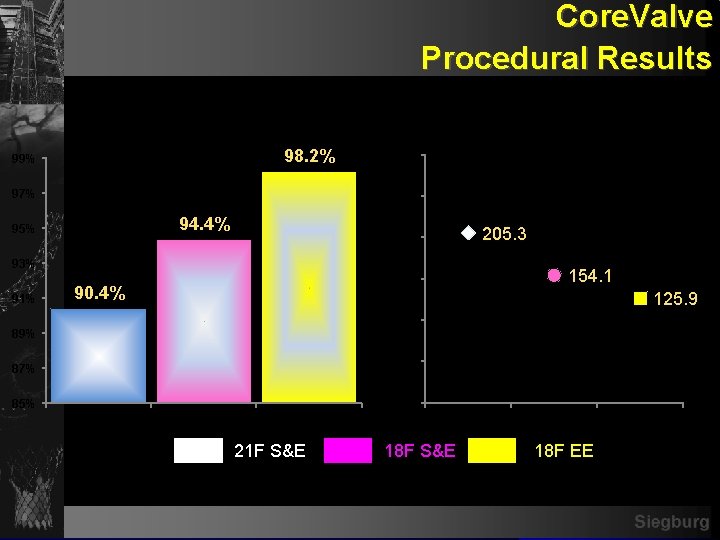
Core. Valve Procedural Results Procedure Success 98. 2% 99% 97% Procedure Mean Time ± SD 300 250 94. 4% 95% 200 93% 91% 150 90. 4% 205. 3 154. 1 125. 9 100 89% 50 87% 0 85% 21 F S&E 18 F EE Siegburg
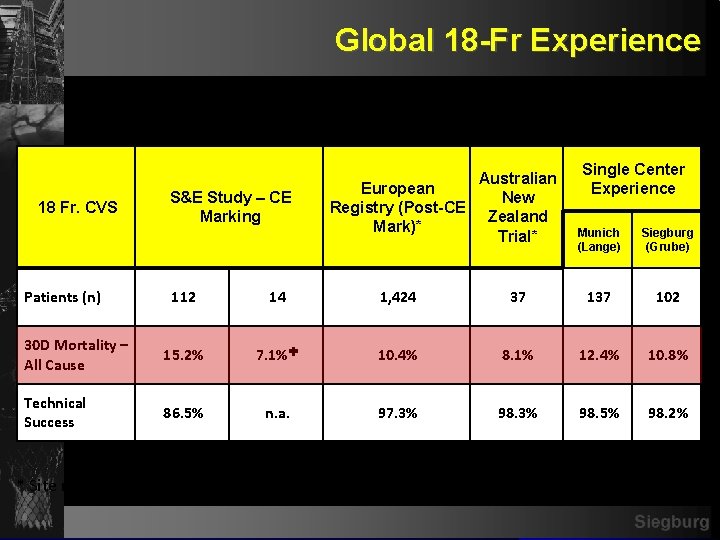
Global 18 -Fr Experience S&E Study – CE Marking 18 Fr. CVS Patients (n) Australian European New Registry (Post-CE Zealand Mark)* Trial* Single Center Experience Munich (Lange) Siegburg (Grube) 112 14 1, 424 37 102 30 D Mortality – All Cause 15. 2% 7. 1%✚ 10. 4% 8. 1% 12. 4% 10. 8% Technical Success 86. 5% n. a. 97. 3% 98. 5% 98. 2% * Site reported ✚ Un-adjudicated Siegburg
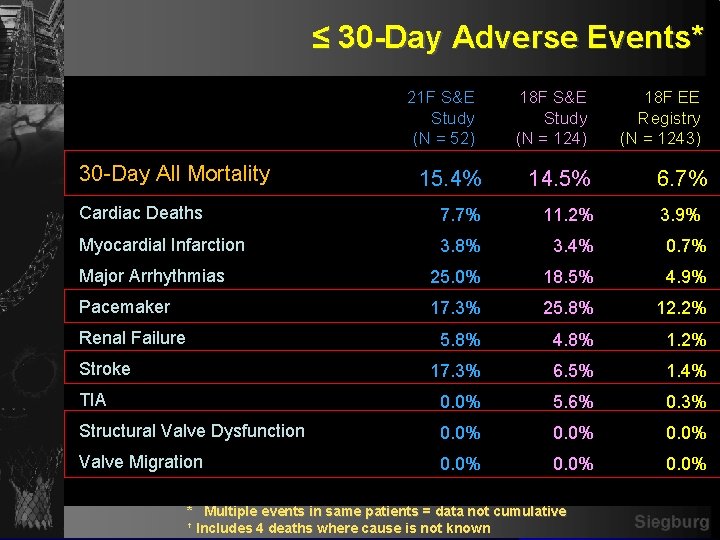
≤ 30 -Day Adverse Events* 21 F S&E Study (N = 52) 30 -Day All Mortality 18 F S&E Study (N = 124) 18 F EE Registry (N = 1243) 15. 4% 14. 5% 6. 7% Cardiac Deaths 7. 7% 11. 2% 3. 9%† Myocardial Infarction 3. 8% 3. 4% 0. 7% Major Arrhythmias 25. 0% 18. 5% 4. 9% Pacemaker 17. 3% 25. 8% 12. 2% 5. 8% 4. 8% 1. 2% 17. 3% 6. 5% 1. 4% TIA 0. 0% 5. 6% 0. 3% Structural Valve Dysfunction 0. 0% Valve Migration 0. 0% Renal Failure Stroke * Multiple events in same patients = data not cumulative † Includes 4 deaths where cause is not known Siegburg

