Training in Stable Isotope Methods Mass Spectrometery Isotopology

Training in Stable Isotope Methods, Mass Spectrometery & Isotopology IB 400 Todd Dawson (2 -6090) tdawson@berkeley. edu Paul Brooks (3 -1748) isotopes@berkeley. edu Kevin Simonin (2 -1054) ksimonin@berkeley. edu & Elizabeth Wenk for all her powerpoint magic 9 -28 -05 Version

Training in Stable Isotope Methods, Mass Spectrometery & Isotopology Goal: provide some of the fundamentals needed to understand isotopes and their use in the biological sciences Six 1. 2. 3. 4. 4. 5. 6. 6. sessions: Fundamentals of isotope physics & chemistry Case studies of how stable isotopes have been applied in natural systems Sampling issues: what, where, how and help! of isotope sample collection and preparation The isotope ratio mass spectrometer: how it works and how you make it work for your research 5. Correcting your data - tour de Excel! Discussion about isotope applications & your work - the questions you face and the challenges you need to resolve » Introductions: who you are, what you do, why isotopes?

TODAY: Introduction to Stable Isotopes 1. Introduction to isotopes 2. Isotopes used in ecological studies 3. Stable isotope notation 4. Correct usage of stable isotope expressions 5. Causes of variation in stable isotope abundances 6. Fractionation factors 7. Rayleigh distillation: using fractionation factors

Why use stable isotopes? » They are non-radioactive TRACERS of resource origin, fate and flux including: 1. organismal movements 2. energy or resource flow across levels of ecological organization » They are non-destructive and non-disruptive INTEGRATORS of ‘system’ processes including: 1. organismal function/tradeoffs 2. spatial and temporal responses to environment

What are isotopes?

History of Isotopy: • 1914 Fredrick Soddy proposed that any place on periodic table can be occupied by more than one kind of atom He proposed isotope ---- meaning “same place” Isotopes Nuclides of single element having different atomic weights • Presently more than 2500 nuclides are known from ~110 elements

To be or not to be? - A stable isotope that is!

Nuclear Entities These entities are of greatest interest to us

Atoms First: First An atom is composed of three types of particles: PROTONS, PROTONS NEUTRONS, NEUTRONS and ELECTRONS Each element has a unique number of protons, its atomic number. The number of protons (Z) determines many of the chemical and physical properties associated with an atom. The atomic mass is thenumber sum of protons and neutrons (N), The atomic = Z (Protons) particles with nearly identical weights. (Electrons, negatively charged particles, have insignificant weight – The atomic mass = Z + N (Protons + Neutrons) to ecologists!)

Partial chart of the elements Each square represents a nuclide, an isotope specific atom Atomic number = Z (Protons): bottom left of each atom Atomic mass = Z + N (Protons + Neutrons): top left of each atom 13 8 Proton Number (Z) 8 O 7 6 9 6 5 8 5 4 6 4 3 5 3 3 2 2 1 1 1 H 0 He 2 1 H 1 4 2 Be Li He 3 1 7 4 10 6 Li He C 9 5 O N 13 7 N 14 7 11 6 C 12 6 C 13 6 10 5 8 4 Be 9 4 Be 10 4 7 3 Li 8 3 Li 9 3 6 2 15 8 12 7 B B Be 6 3 5 2 C 14 8 B 11 5 8 2 He B Be N C 12 5 11 4 O B Be 16 8 O 17 8 O 18 8 O 15 7 N 16 7 N 17 7 N 14 6 C 15 6 C 16 6 C 19 8 18 7 O 20 8 O N 13 4 B 12 4 Be Li He H 2 3 4 5 6 7 8 Neutron Number (N) 9 10 11 12 13
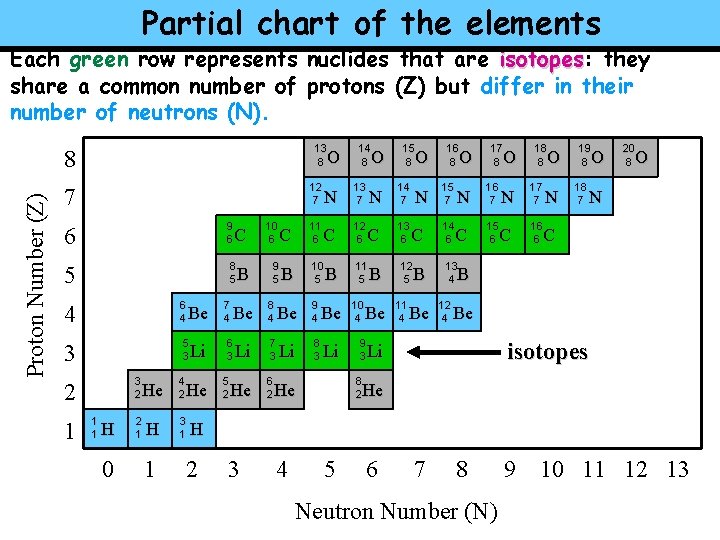
Partial chart of the elements Each green row represents nuclides that are isotopes: isotopes they share a common number of protons (Z) but differ in their number of neutrons (N). 13 8 Proton Number (Z) 8 O 12 7 7 6 9 6 5 8 5 4 6 4 3 5 3 3 2 2 1 1 1 H 0 He 2 1 H 1 4 2 Be Li He 3 1 7 4 B Be 6 3 5 2 C Li He 10 6 9 5 8 4 O N 13 7 C 12 6 B 10 5 Be 9 4 Be 10 4 8 3 Li 9 3 7 3 6 2 11 6 C 14 8 Li B 15 8 O N 14 7 C 13 6 11 5 B Be N C 12 5 B 11 4 Be 16 8 O 15 7 17 8 O N 16 7 C 15 6 14 6 18 8 O N 17 7 C 16 6 N 19 8 O 18 7 20 8 O N C 13 4 B 12 4 Be isotopes Li 8 2 He He H 2 3 4 5 6 7 8 Neutron Number (N) 9 10 11 12 13

Partial chart of the elements Each green row represents nuclides that are isobars: isobars they share a common atomic weight (N + Z). 13 8 Proton Number (Z) 8 O 12 7 7 6 9 6 5 8 5 C B 4 6 4 Be 7 4 3 5 3 6 3 3 2 2 1 1 1 H 0 He 2 1 H 1 Li 4 2 He 3 1 5 2 Be Li He 10 6 C 9 5 8 4 O N 13 7 C 12 6 B 10 5 Be 9 4 Be 10 4 8 3 Li 9 3 7 3 6 2 11 6 14 8 Li B 15 8 O N 14 7 C 13 6 11 5 B Be N C 12 5 B 11 4 Be He 3 4 5 15 7 17 8 N 16 7 C 15 6 14 6 18 8 O O N 17 7 C 16 6 N 19 8 O 18 7 20 8 O N C 13 4 B 12 4 Be isobars H 2 O Li 8 2 He 16 8 6 7 8 Neutron Number (N) 9 10 11 12 13

Partial chart of the elements Each green row represents nuclides that are isotones: isotones they share a common number of neutrons (N). 13 8 Proton Number (Z) 8 O 12 7 7 6 9 6 5 8 5 C B 4 6 4 Be 7 4 3 5 3 6 3 3 2 2 1 1 1 H 0 He 2 1 H 1 Li 4 2 He 3 1 5 2 Be Li He 10 6 C 9 5 8 4 O N 13 7 C 12 6 B 10 5 Be 9 4 Be 10 4 8 3 Li 9 3 7 3 6 2 11 6 14 8 Li B 15 8 O N 14 7 C 13 6 11 5 B Be N C 12 5 B 11 4 Be 16 8 O 15 7 17 8 O N 16 7 C 15 6 14 6 18 8 O N 17 7 C 16 6 N 19 8 O 18 7 20 8 O N C 13 4 B 12 4 Be Li isotones 8 2 He He H 2 3 4 5 6 7 8 Neutron Number (N) 9 10 11 12 13
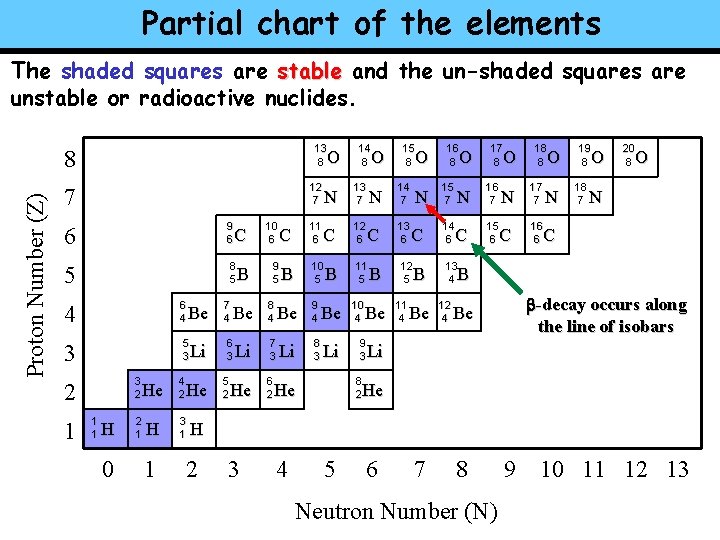
Partial chart of the elements The shaded squares are stable and the un-shaded squares are unstable or radioactive nuclides. 13 8 Proton Number (Z) 8 O 12 7 7 6 9 6 5 8 5 4 6 4 3 5 3 3 2 2 1 1 1 H 0 He 2 1 H 1 4 2 Be Li He 3 1 7 4 B Be 6 3 5 2 C Li He 10 6 9 5 8 4 O N 13 7 C 12 6 B 10 5 Be 9 4 Be 10 4 8 3 Li 9 3 7 3 6 2 11 6 C 14 8 Li B 15 8 O N 14 7 C 13 6 11 5 B Be N C 12 5 B 11 4 Be 16 8 O 15 7 17 8 O N 16 7 C 15 6 14 6 18 8 O N 17 7 C 16 6 N 19 8 O 18 7 20 8 O N C 13 4 B 12 4 -decay occurs along the line of isobars Be Li 8 2 He He H 2 3 4 5 6 7 8 Neutron Number (N) 9 10 11 12 13

Stable isotope trends 3 important points: 1. Stable isotopes tend to have an N/Z near 1 for masses less than 20 2. Stable isotopes tend to have an even Z-number for masses greater than 20 3. Most biologically important elements have masses less than 20 N N/Z = 1 O H C Z N

What isotopes are used in ecological studies?
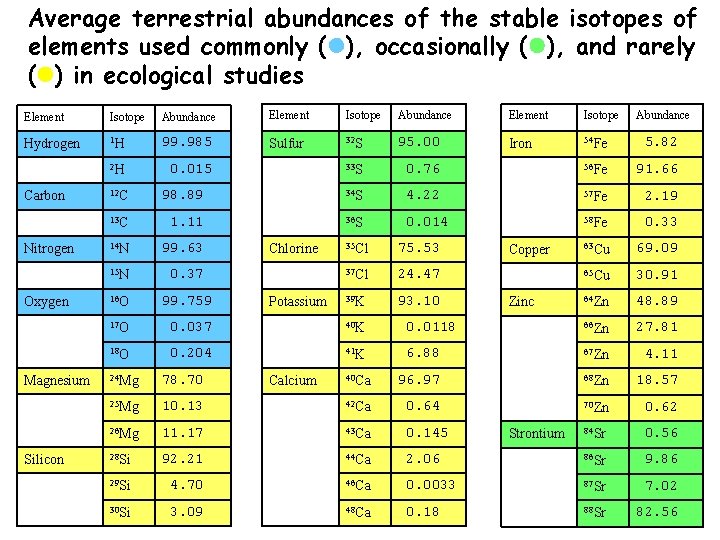
Average terrestrial abundances of the stable isotopes of elements used commonly ( ), occasionally ( ), and rarely ( ) in ecological studies Element Isotope Abundance Element Isotope Hydrogen 1 H 99. 985 Sulfur 32 S 95. 00 Iron 54 Fe 5. 82 2 H 0. 015 33 S 0. 76 56 Fe 91. 66 Carbon Nitrogen Oxygen Magnesium Silicon Abundance 12 C 98. 89 34 S 4. 22 57 Fe 2. 19 13 C 1. 11 36 S 0. 014 58 Fe 0. 33 14 N 99. 63 63 Cu 69. 09 15 N 0. 37 65 Cu 30. 91 16 O 99. 759 64 Zn 48. 89 17 O 0. 037 40 K 0. 0118 66 Zn 27. 81 18 O 0. 204 41 K 6. 88 67 Zn 4. 11 40 Ca 96. 97 68 Zn 18. 57 70 Zn 0. 62 84 Sr 0. 56 Chlorine Potassium 35 Cl 75. 53 37 Cl 24. 47 39 K 93. 10 Copper Zinc 24 Mg 78. 70 25 Mg 10. 13 42 Ca 0. 64 26 Mg 11. 17 43 Ca 0. 145 28 Si 92. 21 44 Ca 2. 06 86 Sr 9. 86 29 Si 4. 70 46 Ca 0. 0033 87 Sr 7. 02 30 Si 3. 09 48 Ca 0. 18 88 Sr 82. 56 Calcium Strontium

