Topic Trends in the Periodic Table Ionization Energy

Topic: Trends in the Periodic Table: Ionization Energy and Electronegativity

REVIEW: How do you know if an atom gains or loses electrons? • Metals tend to lose electrons • Nonmetals tend to gain electrons

OR

Ionization Energy • the amount energy required to remove a valence electron from an atom in gas phase

previous | index | next • Cs valence electron lot farther away from nucleus than Li • attraction much weaker so easier to steal electron away from Cs • THEREFORE, Li has a higher Ionization energy then Cs
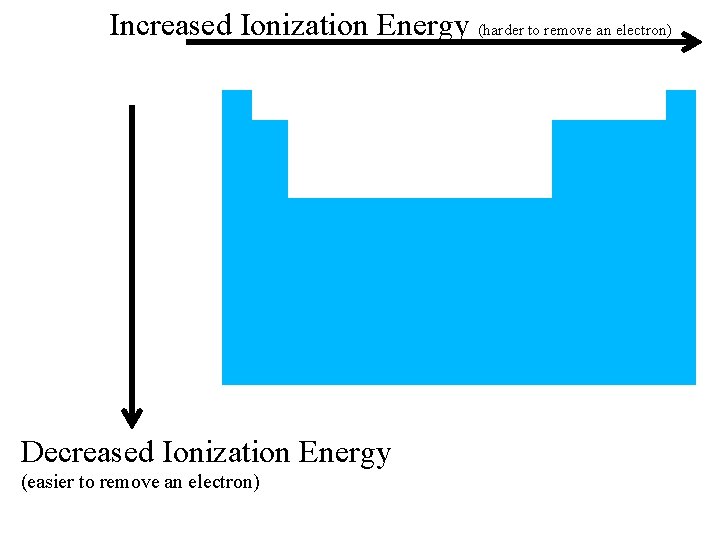
Increased Ionization Energy (harder to remove an electron) Decreased Ionization Energy (easier to remove an electron)

Electronegativity • ability of atom to attract electrons in bond • noble gases tend not to form bonds, so don’t have electronegativity values • Unit = Pauling • Fluorine: most electronegative element = 4. 0 Paulings
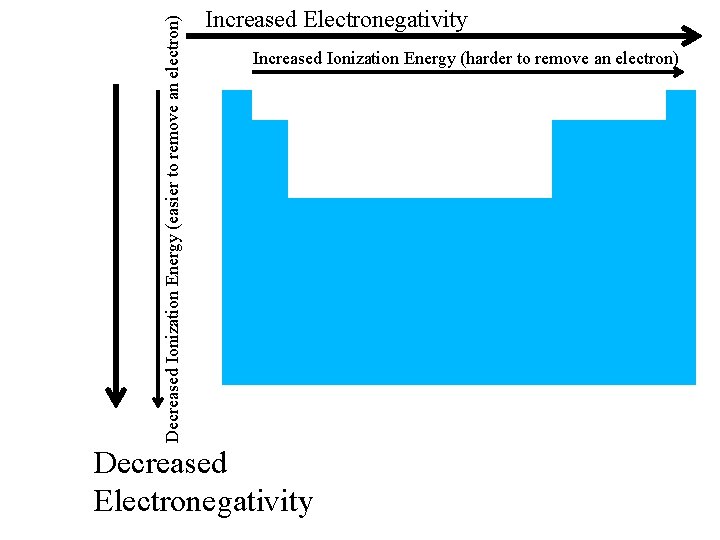
Decreased Ionization Energy (easier to remove an electron) Increased Electronegativity Increased Ionization Energy (harder to remove an electron) Decreased Electronegativity

Reactivity of Metals • judge reactivity of metals by how easily give up electrons (they’re losers) • So the easier it is to remove an electron the more reactive – Lower ionization energy = more reactive = more metallic
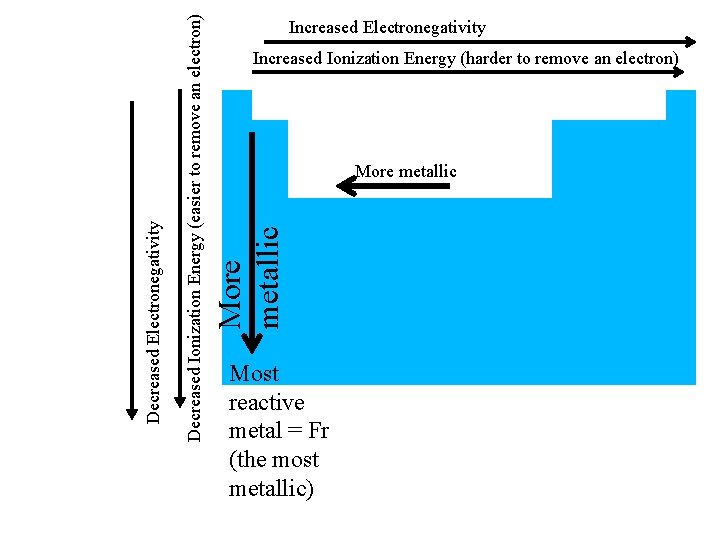
Increased Ionization Energy (harder to remove an electron) More metallic Decreased Ionization Energy (easier to remove an electron) Decreased Electronegativity Increased Electronegativity Most reactive metal = Fr (the most metallic)

Reactivity of Non-metals • judge reactivity of non-metals by how easily gain electrons (they are winners) • So the greater the affinity for electrons, the more reactive – Higher electronegativity= more reactive
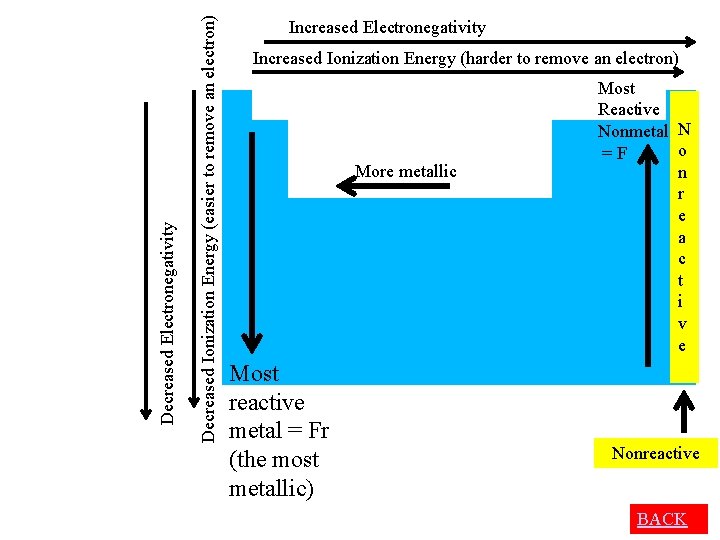
Decreased Ionization Energy (easier to remove an electron) Decreased Electronegativity Increased Ionization Energy (harder to remove an electron) More metallic Most reactive metal = Fr (the most metallic) Most Reactive Nonmetal N o = F n r e a c t i v e Nonreactive BACK
- Slides: 13