Siegburg Core. Valve Experience Total number of patients*: 387 Gen 1 (2005) 10 Gen 2 (2005 -2006) 26 Gen 3 (since 2006) 257 in 2008 217 in 2009 6 -10/week 30 Day Mortality: Gen 1 Gen 2 Gen 3 in 2008 *Status of Nov 2008 40. 0% 20. 8% 8. 6% 4. 8% Siegburg

TAVI Candidate Today: Who is Eligible? Morphological Criteria: (Mandatory) Clinical Criteria: ? • • Native Aortic Valve Disease Severe AS: AVAI ≤ 0. 6 cm 2/m 2 27 mm ≥AV annulus ≥ 20 mm Sino-tubular Junction ≤ 43 mm Logistic Euro. SCORE ≥ 20% (21 F) ≥ 15% (18 F) Age ≥ 80 y (21 F) ≥ 75 y (18 F) Age ≥ 65 y plus 1+ of the following: • • • Liver cirrhosis (Child A or B) Pulmonary insufficiency: FEV 1<1 L Previous cardiac surgery PHT (PAP>60 mm. Hg) Recurrent P. E’s RV failure Hostile thorax (radiation, burns, etc) Severe connective tissue disease Cachexia Siegburg
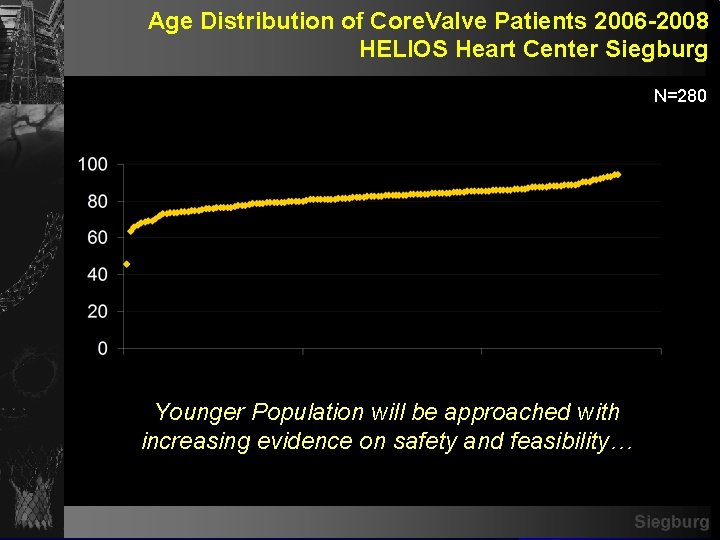
Age Distribution of Core. Valve Patients 2006 -2008 HELIOS Heart Center Siegburg N=280 Younger Population will be approached with increasing evidence on safety and feasibility… Siegburg
Morphologic Criteria must be met Morphological Quantification Siegburg