Common isotopes When we see this list of isotopes used in ecological studies, note that it includes many of the most common isotopes in the solar system: The 10 most common isotopes in the solar system are: H >> 4 He >> 16 O > 12 C >> 20 Ne > 14 N > 24 Mg > 28 Si > 56 Fe > 32 S The isotopes we study occur throughout the solar system and are important in lots of processes!
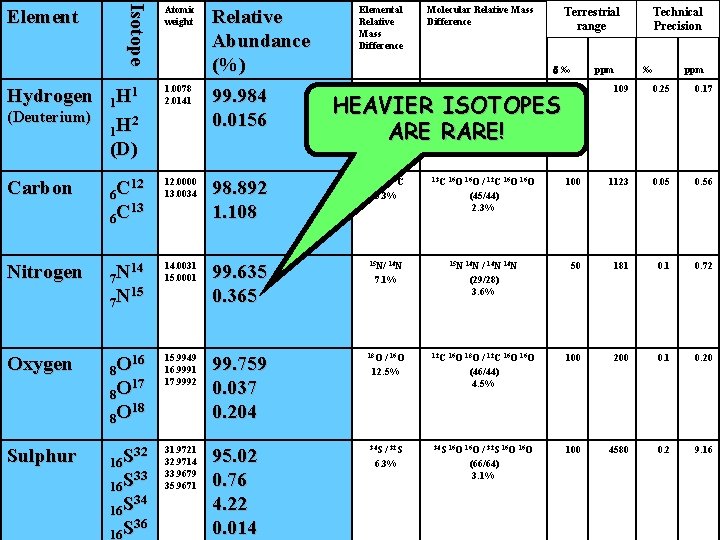
Isotope Element Atomic weight Relative Abundance (%) Elemental Relative Mass Difference 1. 0078 2. 0141 99. 984 0. 0156 Carbon 12 6 C 13 6 C 12. 0000 13. 0034 98. 892 1. 108 13 C Nitrogen 14 7 N 15 7 N 14. 0031 15. 0001 Oxygen 16 8 O 17 8 O 18 8 O Sulphur 32 16 S 33 16 S 34 16 S 36 16 S (Deuterium) Terrestrial range d‰ 1 1 H 2 1 H (D) Hydrogen Molecular Relative Mass Difference D/H 100% / 1 H 1 H (3/2) 50% ppm Technical Precision ‰ ppm 1 HD 700 109 0. 25 0. 17 / 12 C 8. 3% 13 C 16 O 100 1123 0. 05 0. 56 99. 635 0. 365 15 N/ 14 N 15 N 14 N 50 181 0. 72 15. 9949 16. 9991 17. 9992 99. 759 0. 037 0. 204 18 O 12 C 16 O 18 O 100 200 0. 1 0. 20 31. 9721 32. 9714 33. 9679 35. 9671 95. 02 0. 76 4. 22 0. 014 34 S 16 O 100 4580 0. 2 9. 16 HEAVIER ISOTOPES ARE RARE! 7. 1% / 16 O 12. 5% / 32 S 6. 3% / 12 C 16 O (45/44) 2. 3% / 14 N (29/28) 3. 6% / 12 C 16 O (46/44) 4. 5% / 32 S 16 O (66/64) 3. 1%
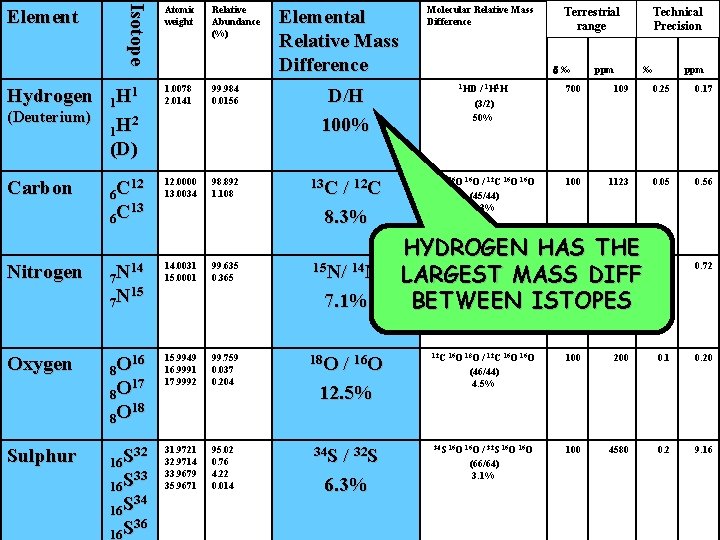
Hydrogen (Deuterium) Carbon Isotope Element Atomic weight Relative Abundance (%) Elemental Relative Mass Difference Molecular Relative Mass Difference Terrestrial range d‰ / 1 H 1 H (3/2) 50% ppm 1 1 H 2 1 H (D) 1. 0078 2. 0141 99. 984 0. 0156 12 6 C 13 6 C 12. 0000 13. 0034 98. 892 1. 108 13 C HYDROGEN HAS THE N N/ N N 50 181 15 N/ 14 N LARGEST (29/28) MASS DIFF 3. 6% 7. 1% BETWEEN ISTOPES ‰ ppm 1 HD 700 109 0. 25 0. 17 13 C 16 O 100 1123 0. 05 0. 56 0. 1 0. 72 D/H 100% / 12 C 8. 3% Technical Precision / 12 C 16 O (45/44) 2. 3% Nitrogen 14 7 N 15 7 N 14. 0031 15. 0001 99. 635 0. 365 Oxygen 16 8 O 17 8 O 18 8 O 15. 9949 16. 9991 17. 9992 99. 759 0. 037 0. 204 18 O 12 C 16 O 18 O 100 200 0. 1 0. 20 Sulphur 32 16 S 33 16 S 34 16 S 36 16 S 31. 9721 32. 9714 33. 9679 35. 9671 95. 02 0. 76 4. 22 0. 014 34 S 16 O 100 4580 0. 2 9. 16 15 / 16 O 12. 5% / 32 S 6. 3% 14 14 14 / 12 C 16 O (46/44) 4. 5% / 32 S 16 O (66/64) 3. 1%
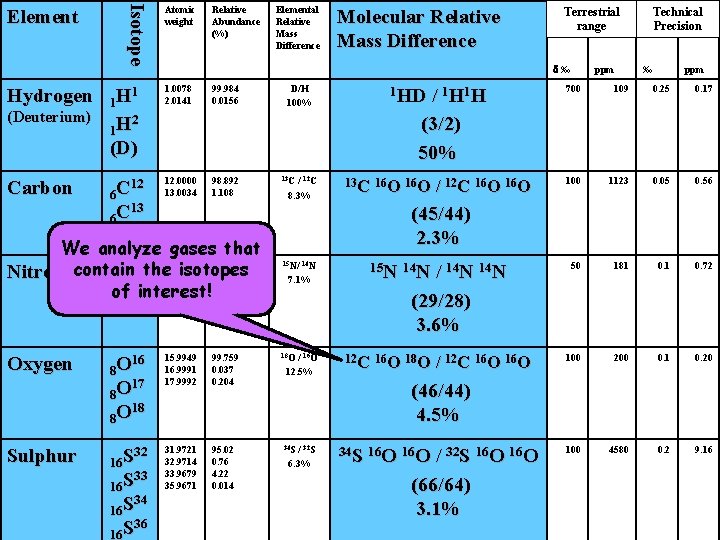
Isotope Element Hydrogen (Deuterium) Carbon Atomic weight Relative Abundance (%) Elemental Relative Mass Difference 1 1 H 2 1 H (D) 1. 0078 2. 0141 99. 984 0. 0156 12 6 C 13 6 C 12. 0000 13. 0034 98. 892 1. 108 D/H 100% 13 C / 12 C 8. 3% 16 8 O 17 8 O 18 8 O 15. 9949 16. 9991 17. 9992 99. 759 0. 037 0. 204 32 16 S 33 16 S 34 16 S 36 16 S 31. 9721 32. 9714 33. 9679 35. 9671 95. 02 0. 76 4. 22 0. 014 1 HD / 1 H (3/2) 50% 13 C 16 O / 12 C 16 O ppm Technical Precision ‰ ppm 700 109 0. 25 0. 17 100 1123 0. 05 0. 56 50 181 0. 72 100 200 0. 1 0. 20 100 4580 0. 2 9. 16 (45/44) 2. 3% 15 N/ 14 N 7. 1% 7 Sulphur Terrestrial range d‰ We analyze gases that 14. 0031 99. 635 14 the isotopes Nitrogencontain 15. 0001 0. 365 7 N of N 15 interest! Oxygen Molecular Relative Mass Difference 18 O / 16 O 12. 5% 15 N 14 N / 14 N (29/28) 3. 6% 12 C 16 O 18 O / 12 C 16 O (46/44) 4. 5% 34 S / 32 S 6. 3% 34 S 16 O / 32 S 16 O (66/64) 3. 1%

Isotope Element Atomic weight Relative Abundance (%) Elemental Relative Mass Difference Molecular Relative Mass Difference HYDROGEN HAS A LARGE TERRESTRIAL RANGE, BUT 1. 0078 99. 984 D/H 1 Hydrogen H 2. 0141 0. 0156 100% 1 RELATIVELY LOW ALSO (Deuterium) 2 1 H PRECISION Terrestrial range d‰ / 1 H 1 H (3/2) 50% Technical Precision ppm ‰ ppm 1 HD 700 109 0. 25+ 0. 17 (D) Carbon 12 6 C 13 6 C 12. 0000 13. 0034 98. 892 1. 108 13 C / 12 C 8. 3% 13 C 16 O 100 1123 0. 05 0. 56 Nitrogen 14 7 N 15 7 N 14. 0031 15. 0001 99. 635 0. 365 15 N/ 14 N 15 N 14 N 50 181 0. 72 16 O 100 200 0. 1 0. 20 / 32 S 16 O (66/64) 3. 1% 100 4580 0. 2 9. 16 Oxygen Sulphur / 12 C 16 O (45/44) 2. 3% / 14 N (29/28) 3. 6% 7. 1% NITROGEN HAS A SMALLER O/ O C O O/ C O 16 15. 9949 99. 759 O 16. 9991 0. 037 12. 5% (46/44) 8 TERRESTRIAL RANGE, BUT 17. 9992 0. 204 4. 5% 17 8 O 18 BETTER TECHNICAL 8 O PRECISION 18 32 16 S 33 16 S 34 16 S 36 16 S 31. 9721 32. 9714 33. 9679 35. 9671 95. 02 0. 76 4. 22 0. 014 16 34 S / 32 S 6. 3% 12 16 18 34 S 16 O 12 16

