TPD and XPS of Adsorbed Xenon Atoms for

TPD and XPS of Adsorbed Xenon Atoms for the Characterization of Reaction Sites on Oxygen-Modified Ni(110) Surfaces Hansheng Guo, Francisco Zaera Department of Chemistry, University of California Riverside, CA 92521 AVS 51 st International Symposium & Exhibition, 11/18/2004, Anaheim, CA and H. -S. Guo and F. Zaera, Nature Materials, 5(6), 489 -493 (2006). (Financial support: US National Science Foundation)

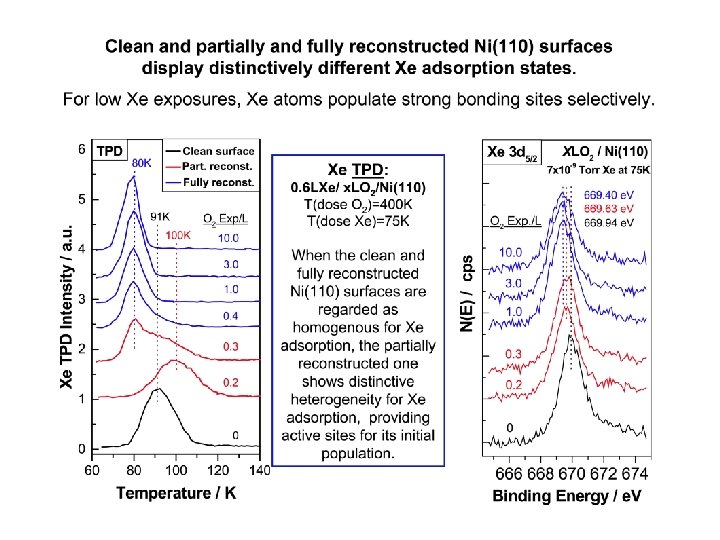
Techniques and research significance Experiment demands – Samples have to be cooled to 75 K to facilitate Xe adsorption. – The first layer saturates around 75 K on Ni(110) surface – No Xe condensation on the surfaces saturated with CO or NH 3. Useful properties of adsorbed Xe – The selective population of specific sites with Xe atoms is governed by the corresponding adsorption energies, and those can be measured by temperatureprogrammed desorption (TPD) – The electronic property of adsorbed Xe is site-sensitive, it depends on the local chemical environment. This can be characterized by X-ray photoelectron spectroscopy (XPS). – A combination of TPD with XPS of adsorbed Xe can therefore be used to characterize local surface heterogeneities such as catalytic active sites.
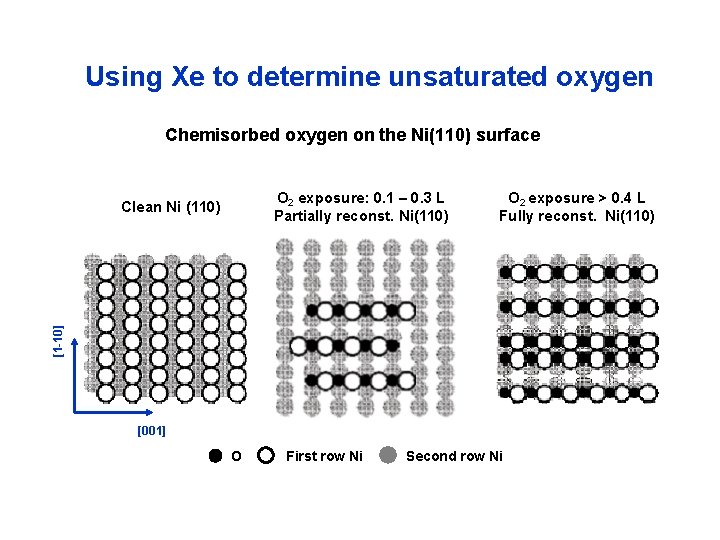
Using Xe to determine unsaturated oxygen Chemisorbed oxygen on the Ni(110) surface O 2 exposure: 0. 1 – 0. 3 L Partially reconst. Ni(110) O 2 exposure > 0. 4 L Fully reconst. Ni(110) [1 -10] Clean Ni (110) [001] O First row Ni Second row Ni

