Toxic Elements Dr Sherin Bakhashab Introduction Heavy metals

Toxic Elements Dr. Sherin Bakhashab

Introduction Heavy metals are toxic to human health Most common heavy metals are lead(Pb), mercury(Hg), cadmium(Cd) and arsenic(As) Indoor concentration of heavy metals is generally less than their outdoor concentration They are mainly produced by industrial activities, and deposit slowly in the surrounding water and soil
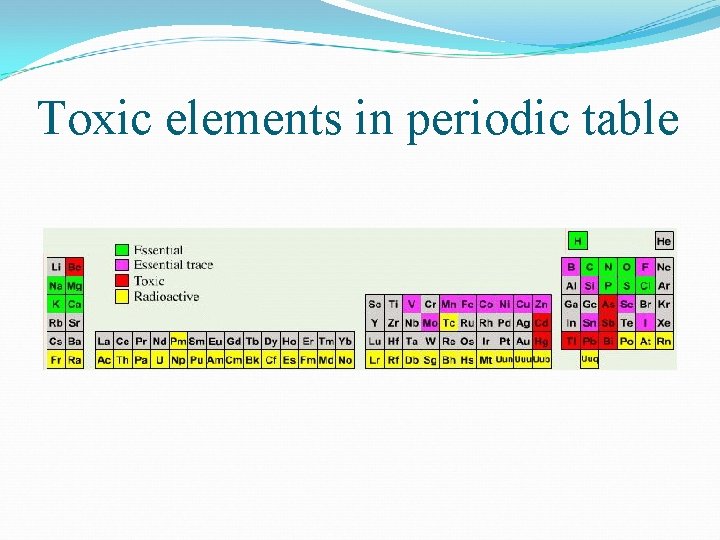
Toxic elements in periodic table

Properties of heavy metals They occur near the bottom of the periodic table Have high densities Toxic in nature Nondegradable Note: Arsenic is not actually a metal but is a semimetal i. e. its properties are intermediate between those of metals and nonmetals.

Transport phenomenon Water Food Air Adsorption or absorption onto various materials Example: Over half of the heavy metal input into Great Lakes is due to deposition from air.

Toxicity of heavy metals Mercury is highly toxic in vapor form but lead, cadmium and arsenic are more toxic in their cationic form Toxicity arises from strong affinity of the heavy metal cations for sulfur Medicinal treatment for heavy metal poisoning is done by chelation therapy by administering compounds known as chelates Example : British Anti-Lewisite(BAL), ethylene diamine tetra acetic acid(EDTA).
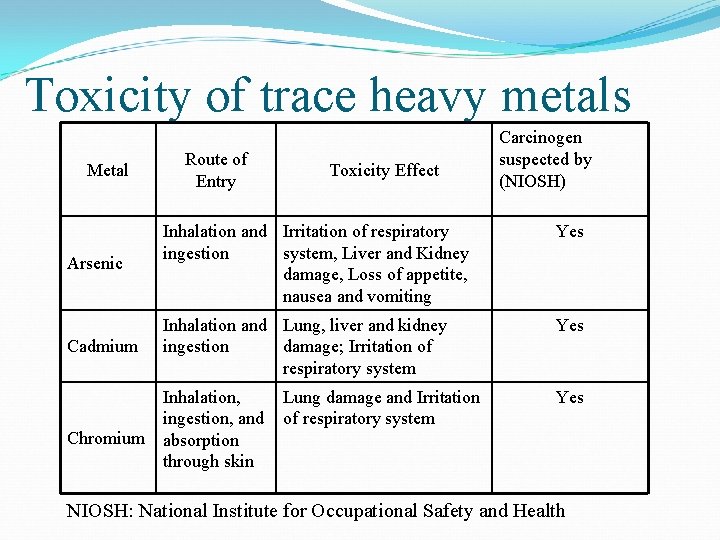
Toxicity of trace heavy metals Metal Route of Entry Toxicity Effect Carcinogen suspected by (NIOSH) Yes Arsenic Inhalation and Irritation of respiratory ingestion system, Liver and Kidney damage, Loss of appetite, nausea and vomiting Yes Cadmium Inhalation and Lung, liver and kidney ingestion damage; Irritation of respiratory system Inhalation, ingestion, and absorption through skin Yes Chromium Lung damage and Irritation of respiratory system NIOSH: National Institute for Occupational Safety and Health
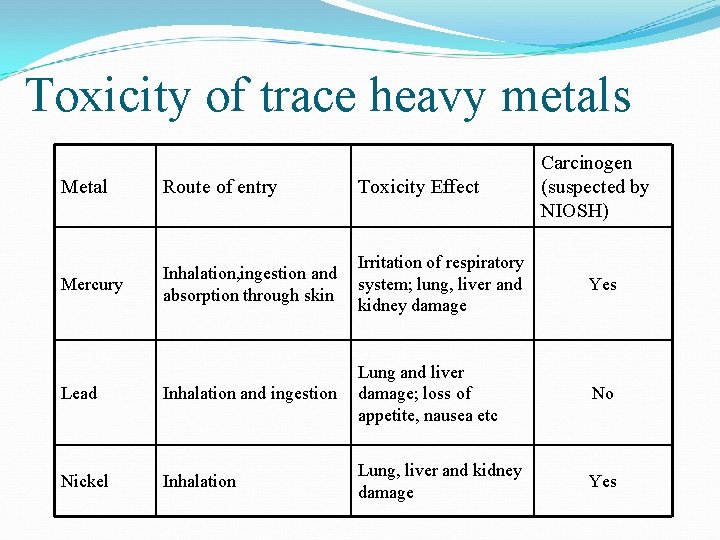
Toxicity of trace heavy metals Carcinogen (suspected by NIOSH) Metal Route of entry Toxicity Effect Mercury Inhalation, ingestion and absorption through skin Irritation of respiratory system; lung, liver and kidney damage Yes Lead Inhalation and ingestion Lung and liver damage; loss of appetite, nausea etc No Nickel Inhalation Lung, liver and kidney damage Yes