Expressing differences in stable isotope abundance
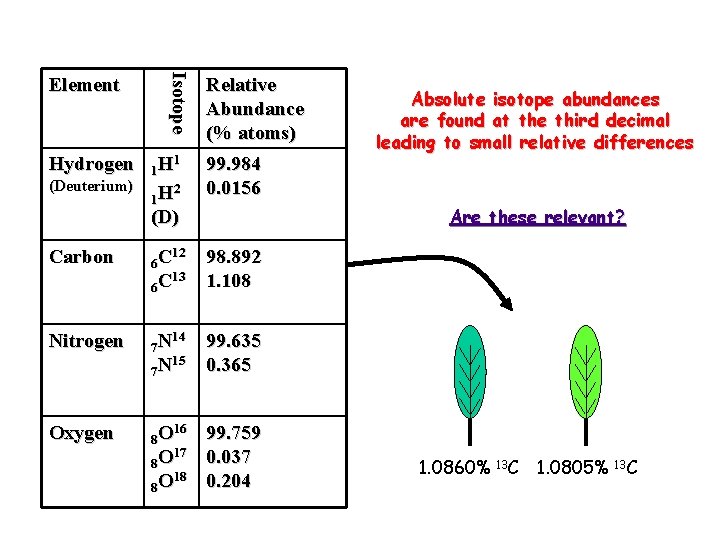
Hydrogen Isotope Element Relative Abundance (% atoms) 1 1 H 2 1 H 99. 984 0. 0156 Carbon 12 6 C 13 6 C 98. 892 1. 108 Nitrogen 14 7 N 15 7 N 99. 635 0. 365 Oxygen 16 8 O 17 8 O 18 8 O 99. 759 0. 037 0. 204 (Deuterium) Absolute isotope abundances are found at the third decimal leading to small relative differences Are these relevant? (D) 1. 0860% 13 C 1. 0805% 13 C
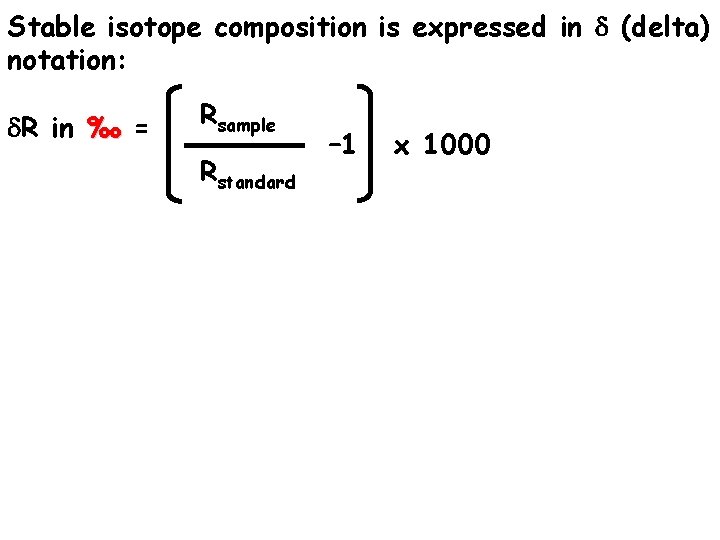
Stable isotope composition is expressed in d (delta) notation: d. R in ‰ = Rsample Rstandard – 1 x 1000
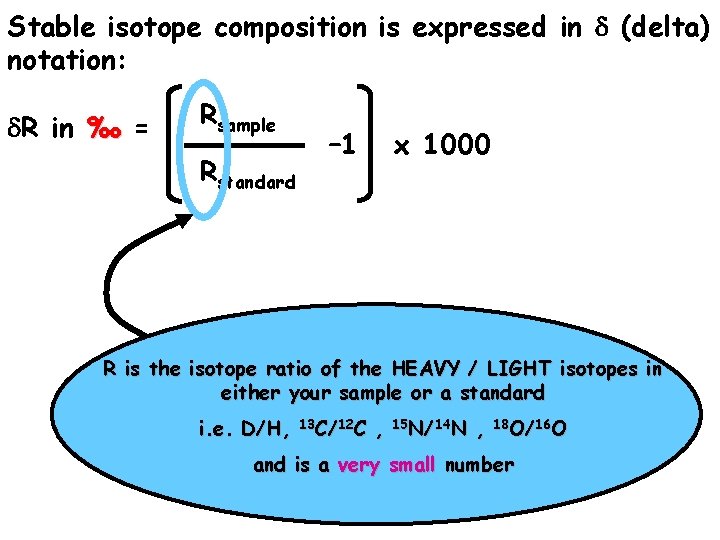
Stable isotope composition is expressed in d (delta) notation: d. R in ‰ = Rsample Rstandard – 1 x 1000 R is the isotope ratio of the HEAVY / LIGHT isotopes in either your sample or a standard i. e. D/H, 13 C/12 C , 15 N/14 N , 18 O/16 O and is a very small number
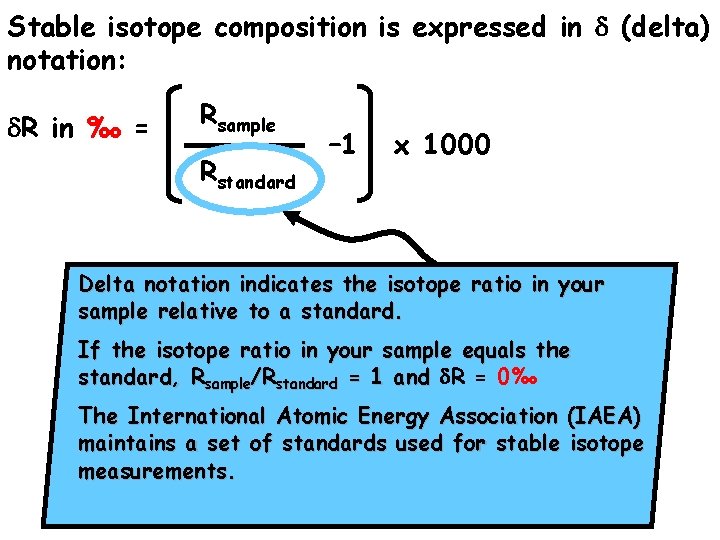
Stable isotope composition is expressed in d (delta) notation: d. R in ‰ = Rsample Rstandard – 1 x 1000 Delta notation indicates the isotope ratio in your sample relative to a standard. If the isotope ratio in your sample equals the standard, Rsample/Rstandard = 1 and d. R = 0‰ The International Atomic Energy Association (IAEA) maintains a set of standards used for stable isotope measurements.
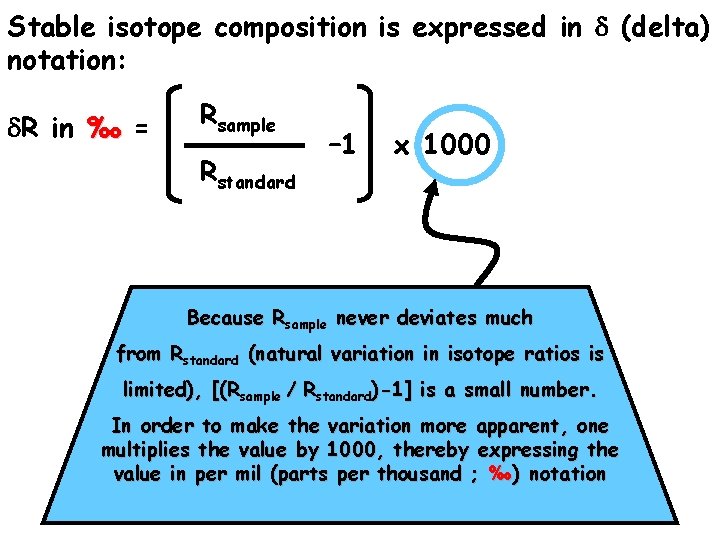
Stable isotope composition is expressed in d (delta) notation: d. R in ‰ = Rsample Rstandard – 1 x 1000 Because Rsample never deviates much from Rstandard (natural variation in isotope ratios is limited), [(Rsample / Rstandard)-1] is a small number. In order to make the variation more apparent, one multiplies the value by 1000, thereby expressing the value in per mil (parts per thousand ; ‰) notation
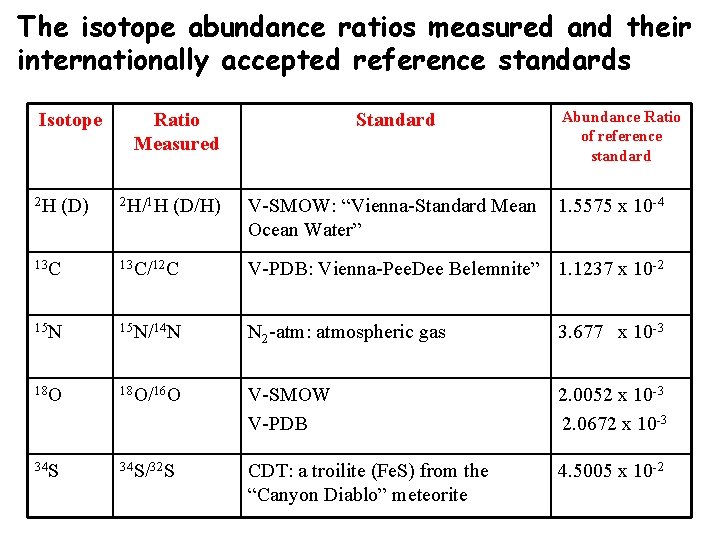
The isotope abundance ratios measured and their internationally accepted reference standards Isotope 2 H (D) Ratio Measured 2 H/1 H (D/H) Standard V-SMOW: “Vienna-Standard Mean Ocean Water” Abundance Ratio of reference standard 1. 5575 x 10 -4 13 C/12 C V-PDB: Vienna-Pee. Dee Belemnite” 1. 1237 x 10 -2 15 N/14 N N 2 -atm: atmospheric gas 3. 677 x 10 -3 18 O/16 O V-SMOW V-PDB 2. 0052 x 10 -3 2. 0672 x 10 -3 34 S/32 S CDT: a troilite (Fe. S) from the “Canyon Diablo” meteorite 4. 5005 x 10 -2
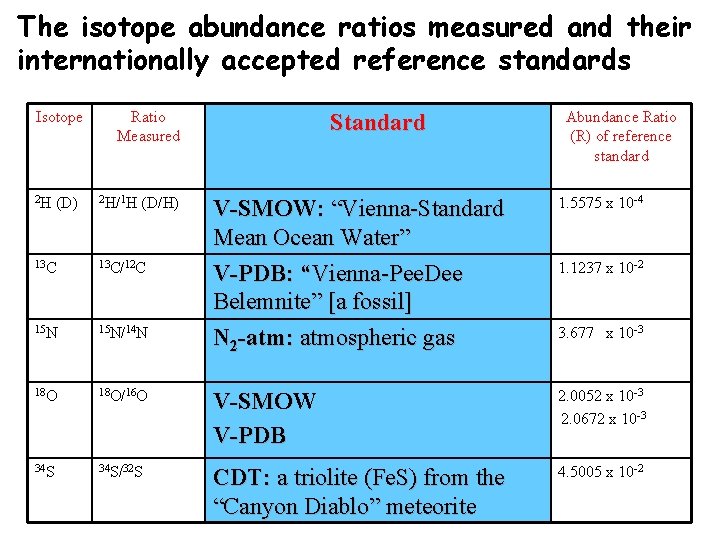
The isotope abundance ratios measured and their internationally accepted reference standards Isotope 2 H (D) Ratio Measured 2 H/1 H (D/H) Standard Abundance Ratio (R) of reference standard V-SMOW: “Vienna-Standard Mean Ocean Water” V-PDB: “Vienna-Pee. Dee Belemnite” [a fossil] N 2 -atm: atmospheric gas 1. 5575 x 10 -4 13 C/12 C 1. 1237 x 10 -2 15 N/14 N 18 O/16 O V-SMOW V-PDB 2. 0052 x 10 -3 2. 0672 x 10 -3 34 S/32 S CDT: a triolite (Fe. S) from the “Canyon Diablo” meteorite 4. 5005 x 10 -2 3. 677 x 10 -3

The isotope abundance ratios measured and their internationally accepted reference standards Isotope 2 H (D) Ratio Measured 2 H/1 H (D/H) Standard V-SMOW Abundance Ratio of reference standard (w/ 95% CI) 1. 5575 x 10 -4 ±. 001 13 C/12 C V-PDB 1. 1237 x 10 -2 ±. 0009 15 N/14 N N 2 -atm 3. 677 x 10 -3 ±. 00081 18 O/16 O V-SMOW V-PDB 2. 0052 x 10 -3 ±. 00043 2. 0672 x 10 -3 ±. 0021 17 O/16 O V-SMOW V-PDB 34 S/32 S CDT 373 x 10 -6 ± 15 379 x 10 -6 ± 15 4. 5005 x 10 -2 These values are the ratios of atoms in the standards and reflect the very low abundance of the heavier isotope

Standards Some other international standards of known d value: Standard Light Antarctic Precipitation (SLAP) with values: d. D = -428‰ d 18 O = -55. 5‰ Greenland Icesheet Precipitation (GISP) with values: d. D = -189. 7‰ d 18 O = -24. 8‰

Working standards The internationally accepted reference standards are obviously in limited supply, expensive, and cannot be used as the daily reference standard in labs around the world. Instead isotope labs employ WORKING STANDARDS. Working standards are: used on a regular (daily) basis homogeneous well matched to your analyses easily obtained or made easily corrected back to the international standards
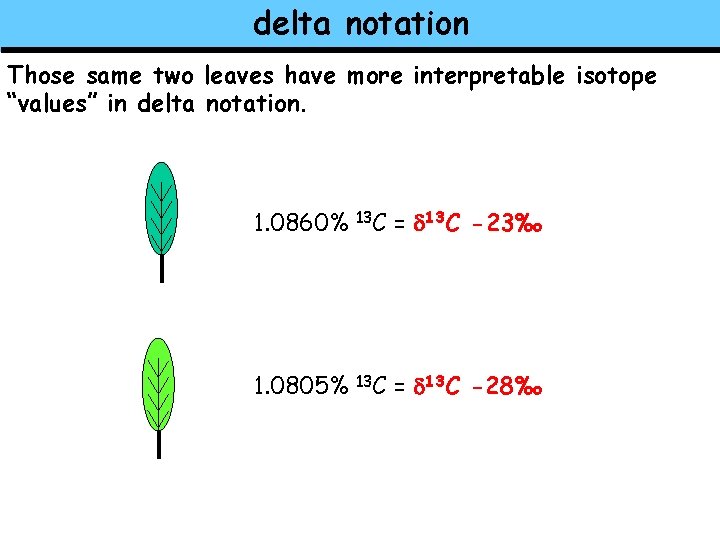
delta notation Those same two leaves have more interpretable isotope “values” in delta notation. 1. 0860% 13 C = d 13 C -23‰ 1. 0805% 13 C = d 13 C -28‰

delta notation A quick note, to be elaborated on in lecture 5: Ø You can’t do chemical calculations with d units. Ø They are just for comparative purposes. Ø That said, for small ranges of d units “you are allowed” to be sloppy.