Which is the preferred access? Surgical Transapical Subclavian Interventional Transfemoral Complexity / Invasiveness Siegburg
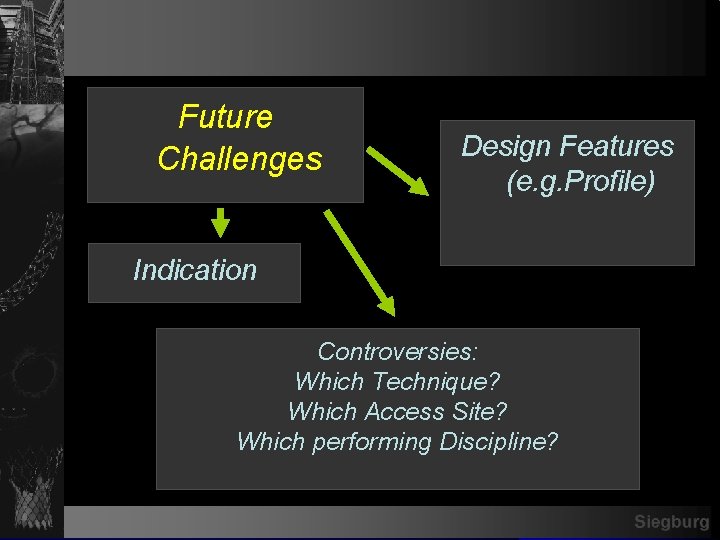
Future Challenges Design Features (e. g. Profile) Indication Controversies: Which Technique? Which Access Site? Which performing Discipline? Siegburg

‘Percutaneous Devices for Aortic Valve Replacement’ Potential problems of current devices – – Paravalvular leackage Inaccuracies in Positioning Embolization (Edwards prosthesis) ‘One shot’ procedure Siegburg
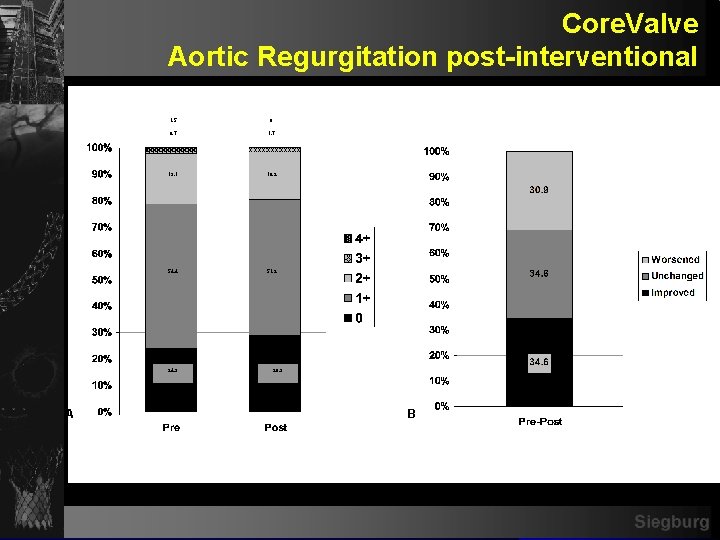
Core. Valve Aortic Regurgitation post-interventional 1. 5 0 0. 7 19. 1 18. 2 54. 4 51. 2 24. 3 A 28. 9 B Siegburg