Mercury

Mercury Most volatile of all metals Highly toxic in vapor form Liquid mercury itself is not highly toxic, and most of that ingested is excreted

Sources of Mercury Elemental mercury is employed in many applications due to its unusual property of being a liquid that conducts electricity Used in electrical switches, fluorescent light bulbs and mercury lamps Emission of mercury vapor from large industrial operations Unregulated burning of coal and fuel oil Emissions from mercury containing products : batteries, thermometers, etc. Mercury amalgams: dental fillings

Health effects Skin burns Irritation of nose and skin Rashes Excessive perspiration Damage to the kidneys Damage to vision Minamata disease: sever neurological disease Dysfunctions of the central nervous system Loss of hearing and muscle coordination Sever brain damage Death

Lead

Lead Has a very low melting point of 327 degrees C Romans used it in water ducts and in cooking vessels Analysis of ice-core samples from Greenland indicate that atmospheric lead concentration reached a peak in roman times

Sources of lead Commonly used in the building industry for roofing and flashing and for soundproofing Used in pipes When combined with tin, it forms solder, used in electronics and in other applications to make connections between solid metals Lead is also used in ammunition Note: Lead shots have been banned in United States, Canada, Netherlands, Norway and Denmark Lead is used in batteries and sinkers in fishing Ammunition

Sources (contd. ) Used in paints Lead chromate is the yellow pigment used in paints usually applied to school buses. Lead is also used in corrosion-resistant paints and has a bright red color Used in ceramics and dishware The leaching of lead from glazed ceramics used to prepare food is a major source of dietary lead, especially in Mexico In the past, lead salts were used as coloring agents in various foods Lead is used in some types of PVC miniblinds

Health effects At high levels, inorganic lead is a general metabolic poison Lead poisoning effects the neurological and reproductive systems Lead breaks the blood-brain barrier and interferes with the normal development of brain in infants

Health effects(contd. ) Lead is observed to lower IQ levels in children Lead is transferred postnatally from the mother in her breast milk At elevated levels, lead poisoning would eventually result in death

Facts about lead poisoning The human groups most at risk of lead poisoning are fetuses and children under the age of seven Episodes of lead poisoning were recorded through the middle ages and even until recent times A recent study in Mexico indicated that pregnant women can decrease the lead levels in their blood and presumably in the blood of their developing fetus by taking calcium supplements.

Cadmium

Cadmium lies in the same subgroup of the periodic table as zinc and mercury, but is more similar to zinc Coal burning is the main source of environmental cadmium Burning of wastes containing cadmium is an important source of the metal in the environment Cadmium is most toxic in its ionic form unlike mercury Note: Mercury is most toxic in vapor form and lead, cadmium and arsenic are most toxic in their ionic forms.

Sources of Cadmium is used as an electrode in “Ni. Cad” batteries Cadmium is used as a pigment in paints(yellow color) It is also used in photovoltaic devices and in TV screens Cigarette smoke Fertilizers and pesticides Note: The greatest proportion of our exposure to cadmium comes from our food supply- seafood, organ meats, particularly kidneys, and also from potatoes, rice, and other grains.

Health effects Severe pain in joints Bone diseases Kidney problems Its lifetime in the body is several years Areas of greatest risk are Japan and central Europe In very high levels it poses serious health problems related to bones, liver and kidneys and can eventually cause death.