Atom % notation Xheavy Atom % X= heavy + Xlight Xheavy • 100 = Xtotal • 100 Where X is the FRACTION of the heavy or light isotope in a mixture. Unlike delta notation, atom % notation does not accentuate small changes in isotope abundance. You will NOT see this notation used in the NATURAL ABUNDANCE stable isotope literature You WILL see this notation used if you are working with ENRICHED stable isotope methods

Using and referring to the delta values

Some comparative terms: LIGHT vs. HEAVY SAMPLES a LIGHTER sample contains more of the lighter isotope, relative to another sample a HEAVIER samples contains more of the heavier isotope, relative to another sample DEPLETED vs. ENRICHED SAMPLES a sample “DEPLETED” IN THE HEAVY ISOTOPE contains less of the heavy isotope and more of the light isotope, relative to another sample a sample “ENRICHED” IN THE LIGHT ISOTOPE contains more of the light isotope and less of the heavy isotope, relative to another sample
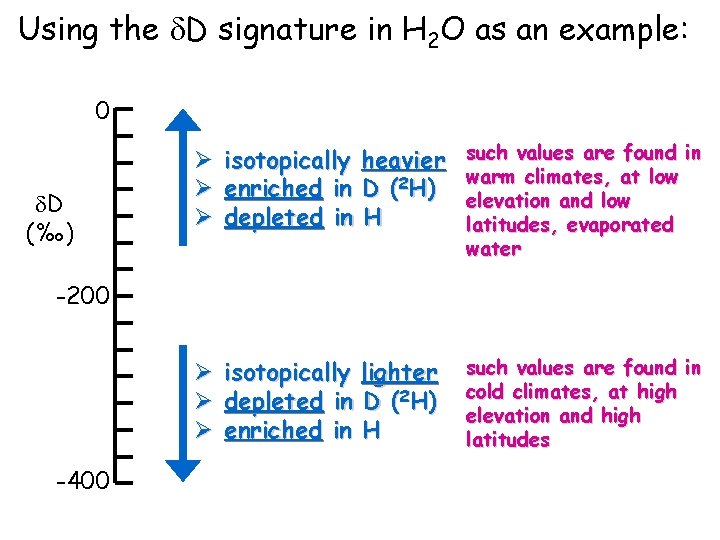
Using the d. D signature in H 2 O as an example: 0 d. D (‰) Ø Ø Ø isotopically heavier enriched in D (2 H) depleted in H such values are found in warm climates, at low elevation and low latitudes, evaporated water -200 Ø isotopically lighter Ø depleted in D (2 H) Ø enriched in H -400 such values are found in cold climates, at high elevation and high latitudes

Some causes of variation in stable isotope values

Isotope Effects Urey’s Axiom: “The heavy isotopes concentrate in the compound in which the element is most strongly held” If Urey is correct then this implies that issues such as: Bond-strength, Mass of an element, isotope or atom, Rates of a chemical reaction (chemical behavior), System properties (open vs. closed), Etc. ……. Could all have effects on isotope distributions in the materials we measure and help explain the variation in stable isotope composition (e. g. , variation in d).

Key points about isotopes 1. Chemical properties of any element are largely determined by the number and configuration of electrons (e-) Ø Since isotopes have the same number and configuration of electrons. . . isotopes have the same chemical properties 13 CO 2 is chemically identical to 12 CO 2

Key points about isotopes 1. isotopes have the same chemical properties 2. However, isotopes differ in then number of neutrons, N they possess, and therefore in mass How do mass differences lead to variation is isotope abundance? Mass differences influence chemical BEHAVIOR in reactions or mixtures

Isotope mass effects Differences in mass influence: 1. The RATES at which the isotopes react Lighter isotopes react faster. Therefore different isotopes involved in a chemical reaction display differential representation in different phases of the reaction 2. The PHYSIO-CHEMICAL properties of molecules composed of different isotopes That is, factors including vapor pressure, boiling temperature, freezing point, and melting point are affected by the isotope composition of a molecule. As we’ll see next, for water composed of different isotopes this has a large, measurable, and significant influence.
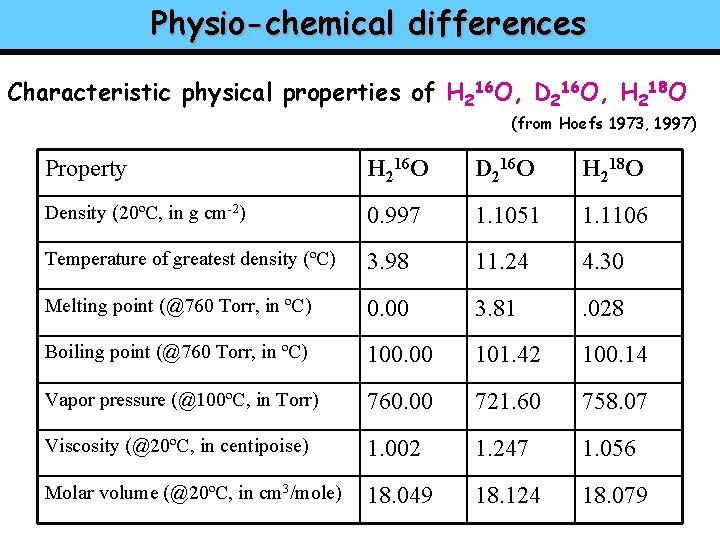
Physio-chemical differences Characteristic physical properties of H 216 O, D 216 O, H 218 O (from Hoefs 1973, 1997) Property H 216 O D 216 O H 218 O Density (20ºC, in g cm-2) 0. 997 1. 1051 1. 1106 Temperature of greatest density (ºC) 3. 98 11. 24 4. 30 Melting point (@760 Torr, in ºC) 0. 00 3. 81 . 028 Boiling point (@760 Torr, in ºC) 100. 00 101. 42 100. 14 Vapor pressure (@100ºC, in Torr) 760. 00 721. 60 758. 07 Viscosity (@20ºC, in centipoise) 1. 002 1. 247 1. 056 Molar volume (@20ºC, in cm 3/mole) 18. 049 18. 124 18. 079
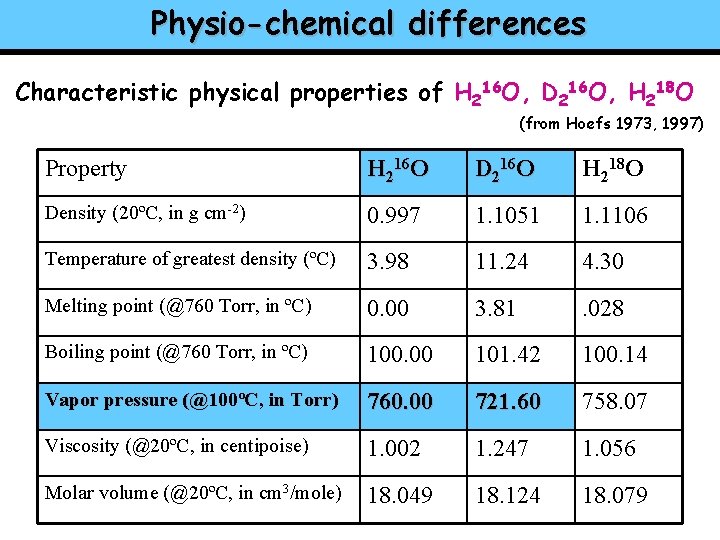
Physio-chemical differences Characteristic physical properties of H 216 O, D 216 O, H 218 O (from Hoefs 1973, 1997) Property H 216 O D 216 O H 218 O Density (20ºC, in g cm-2) 0. 997 1. 1051 1. 1106 Temperature of greatest density (ºC) 3. 98 11. 24 4. 30 Melting point (@760 Torr, in ºC) 0. 00 3. 81 . 028 Boiling point (@760 Torr, in ºC) 100. 00 101. 42 100. 14 Vapor pressure (@100ºC, in Torr) 760. 00 721. 60 758. 07 Viscosity (@20ºC, in centipoise) 1. 002 1. 247 1. 056 Molar volume (@20ºC, in cm 3/mole) 18. 049 18. 124 18. 079
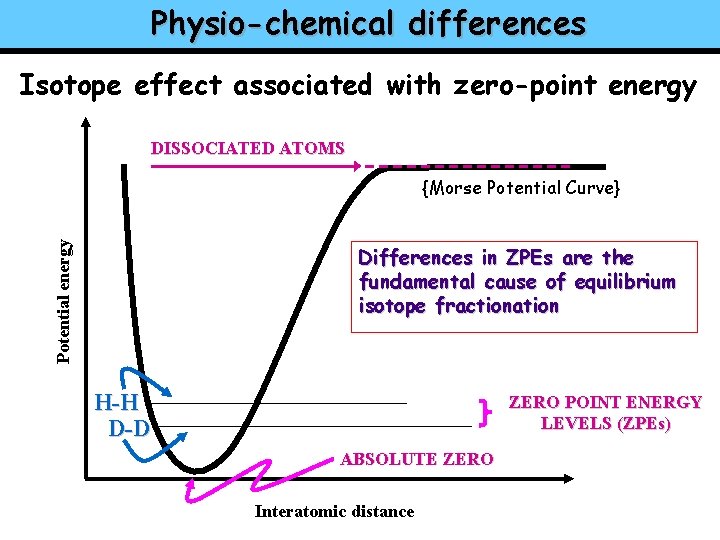
Physio-chemical differences Isotope effect associated with zero-point energy DISSOCIATED ATOMS Potential energy {Morse Potential Curve} Differences in ZPEs are the fundamental cause of equilibrium isotope fractionation H-H D-D ZERO POINT ENERGY LEVELS (ZPEs) ABSOLUTE ZERO Interatomic distance
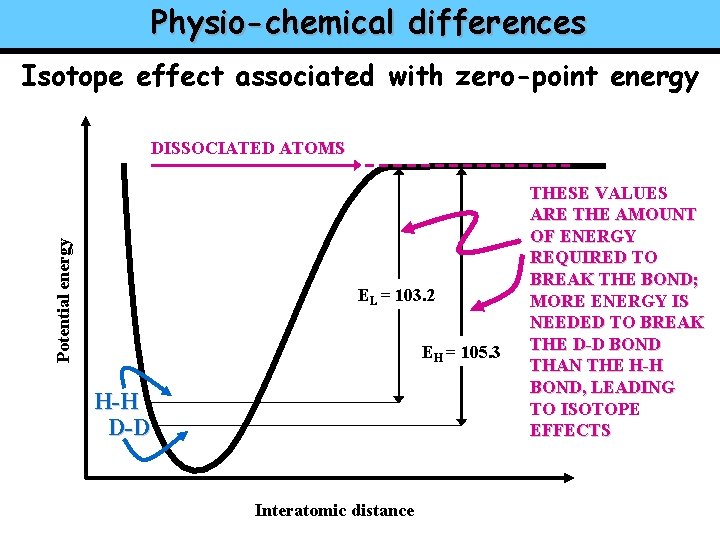
Physio-chemical differences Isotope effect associated with zero-point energy Potential energy DISSOCIATED ATOMS EL = 103. 2 EH = 105. 3 H-H D-D Interatomic distance THESE VALUES ARE THE AMOUNT OF ENERGY REQUIRED TO BREAK THE BOND; MORE ENERGY IS NEEDED TO BREAK THE D-D BOND THAN THE H-H BOND, LEADING TO ISOTOPE EFFECTS

Physio-chemical differences In summary: Higher vibrational frequency, the stretching and compressing of chemical bonds between atoms, leads to a higher zero point energy and lower stability Bond strengths are proportional to isotope mass, so an isotope with a higher mass has a higher bond strength Molecules with heavier isotopes will be more stable than light isotopes but diffuse more slowly Therefore, partial vaporization of a liquid pool will lead to increased concentration of the lighter isotope in the vapor phase

Reaction rate differences Differences in mass also influence the RATES at which the isotopes react The lighter isotope reacts at a faster rate, leading to a heavier d value in the remaining substrate relative to the product. -- Therefore, differences in MASS influence RATES and lead to ISOTOPE FRACTIONATION