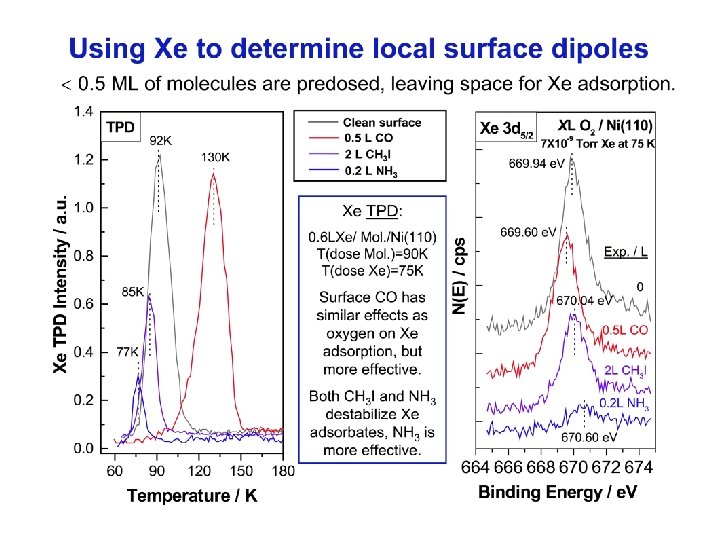

Xe TPD On the Ni(110) surface with these polar molecules ( 0. 5 ML), Xe atoms tend to adsorb next to the CO sites but away from the NH 3. CH 3 I adsorbates seem to have a similar but less marked effect on the adsorption of xenon. Xe (3 d 5/2) XPS Dipolar adsorbates cause shifts on the Xe 3 d 5/2 level that depend on dipole moment and orientation. This is associated with the local dipole interaction or the local WF change ( ). Photoelectron ejected from Xe atoms sense the individual neighboring dipoles Xe+CO / Ni(110) sense the negative end: EK slightly increases EB slightly decreases + Xe O C Local work function : increased EB (Xe 3 d 5/2) h - Xe+NH 3 / Ni(110) sense the positive end: EK decreases EB increases Xe+CH 3 I / Ni(110) sense the positive end: EK slightly decreases EB slightly increases + + Xe Xe decreased N decreased C I
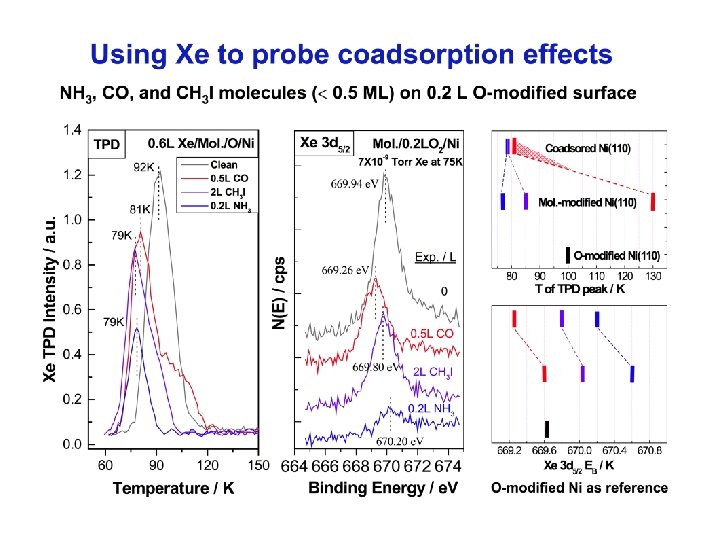

Arrangement of the molecules on the 0. 2 L O-modified surface 1. NH 3 on O/Ni(110) The terminating atoms of the -Ni-O- added rows provide active sites for the initial population of NH 3. The predosed oxygen shows compensation effects on the NH 3 -induced heterogeneity for Xe adsorption (Td and Xe 3 d 5/2). 2. CO on O/Ni(110) CO adsorption on the O-modified surface does not show an evident site selectivity between the bare nickel and the end of the -Ni-O- added rows. Both CO and O modify Xe adsorption in a similar way. 3. CH 3 I on O/Ni(110) CH 3 I behaves similarly to NH 3 on the surface, but shows less site selectivity. CH 3 I exerts a lesser effect on Xe adsorption when it is dosed alone or with O 2.

Summary XPS and TPD of adsorbed Xe can provide valuable molecular-level information about specific sites on heterogeneous surfaces. • Why Xe atoms 1. Xe exhibits the highest induced dipole moment ( ). 2. , adsorption energies (Ead), and core level energies (EB) are all site specific and vary with surface coordination and neighboring environment. 3. Xe adsorbates tend to form hexagonally close-packed overlayer of known atom density, and that allows for an easy calibration to count active sites. • Advantages 1. Selective population of specific sites active sites. 2. Does not affecti surface reactions. • Limitations 1. The number of different sites should not exceed 2 or 3. 2. Knowledge of surface geometry desirable. 3. Data interpretation complicated by coordination numbers, Van der Waals interactions, charge transfers, surface dipoles, etc.
- Slides: 10