The Sadra Lotus Valve - Device Features and Rationale TM Locking mechanism Adaptive. TM Seal
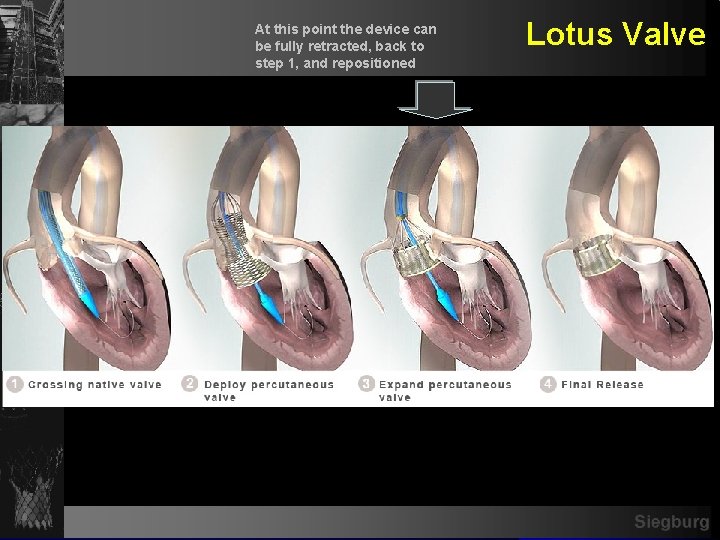
At this point the device can be fully retracted, back to step 1, and repositioned Lotus Valve Siegburg
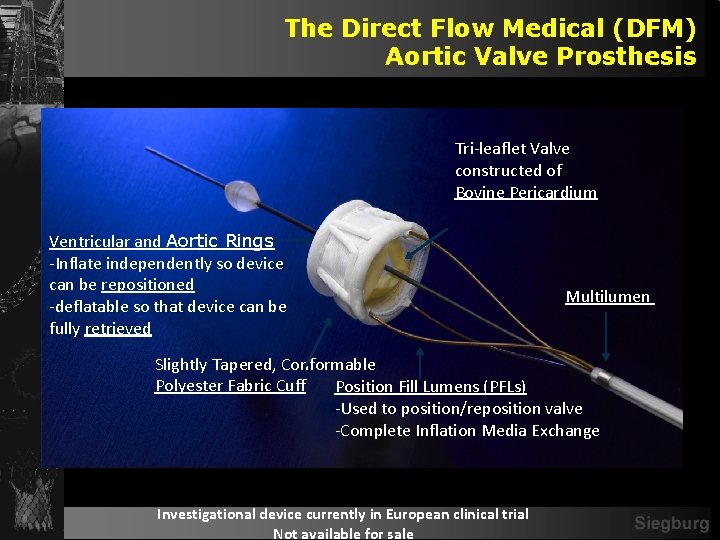
The Direct Flow Medical (DFM) Aortic Valve Prosthesis Tri-leaflet Valve constructed of Bovine Pericardium Ventricular and Aortic Rings -Inflate independently so device can be repositioned -deflatable so that device can be fully retrieved Multilumen Slightly Tapered, Conformable Polyester Fabric Cuff Position Fill Lumens (PFLs) -Used to position/reposition valve -Complete Inflation Media Exchange Investigational device currently in European clinical trial Not available for sale
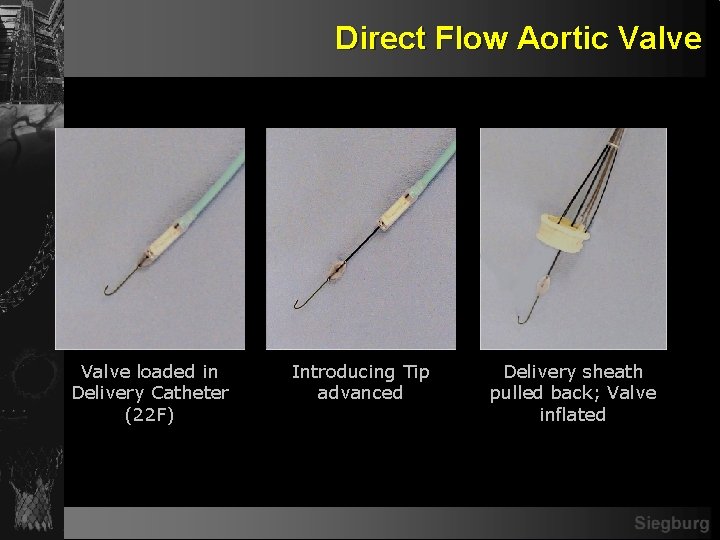
Direct Flow Aortic Valve loaded in Delivery Catheter (22 F) Introducing Tip advanced Delivery sheath pulled back; Valve inflated

My Prediction: Repetition of an Old Story PCI TAVI CABG 1980’s, 1990’s s. AVR 2000’s, 2010’s With the same result… Siegburg
Transcatheter AVR My Rosey Prophecy Surgery – The PAST In the next 5 -10 years, most patients with severe AS requiring AVR will be treated using transcatheter lesser-invasive modalities! TAVR – The Future Siegburg

Vielen Dank Siegburg
- Slides: 35