Fractionation Both the differences in physiochemical properties and reaction rates lead to the REDISTRIBUTION of ISOTOPES This process is known as FRACTIONATION Fractionation can be caused by either BI-DIRECTIONAL or UNIDIRECTIONAL reactions
![Types of fractionation 1. Exchange/equilibrium [BI-directional] • Complete back-reaction • Product/reactant offset by constant Types of fractionation 1. Exchange/equilibrium [BI-directional] • Complete back-reaction • Product/reactant offset by constant](http://slidetodoc.com/presentation_image_h/8a96a20ed3bf8f923cfce73a50ec9588/image-52.jpg)
Types of fractionation 1. Exchange/equilibrium [BI-directional] • Complete back-reaction • Product/reactant offset by constant fractionation factor 2. Kinetic [UNI-directional] = “biological” • Incomplete back-reaction (extreme case. Rayleigh distillation) • Product and reactant d can evolve in concert (closed system), or product composition determines reactant composition (open system) 3. Transport/Diffusion [UNI-directional] • Subset of kinetic fractionation reactions involving flux along a concentration gradient

Fractionation BI-DIRECTIONAL REACTIONS: Known as EQUILIBRIUM FRACTIONATION Differences in physio-chemical properties and sometimes reaction rates (very fast) allow bi-directional exchange of isotopes In such a reaction the difference in d value between the two pools REMAINS CONSTANT when there is CONTINUOUS EXCHANGE between the substrate and the product

Fractionation An example on an EQUILIBRIUM REACTION: CO 2 + H 2 O H 2 CO 3 Or alternatively: CO 2 H 2 O since we are interested in the exchange of oxygen atoms between CO 2 and H 2 O Initially the H 2 O and CO 2 have different isotope compositions, but as they exchange 18 O and 16 O’s back and forth they reach an equilibrium. If fractionation occurs that the concentration of each isotope species will not be the same in both the H 2 O and CO 2 pools [in this case, if the initial d 18 O of H 2 O is – 12. 95‰ the CO 2’s d 18 O – 12. 95 will be 28. 83‰ - we’ll see this next] 28. 83

Fractionation UNI-DIRECTIONAL REACTIONS: Known as KINETIC FRACTIONATION Differences in physio-chemical properties and reaction rates never result in uni-directional exchange of isotopes (there is no back-reaction) Referred to as DISCRIMINATION if it is biologically (enzyme mediated) fractionation

Attaching a number to “fractionation”

Fractionation Factors The d values of the substrate and the product are related to one another through a: FRACTIONATION FACTOR, a a defines the relationship between the substrate (A) and product (B) in either an equilibrium or kinetic reaction such that, a. AB= RA / RB [R is the isotope ratio]

Fractionation Factors If a = 1, no fractionation is occurring If a > 1, there is more of the heavier isotope in the substrate than before the reaction began If a < 1, there is more of the lighter isotope in the substrate than before the reaction began

Fractionation Factors How to relate a and d values: We know: a. AB = RA/RB In addition: a. AB = (1000 + d. A) / (1000 + d. B) And since: d. A = ((RA/RS) – 1) • 1000 Derivation: Actual value a. AB = [((RA/RS) – 1) • 1000] + 1000 [((RB/RS) – 1) • 1000] + 1000 = [(1000 • RA / 1000 • RS) – 1000] + 1000 [(1000 • RB / 1000 • RS) – 1000] + 1000 = (1000 • RA / 1000 • RS) (1000 • RB / 1000 • RS) = RA / RB = (1000 + d. A) / (1000 + d. B)

Fractionation Factors In other words you can calculate a using: a. AB = RA/RB -ora. AB = (1000 + d. A) / (1000 + d. B)

Fractionation Factors With an equilibrium equation, a is really an EQUILIBRIUM CONSTANT - at equilibrium, it will tell you the distribution of isotopes between two species. With kinetic fractionation it is the same idea, except a. AB = k 1 / k 2, where k 1 and k 2 are the RATE CONSTANTS for the two isotopic species In a multi-step process, equilibrium fractionations are ADDITIVE In a multi-step process, kinetic fractionations are NOT ADDITIVE

Fractionation Adding isotope stoichiometry to our equilibrium reaction example, we get: (CO 16 O)gas + (H 218 O)liquid <=> (CO 18 O)gas + (H 216 O)liquid this is the equilibrium equation for each atom you need to empirically determine the quantities of each species once you have these numbers, you can calculate the fractionation factor, because: K = ((CO 18 O)g • (H 216 O)l) / ((CO 16 O)g • (H 218 O)l) = (18 O/16 O)g/(18 O/16 O)l = R A / RB or a = RA / R B

Fractionation Factors… What is the a-constant in our equilibrium example? H 2 O CO 2 where: d 18 OH 2 O = -12. 95‰ -12. 95 d 18 OCO 2 = 28. 83‰ therefore: RH 2 O = 0. 001979 RCO 2 = 0. 002063 (18 O/16 O)water (18 O/16 O)carbon dioxide = RA RB = 0. 001979 0. 002063 = 0. 95939
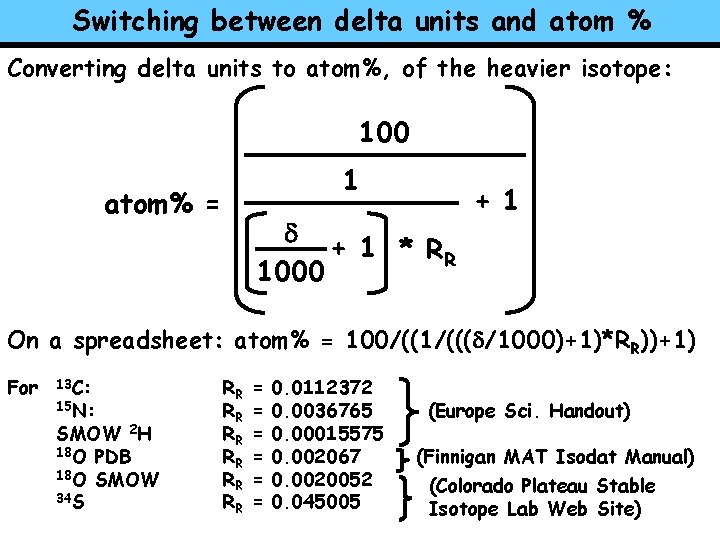
Switching between delta units and atom % Converting delta units to atom%, of the heavier isotope: 100 1 atom% = d + 1 * RR 1000 + 1 On a spreadsheet: atom% = 100/((1/(((d/1000)+1)*RR))+1) For 13 C: 15 N: SMOW 2 H 18 O PDB 18 O SMOW 34 S RR RR RR = = = 0. 0112372 0. 0036765 0. 00015575 0. 002067 0. 0020052 0. 045005 (Europe Sci. Handout) (Finnigan MAT Isodat Manual) (Colorado Plateau Stable Isotope Lab Web Site)

Calculating RA from atom% You’ve now calculated Atom% 16 O RA = Atom% = 1 – Atom% 18 O / Atom% 18 O 16 O 18 O

Fractionation Factors In our kinetic example: CO 2 + H 2 O carbohydrate What is the a-constant of the enzyme? If the CO 2 being used has a value of – 12. 4‰ and – 12. 4 the carbohydrates have a value of -28‰, we can calculate that they have atomic ratios of 0. 01110 and 0. 01092 respectively. Next: (13 C/12 C)carbon dioxide (13 C/12 C)carbohydrate RA 0. 01110 = 1. 01605 = = RB 0. 01092

More fractionation terminology There are many other terms which tell you the ‘per mil’ difference between compound A and compound B. Some are used in the biological literature and others in the geological literature. Although the numbers they yield are not identical, they are close approximations of one another. When you work up numbers it is very important that you indicate which calculation you are using.

More fractionation terminology By definition: DAB = d. A - d. B and you should therefore use the previous equations to obtain d. A and d. B from a to calculate DAB

More fractionation terminology By definition: DAB = d. A - d. B and you should therefore, use the previous equations to obtain d. A and d. B to calculate DAB But you will also see a jumble of other calculations, like: Isotopic enrichment a. In the geological literature: e. AB = (a. AB – 1) • 1000 b. In the biological literature: e. AB = 103 lna. AB Isotopic separation (big Delta, D) a. In the geological literature: DAB = 103 lna. AB da – db Isotope discrimination a. Used in the biological literature and refers specifically to enzymemediated fractionation where A is the source and B is the product b. DAB = (a. AB – 1) • 1000 It is much better to use the (a. AB – 1) • 1000 calculation. There is no mathematical reason to use an equation with “ln”

More fractionation terminology Just to compare some D numbers: a. AB (a. AB– 1) • 1000*lna. AB d. A - d. B Equilibrium 0. 95939 example - 42. 33 - 40. 61 - 41. 77 Kinetic example 16. 05 15. 92 16. 4 1. 01605 It is very important to know what terminology is being used!

TEMPERATURE: A key cause for variation in a

Temperature dependence of a This graph of a values for 18 O and D in water shows that fractionation increases as the temperature decreases!
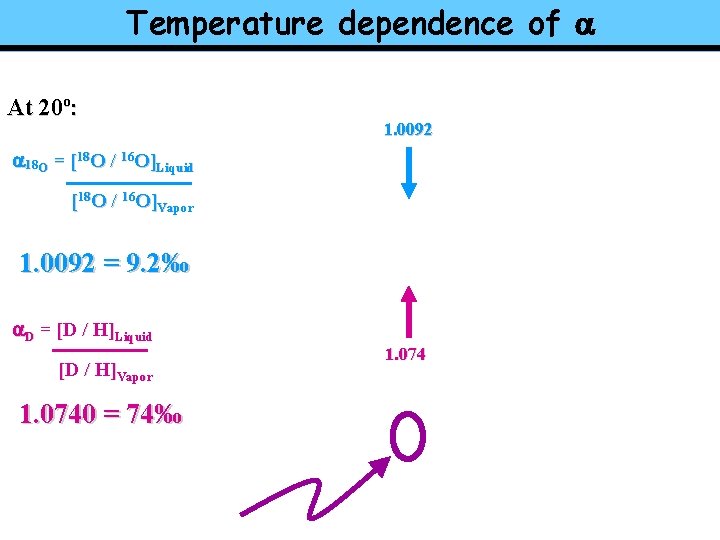
Temperature dependence of a At 20º: 1. 0092 a 18 O = [18 O / 16 O]Liquid [18 O / 16 O]Vapor 1. 0092 = 9. 2‰ a. D = [D / H]Liquid [D / H]Vapor 1. 0740 = 74‰ 1. 074
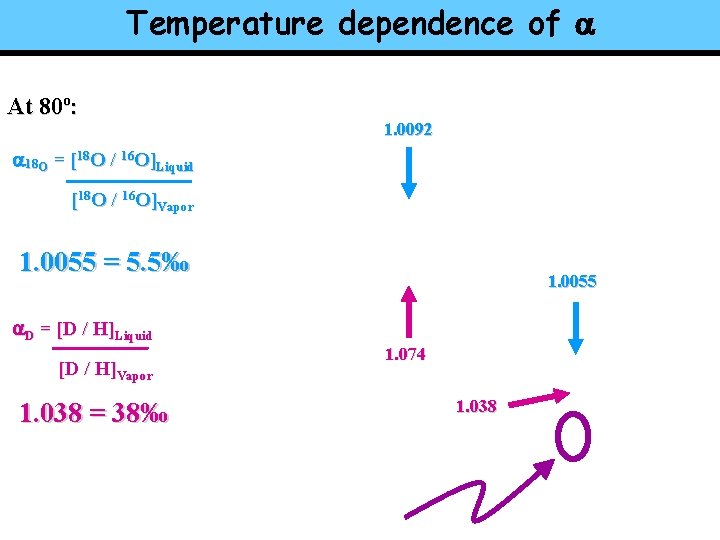
Temperature dependence of a At 80º: 1. 0092 a 18 O = [18 O / 16 O]Liquid [18 O / 16 O]Vapor 1. 0055 = 5. 5‰ 1. 0055 a. D = [D / H]Liquid [D / H]Vapor 1. 038 = 38‰ 1. 074 1. 038