Arsenic

Arsenic oxides were the common poisons used for murder and suicide from roman times through to the middle ages Arsenic compounds were used widely as pesticides before the organic chemicals era Arsenic is very much similar to phosphorous

Sources of Arsenic Pesticides Mining, melting of gold, lead, copper and nickel Production of iron and steel Combustion of coal Used as a wood preservative Herbicides Tobacco smoke Wallpaper paste and pigments in wallpaper

Health effects Birth defects Carcinogen: Lung cancer results from the inhalation of arsenic and probably also from its ingestion. Skin and liver cancer, and perhaps cancers of the bladder and kidneys, arise from ingested arsenic Gastrointestinal damage Severe vomiting Diarrhea Death

Recent studies on arsenic exposure Arsenic emitted from a copper-melting plant in Bulgaria has been shown recently to have produced a three-fold increase in birth defects in new born children in that area Most daily exposure of arsenic by north American adults is due to food intake, especially of meat and seafood

Recent studies. . (contd. ) Recent studies have shown that about 1% of Americans consume drinking water that has arsenic levels of 25 ppb or more, and in Utah and California water supplies have been found to contain as much as 500 ppb Scientists have estimated that there is a one-in-athousand lifetime risk of dying from cancer induced by normal background levels of arsenic ( this equals the risk estimate due to tobacco smoke and radon exposure ).

General sources of heavy metals in residential houses Infiltration from outside, along with the dust carried on shoes and clothes Indoor sources include old-lead and latex based paints, domestic water supply, burning of wood, and tobacco smoke Pesticides and fungicides are major sources of arsenic and mercury indoors

Sampling and Measurement

Methods for measurement of trace metals Most common method of collecting particulate matter is through filters Identification and concentration of individual trace metals like lead, cadmium, arsenic, mercury and chromium is determined by Atomic absorption spectrophotometer X-ray fluorescence

Atomic absorption spectrophotometer
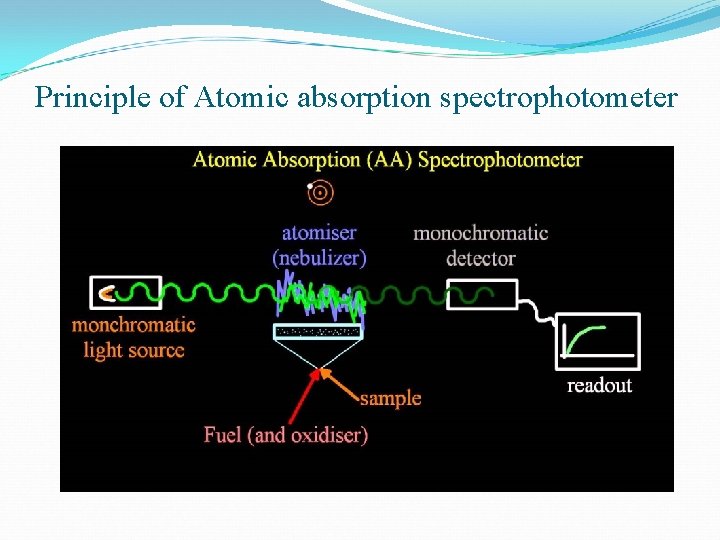
Principle of Atomic absorption spectrophotometer
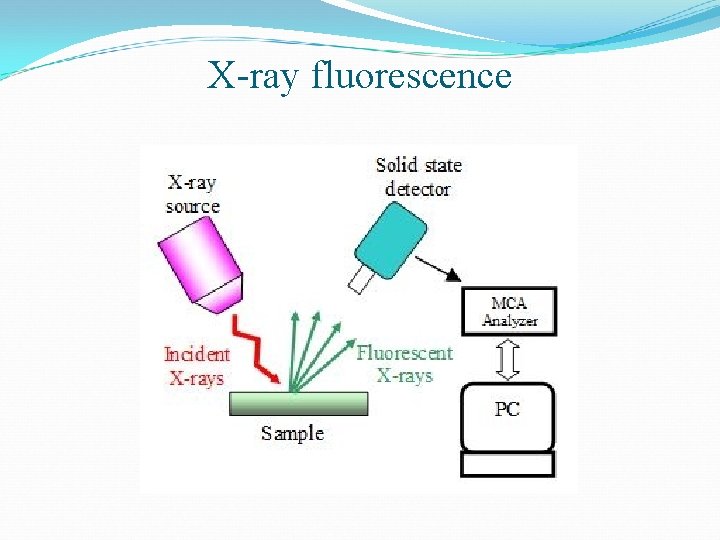
X-ray fluorescence

References Krishnamurthy, S. 1992. Biomethylation and environmental transport of metals. Journal of Chemical Education 69(5) Colin Baird. 2000. Environmental Chemistry, W. H. Freeman and Company, New York.
- Slides: 36