Training course on the Pharmacovigilance of antiretroviral medicines











- Slides: 13

Training course on the Pharmacovigilance of antiretroviral medicines 23 – 28 November 2009 Rationale, Objectives and Expected Outcomes

Why do we need this course l Significant safety issues exist with ARVs l Both short and long-term effects l Good experience in above issues in industrialised world l Insufficient / little information in RLS l Important to address safety issues as relevant to RLS: with characteristic co-morbidities etc l New tools to share 2| TITLE from VIEW and SLIDE MASTER | 19 January 2022

Who is organizing this l WHO HQ Regions Countries – HIV AIDS Department – Essential Medicines Department l With funds from BMGF, EC – HIV PV project – EC malaria project 3| TITLE from VIEW and SLIDE MASTER | 19 January 2022

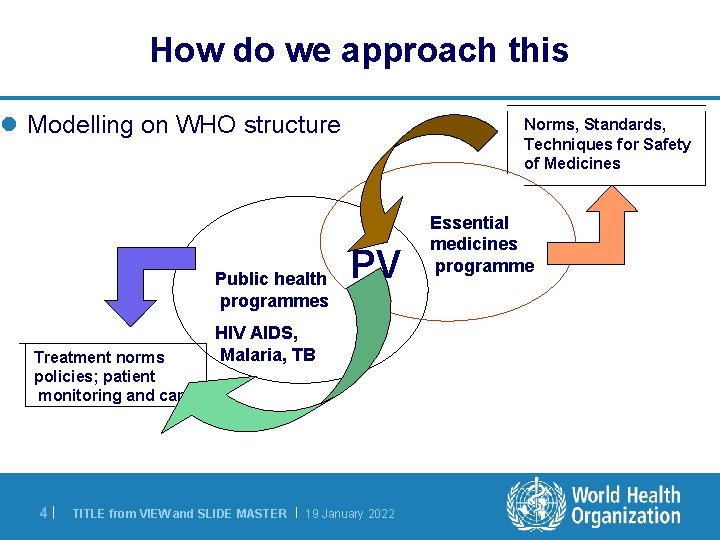
How do we approach this l Modelling on WHO structure Public health programmes Treatment norms policies; patient monitoring and care 4| Norms, Standards, Techniques for Safety of Medicines PV HIV AIDS, Malaria, TB TITLE from VIEW and SLIDE MASTER | 19 January 2022 Essential medicines programme

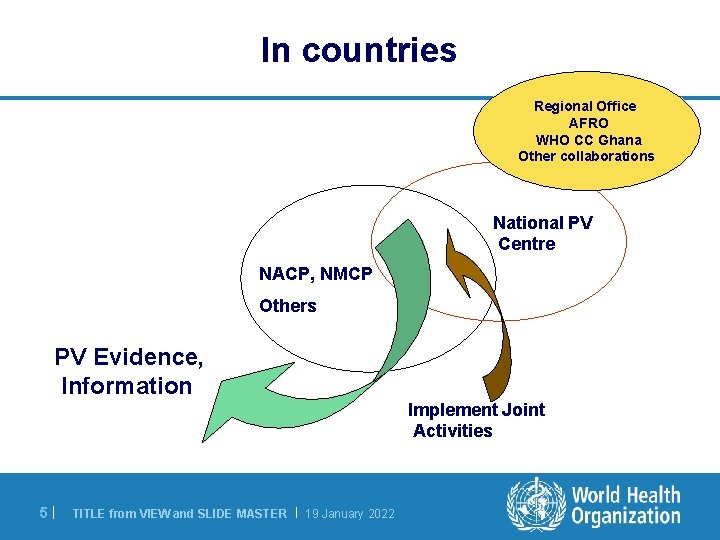
In countries Regional Office AFRO WHO CC Ghana Other collaborations National PV Centre NACP, NMCP Others PV Evidence, Information Implement Joint Activities 5| TITLE from VIEW and SLIDE MASTER | 19 January 2022

Objectives Major: to agree upon a common approach to PV l To build capacity for ARV-PV (in NPVCs & NACP) l To introduce the principles of – PV in HIV programmes – Cohort Event Monitoring for ARVs – The clinical management of adverse reactions with ARVs – PV/HIV training modules l To initiate plans for early implementation of pilot projects 6| TITLE from VIEW and SLIDE MASTER | 19 January 2022

Audience and perspectives Expected outcomes

Audience: Who are we? l PV and HIV experts l From WHO HQ, regions and countries, UMC and UMC -A l 8 Countries invited based on specific strengths and criteria: – "Teams" from members countries of the drug monitoring programme – With experience to share, commitment in HIV and PV – Readiness to implement 8| TITLE from VIEW and SLIDE MASTER | 19 January 2022

A tripartite programme WHO Collaborating Centres, Uppsala Ghana WHO HQ + 6 Regional offices National Programmes & Centres 9| TITLE from VIEW and SLIDE MASTER | 19 January 2022

What do we look at? Norms, Standards, Techniques for Safety of Medicines Public health programmes PV Essential medicines programme HIV AIDS, ? ? Treatment norms policies; Patient monitoring and care ? 10 | TITLE from VIEW and SLIDE MASTER | 19 January 2022 Methods and Tools Strategies Training Plans

Key questions How best could we: l Prevent the HIV "verticalism and exceptionalism": l Integrate – passive and active monitoring – cohorts collaborations – promote cross countries collaborations 11 | TITLE from VIEW and SLIDE MASTER | 19 January 2022

What do we expect? l A critical look at needs and means to address these 12 | TITLE from VIEW and SLIDE MASTER | 19 January 2022

What to bring back? l A proposed plan for modelling PV for ARVs l A training package to be rolled on in countries 13 | TITLE from VIEW and SLIDE MASTER | 19 January 2022