Tying together isotope fractionation concepts: Rayleigh distillation

Rayleigh distillation • Rayleigh fractionation occurs when a parent mass is depleted by equilibrium fractionation to a phase continually removed. The d values of all elements that show mass dependent fractionation are affected; the process occurs in all natural systems. • The equation describing Rayleigh processes is: R 0 f(1 -a) R= R and R 0 are the ratios at t and at t=0 f is the fraction remaining at t a is the fractionation factor “Condensation example”
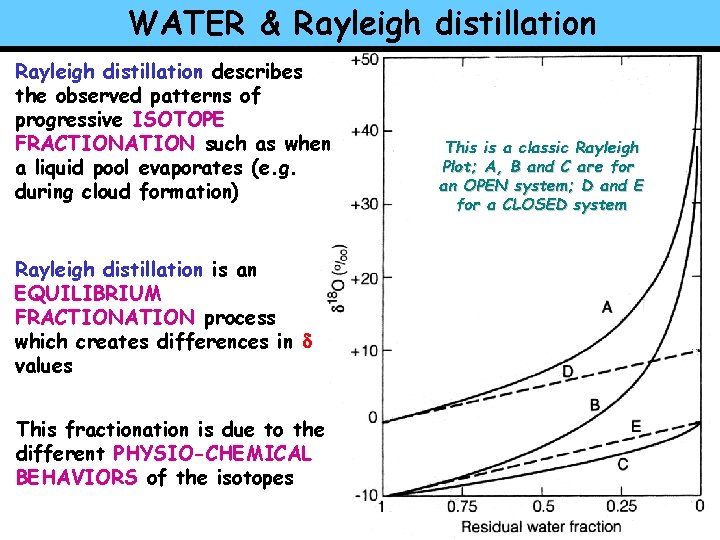
WATER & Rayleigh distillation describes the observed patterns of progressive ISOTOPE FRACTIONATION such as when a liquid pool evaporates (e. g. during cloud formation) Rayleigh distillation is an EQUILIBRIUM FRACTIONATION process which creates differences in d values This fractionation is due to the different PHYSIO-CHEMICAL BEHAVIORS of the isotopes This is a classic Rayleigh Plot; A, B and C are for an OPEN system; D and E for a CLOSED system
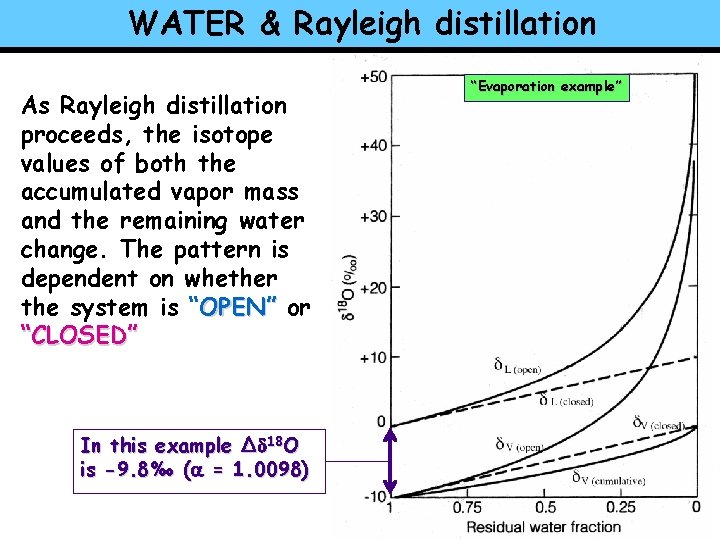
WATER & Rayleigh distillation As Rayleigh distillation proceeds, the isotope values of both the accumulated vapor mass and the remaining water change. The pattern is dependent on whether the system is “OPEN” or “CLOSED” In this example ∆d 18 O is -9. 8‰ (a = 1. 0098) “Evaporation example”
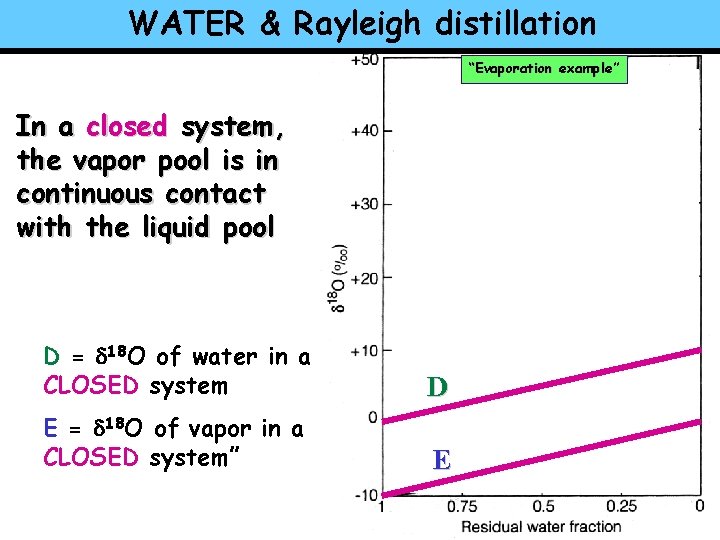
WATER & Rayleigh distillation “Evaporation example” In a closed system, the vapor pool is in continuous contact with the liquid pool D = d 18 O of water in a CLOSED system D E = d 18 O of vapor in a CLOSED system” E
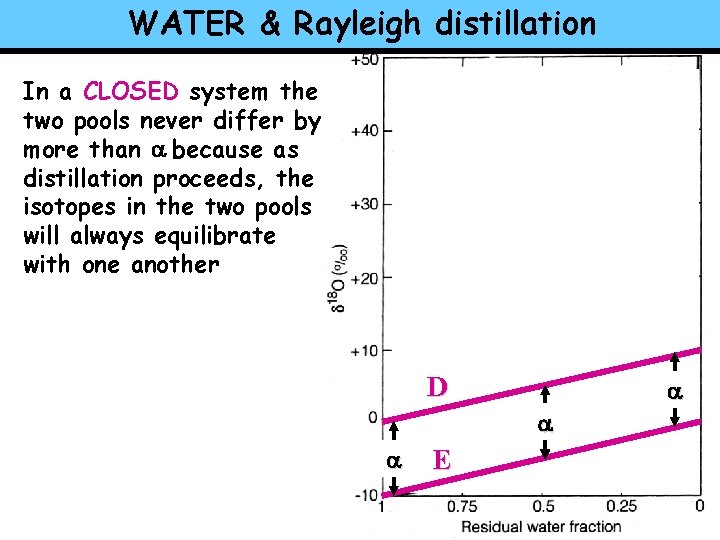
WATER & Rayleigh distillation In a CLOSED system the two pools never differ by more than a because as distillation proceeds, the isotopes in the two pools will always equilibrate with one another D a a a E
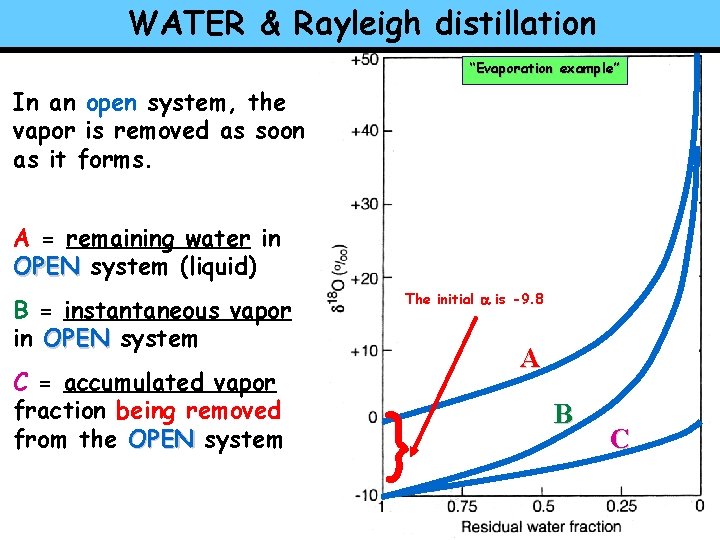
WATER & Rayleigh distillation “Evaporation example” In an open system, the vapor is removed as soon as it forms. A = remaining water in OPEN system (liquid) B = instantaneous vapor in OPEN system C = accumulated vapor fraction being removed from the OPEN system The initial a is -9. 8 A } B C
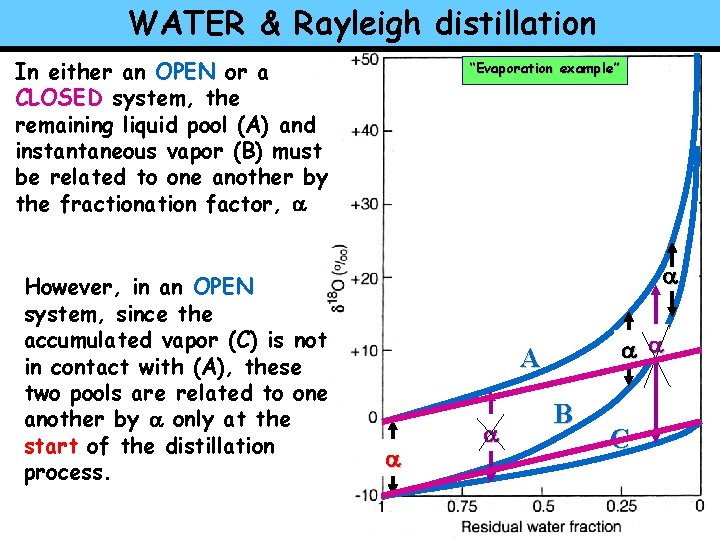
WATER & Rayleigh distillation In either an OPEN or a CLOSED system, the remaining liquid pool (A) and instantaneous vapor (B) must be related to one another by the fractionation factor, a However, in an OPEN system, since the accumulated vapor (C) is not in contact with (A), these two pools are related to one another by a only at the start of the distillation process. “Evaporation example” a a a A a a B C
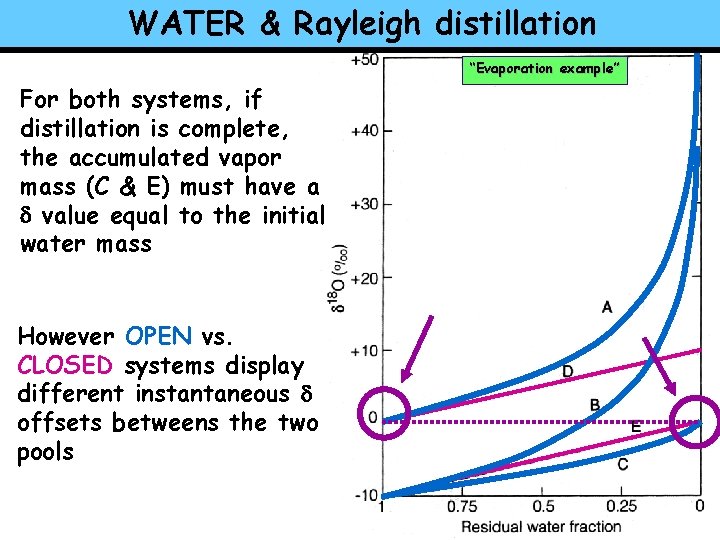
WATER & Rayleigh distillation “Evaporation example” For both systems, if distillation is complete, the accumulated vapor mass (C & E) must have a d value equal to the initial water mass However OPEN vs. CLOSED systems display different instantaneous d offsets betweens the two pools
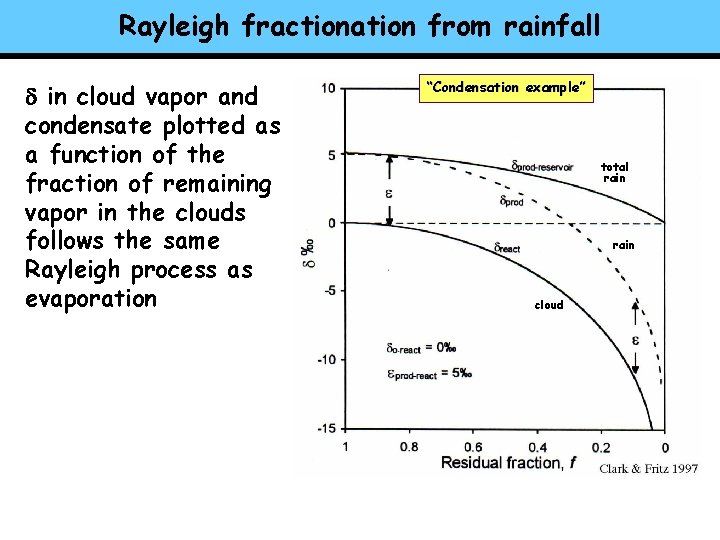
Rayleigh fractionation from rainfall d in cloud vapor and condensate plotted as a function of the fraction of remaining vapor in the clouds follows the same Rayleigh process as evaporation “Condensation example” total rain cloud
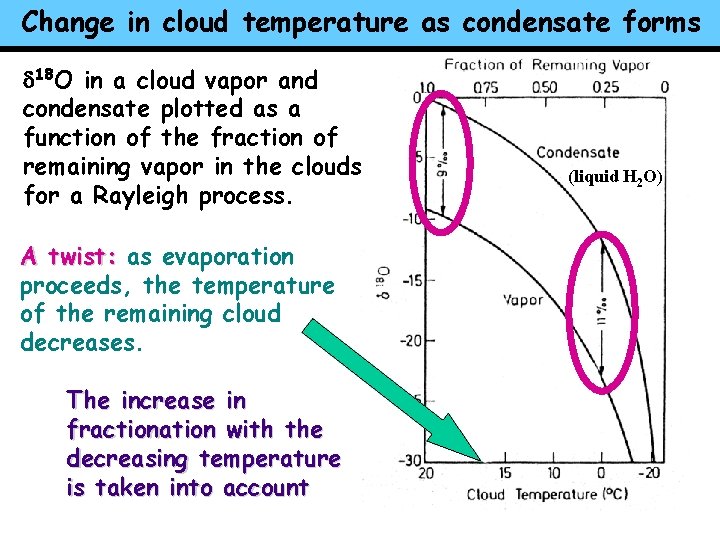
Change in cloud temperature as condensate forms d 18 O in a cloud vapor and condensate plotted as a function of the fraction of remaining vapor in the clouds for a Rayleigh process. A twist: as evaporation proceeds, the temperature of the remaining cloud decreases. The increase in fractionation with the decreasing temperature is taken into account (liquid H 2 O)
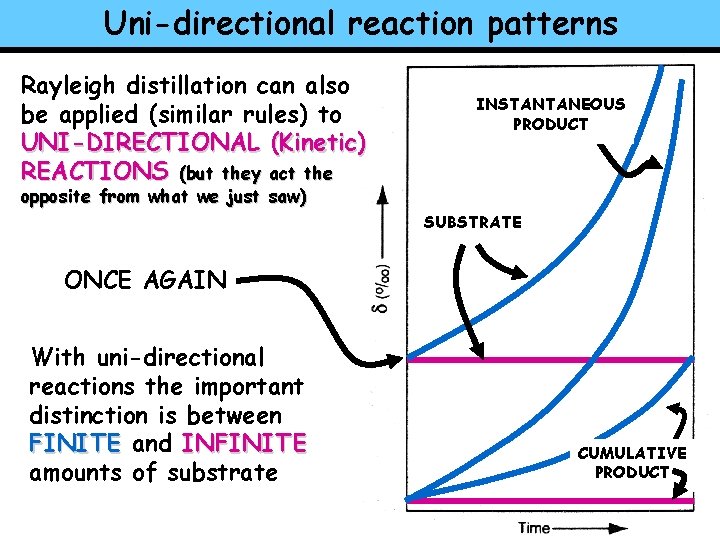
Uni-directional reaction patterns Rayleigh distillation can also be applied (similar rules) to UNI-DIRECTIONAL (Kinetic) REACTIONS (but they act the INSTANTANEOUS PRODUCT opposite from what we just saw) SUBSTRATE ONCE AGAIN With uni-directional reactions the important distinction is between FINITE and INFINITE amounts of substrate CUMULATIVE PRODUCT
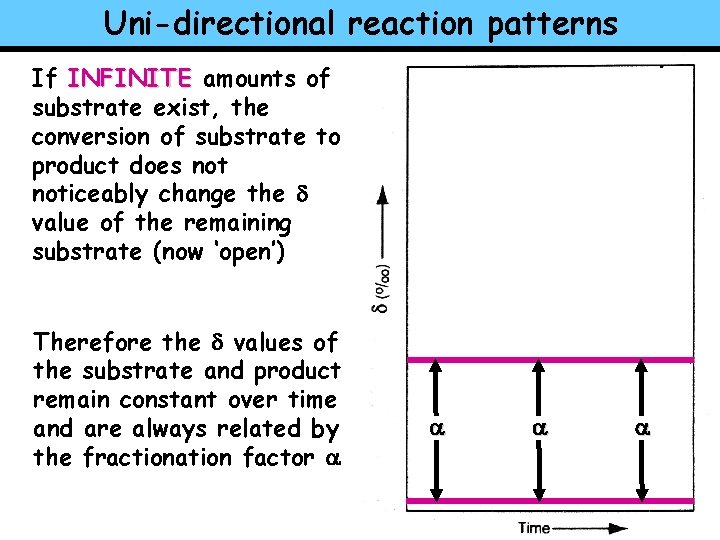
Uni-directional reaction patterns If INFINITE amounts of substrate exist, the conversion of substrate to product does noticeably change the d value of the remaining substrate (now ‘open’) Therefore the d values of the substrate and product remain constant over time and are always related by the fractionation factor a a
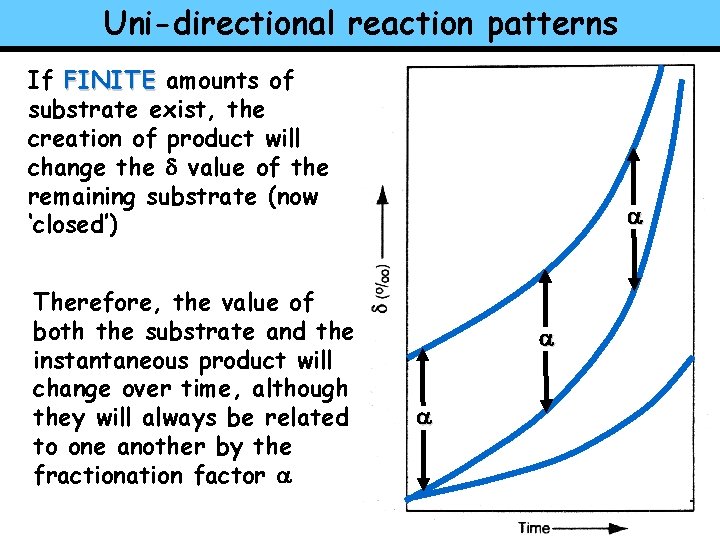
Uni-directional reaction patterns If FINITE amounts of substrate exist, the creation of product will change the d value of the remaining substrate (now ‘closed’) Therefore, the value of both the substrate and the instantaneous product will change over time, although they will always be related to one another by the fractionation factor a a

How can Stable Isotopes be used as a tool? On to some case studies in isotope ecology
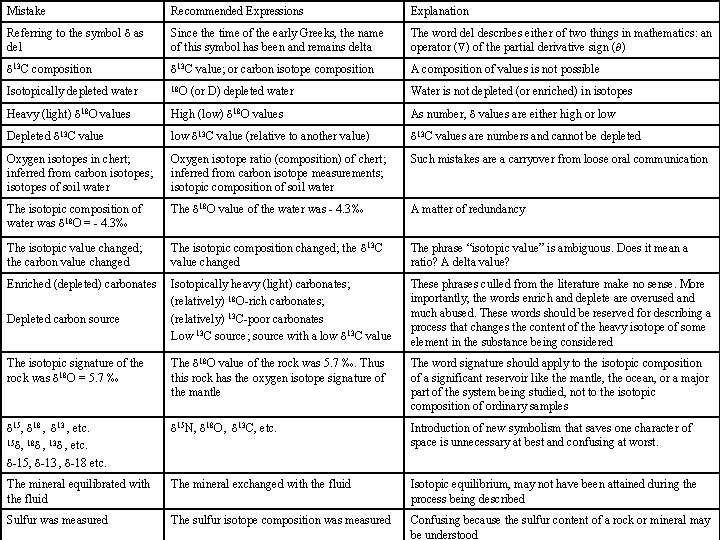
Mistake Recommended Expressions Explanation Referring to the symbol d as del Since the time of the early Greeks, the name of this symbol has been and remains delta The word del describes either of two things in mathematics: an operator ( ) of the partial derivative sign ( ) d 13 C composition d 13 C value; or carbon isotope composition A composition of values is not possible Isotopically depleted water 18 O Water is not depleted (or enriched) in isotopes Heavy (light) d 18 O values High (low) d 18 O values As number, d values are either high or low Depleted d 13 C value low d 13 C value (relative to another value) d 13 C values are numbers and cannot be depleted Oxygen isotopes in chert; inferred from carbon isotopes; isotopes of soil water Oxygen isotope ratio (composition) of chert; inferred from carbon isotope measurements; isotopic composition of soil water Such mistakes are a carryover from loose oral communication The isotopic composition of water was d 18 O = - 4. 3‰ The d 18 O value of the water was - 4. 3‰ A matter of redundancy The isotopic value changed; the carbon value changed The isotopic composition changed; the d 13 C value changed The phrase “isotopic value” is ambiguous. Does it mean a ratio? A delta value? Enriched (depleted) carbonates Isotopically heavy (light) carbonates; (relatively) 18 O-rich carbonates; (relatively) 13 C-poor carbonates Low 13 C source; source with a low d 13 C value These phrases culled from the literature make no sense. More importantly, the words enrich and deplete are overused and much abused. These words should be reserved for describing a process that changes the content of the heavy isotope of some element in the substance being considered The isotopic signature of the rock was d 18 O = 5. 7 ‰ The d 18 O value of the rock was 5. 7 ‰. Thus this rock has the oxygen isotope signature of the mantle The word signature should apply to the isotopic composition of a significant reservoir like the mantle, the ocean, or a major part of the system being studied, not to the isotopic composition of ordinary samples d 15, d 18 , d 13 , etc. 15 d, 18 d , 13 d , etc. d-15, d-13 , d-18 etc. d 15 N, d 18 O , d 13 C, etc. Introduction of new symbolism that saves one character of space is unnecessary at best and confusing at worst. The mineral equilibrated with the fluid The mineral exchanged with the fluid Isotopic equilibrium, may not have been attained during the process being described Sulfur was measured The sulfur isotope composition was measured Confusing because the sulfur content of a rock or mineral may be understood Depleted carbon source (or D) depleted water
Mistake Recommended Expressions Explanation Referring to the symbol d as del Since the time of the early Greeks, the name of this symbol has been and remains delta The word del describes either of two things in mathematics: an operator ( ) of the partial derivative sign ( )

Mistake Recommended Expressions Explanation Referring to the symbol d as del Since the time of the early Greeks, the name of this symbol has been and remains delta The word del describes either of two things in mathematics: an operator ( ) of the partial derivative sign ( ) d 13 C composition d 13 C value; or carbon isotope composition A composition of values is not possible

Mistake Recommended Expressions Explanation Referring to the symbol d as del Since the time of the early Greeks, the name of this symbol has been and remains delta The word del describes either of two things in mathematics: an operator ( ) of the partial derivative sign ( ) d 13 C composition d 13 C value; or carbon isotope composition A composition of values is not possible Isotopically depleted water 18 O Water is not depleted (or enriched) in isotopes (or D) depleted water

Mistake Recommended Expressions Explanation Referring to the symbol d as del Since the time of the early Greeks, the name of this symbol has been and remains delta The word del describes either of two things in mathematics: an operator ( ) of the partial derivative sign ( ) d 13 C composition d 13 C value; or carbon isotope composition A composition of values is not possible Isotopically depleted water 18 O Water is not depleted (or enriched) in isotopes Heavy (light) d 18 O values High (low) d 18 O values (or D) depleted water As number, d values are either high or low
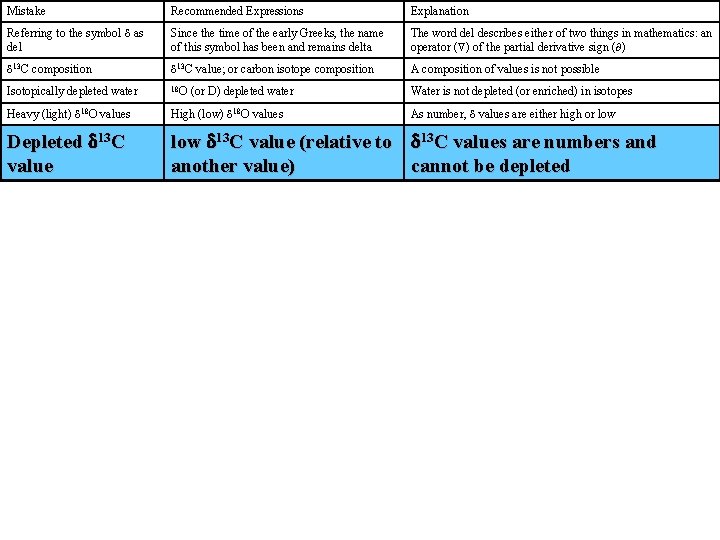
Mistake Recommended Expressions Explanation Referring to the symbol d as del Since the time of the early Greeks, the name of this symbol has been and remains delta The word del describes either of two things in mathematics: an operator ( ) of the partial derivative sign ( ) d 13 C composition d 13 C value; or carbon isotope composition A composition of values is not possible Isotopically depleted water 18 O Water is not depleted (or enriched) in isotopes Heavy (light) d 18 O values High (low) d 18 O values As number, d values are either high or low Depleted d 13 C value low d 13 C value (relative to another value) d 13 C values are numbers and cannot be depleted (or D) depleted water
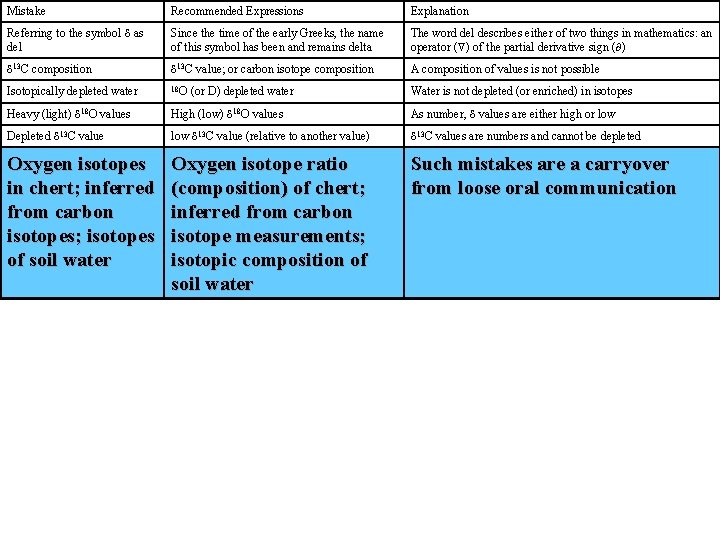
Mistake Recommended Expressions Explanation Referring to the symbol d as del Since the time of the early Greeks, the name of this symbol has been and remains delta The word del describes either of two things in mathematics: an operator ( ) of the partial derivative sign ( ) d 13 C composition d 13 C value; or carbon isotope composition A composition of values is not possible Isotopically depleted water 18 O Water is not depleted (or enriched) in isotopes Heavy (light) d 18 O values High (low) d 18 O values As number, d values are either high or low Depleted d 13 C value low d 13 C value (relative to another value) d 13 C values are numbers and cannot be depleted Oxygen isotopes in chert; inferred from carbon isotopes; isotopes of soil water Oxygen isotope ratio (composition) of chert; inferred from carbon isotope measurements; isotopic composition of soil water Such mistakes are a carryover from loose oral communication (or D) depleted water
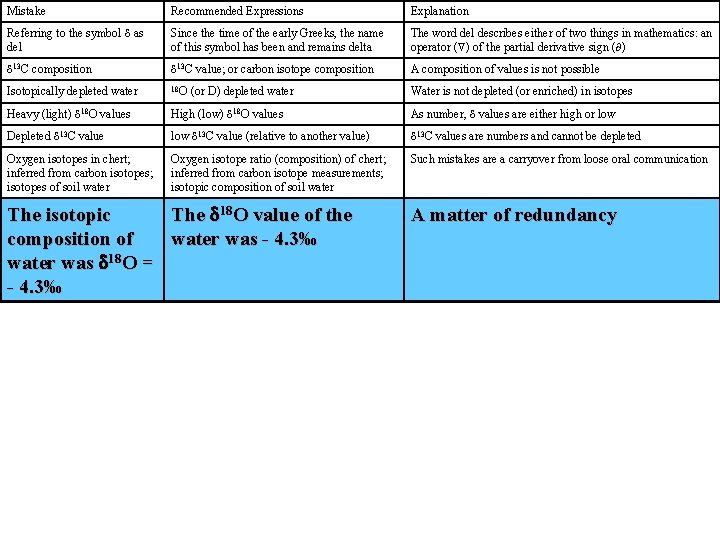
Mistake Recommended Expressions Explanation Referring to the symbol d as del Since the time of the early Greeks, the name of this symbol has been and remains delta The word del describes either of two things in mathematics: an operator ( ) of the partial derivative sign ( ) d 13 C composition d 13 C value; or carbon isotope composition A composition of values is not possible Isotopically depleted water 18 O Water is not depleted (or enriched) in isotopes Heavy (light) d 18 O values High (low) d 18 O values As number, d values are either high or low Depleted d 13 C value low d 13 C value (relative to another value) d 13 C values are numbers and cannot be depleted Oxygen isotopes in chert; inferred from carbon isotopes; isotopes of soil water Oxygen isotope ratio (composition) of chert; inferred from carbon isotope measurements; isotopic composition of soil water Such mistakes are a carryover from loose oral communication (or D) depleted water The isotopic The d 18 O value of the composition of water was - 4. 3‰ water was d 18 O = - 4. 3‰ A matter of redundancy
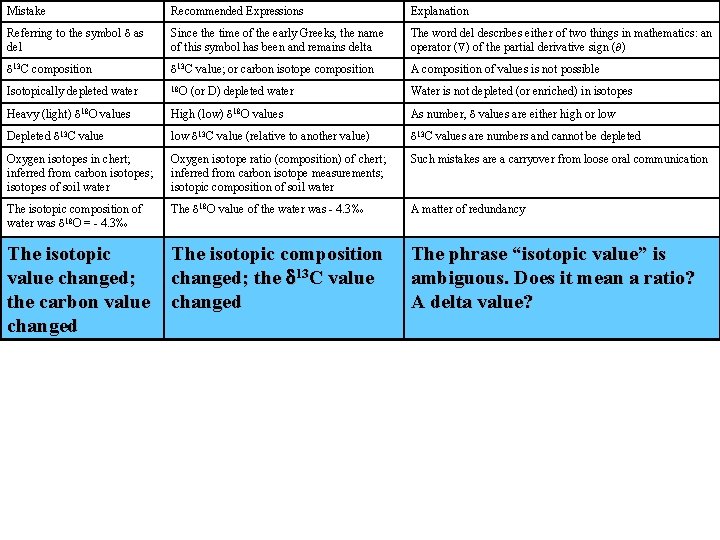
Mistake Recommended Expressions Explanation Referring to the symbol d as del Since the time of the early Greeks, the name of this symbol has been and remains delta The word del describes either of two things in mathematics: an operator ( ) of the partial derivative sign ( ) d 13 C composition d 13 C value; or carbon isotope composition A composition of values is not possible Isotopically depleted water 18 O Water is not depleted (or enriched) in isotopes Heavy (light) d 18 O values High (low) d 18 O values As number, d values are either high or low Depleted d 13 C value low d 13 C value (relative to another value) d 13 C values are numbers and cannot be depleted Oxygen isotopes in chert; inferred from carbon isotopes; isotopes of soil water Oxygen isotope ratio (composition) of chert; inferred from carbon isotope measurements; isotopic composition of soil water Such mistakes are a carryover from loose oral communication The isotopic composition of water was d 18 O = - 4. 3‰ The d 18 O value of the water was - 4. 3‰ A matter of redundancy The isotopic value changed; the carbon value changed The isotopic composition changed; the d 13 C value changed The phrase “isotopic value” is ambiguous. Does it mean a ratio? A delta value? (or D) depleted water
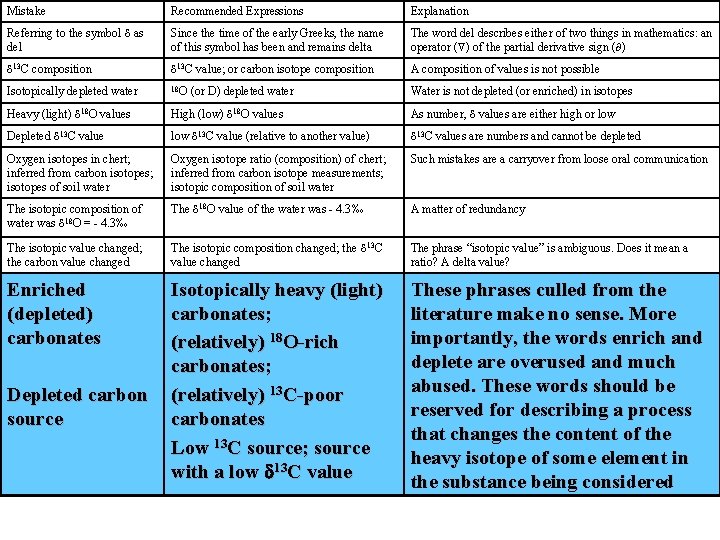
Mistake Recommended Expressions Explanation Referring to the symbol d as del Since the time of the early Greeks, the name of this symbol has been and remains delta The word del describes either of two things in mathematics: an operator ( ) of the partial derivative sign ( ) d 13 C composition d 13 C value; or carbon isotope composition A composition of values is not possible Isotopically depleted water 18 O Water is not depleted (or enriched) in isotopes Heavy (light) d 18 O values High (low) d 18 O values As number, d values are either high or low Depleted d 13 C value low d 13 C value (relative to another value) d 13 C values are numbers and cannot be depleted Oxygen isotopes in chert; inferred from carbon isotopes; isotopes of soil water Oxygen isotope ratio (composition) of chert; inferred from carbon isotope measurements; isotopic composition of soil water Such mistakes are a carryover from loose oral communication The isotopic composition of water was d 18 O = - 4. 3‰ The d 18 O value of the water was - 4. 3‰ A matter of redundancy The isotopic value changed; the carbon value changed The isotopic composition changed; the d 13 C value changed The phrase “isotopic value” is ambiguous. Does it mean a ratio? A delta value? Enriched (depleted) carbonates Isotopically heavy (light) carbonates; (relatively) 18 O-rich carbonates; (relatively) 13 C-poor carbonates Low 13 C source; source with a low d 13 C value These phrases culled from the literature make no sense. More importantly, the words enrich and deplete are overused and much abused. These words should be reserved for describing a process that changes the content of the heavy isotope of some element in the substance being considered Depleted carbon source (or D) depleted water
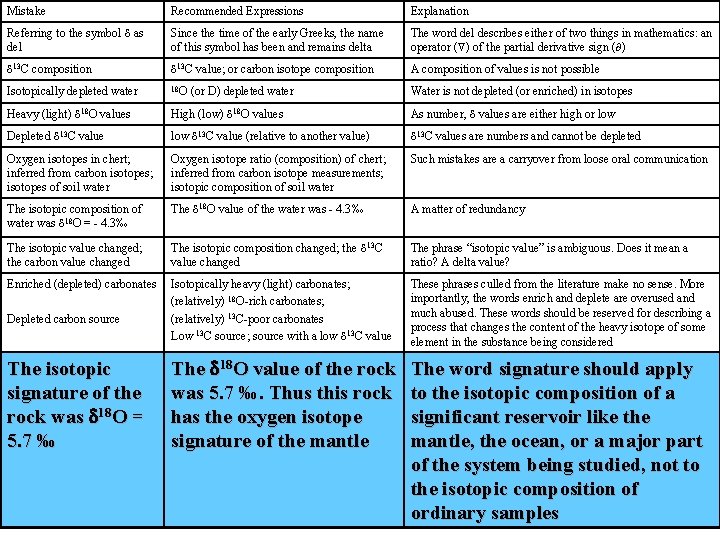
Mistake Recommended Expressions Explanation Referring to the symbol d as del Since the time of the early Greeks, the name of this symbol has been and remains delta The word del describes either of two things in mathematics: an operator ( ) of the partial derivative sign ( ) d 13 C composition d 13 C value; or carbon isotope composition A composition of values is not possible Isotopically depleted water 18 O Water is not depleted (or enriched) in isotopes Heavy (light) d 18 O values High (low) d 18 O values As number, d values are either high or low Depleted d 13 C value low d 13 C value (relative to another value) d 13 C values are numbers and cannot be depleted Oxygen isotopes in chert; inferred from carbon isotopes; isotopes of soil water Oxygen isotope ratio (composition) of chert; inferred from carbon isotope measurements; isotopic composition of soil water Such mistakes are a carryover from loose oral communication The isotopic composition of water was d 18 O = - 4. 3‰ The d 18 O value of the water was - 4. 3‰ A matter of redundancy The isotopic value changed; the carbon value changed The isotopic composition changed; the d 13 C value changed The phrase “isotopic value” is ambiguous. Does it mean a ratio? A delta value? Enriched (depleted) carbonates Isotopically heavy (light) carbonates; (relatively) 18 O-rich carbonates; (relatively) 13 C-poor carbonates Low 13 C source; source with a low d 13 C value These phrases culled from the literature make no sense. More importantly, the words enrich and deplete are overused and much abused. These words should be reserved for describing a process that changes the content of the heavy isotope of some element in the substance being considered The d 18 O value of the rock was 5. 7 ‰. Thus this rock has the oxygen isotope signature of the mantle The word signature should apply to the isotopic composition of a significant reservoir like the mantle, the ocean, or a major part of the system being studied, not to the isotopic composition of ordinary samples Depleted carbon source The isotopic signature of the rock was d 18 O = 5. 7 ‰ (or D) depleted water
- Slides: 100