Topic Solubility and Table G Do Now label
Topic: Solubility and Table G Do Now: label as soluble and insoluble 1. CH 4 2. Ag. Cl 3. C 12 H 22 O 11 4. Na. NO 3 5. KOH

Review: Next 3 slides

Water is often used as a solvent Not everything dissolves in water • • Soluble = dissolves in water = aq Insoluble = doesn’t dissolve in water Miscible = 2 liquids that dissolve Immiscible = 2 liquids that don’t dissolve

How can you tell…. ? !!!? ? • Like Dissolve Like – Water is polar, so most polar compounds will dissolve in water • Things that dissolve in water – Soluble ionic compounds – use Table F – Acids - start with H except H 2 O 2 • ex HCl – Bases - metal + OH and NH 3 • ex. Na. OH – Polar covalent molecules • Remember polar = asymmetrical = poles nonpolar = symmetrical = no poles

Determining if an ionic compound is soluble (aq) or NOT • LOOK AT TABLE F – Li. OH – Cu(NO 3)2 – Ag. Cl 2 – Mg. S – Na. S 2 – KOH – aq – Insoluble = s – aq
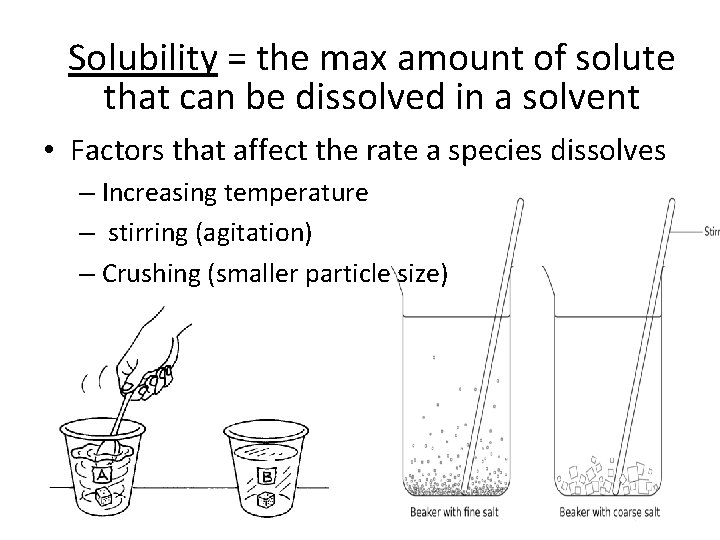
Solubility = the max amount of solute that can be dissolved in a solvent • Factors that affect the rate a species dissolves – Increasing temperature – stirring (agitation) – Crushing (smaller particle size)

Solubility • Many solids and gases dissolve in water • As you increase the temperature, you can dissolve more solid • Does this work the same with gas? • NOT the same for gases – as you increase temp, gas molecules KE ? • To get gases dissolve, decrease temperature and increase pressure

DEMO VIDEO

Summary: Factors Affecting Solubility • Nature of the solvent and the solute: LIKE DISSOLVES LIKE • Temperature • Pressure (for systems with gases)
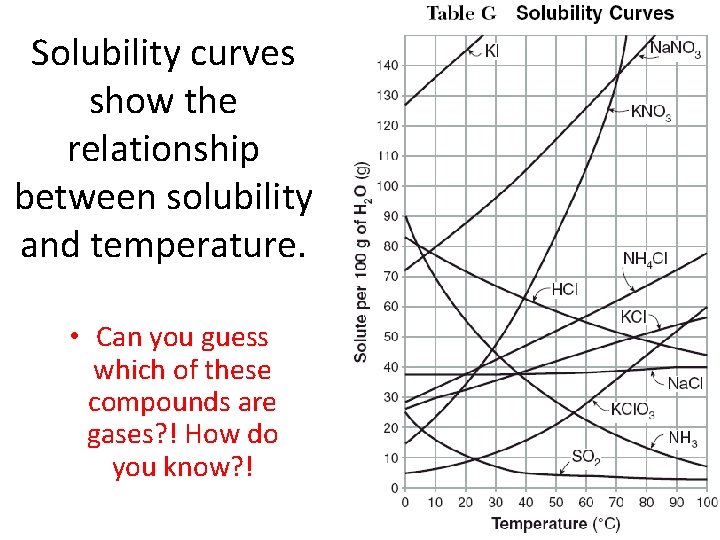
Solubility curves show the relationship between solubility and temperature. • Can you guess which of these compounds are gases? ! How do you know? !
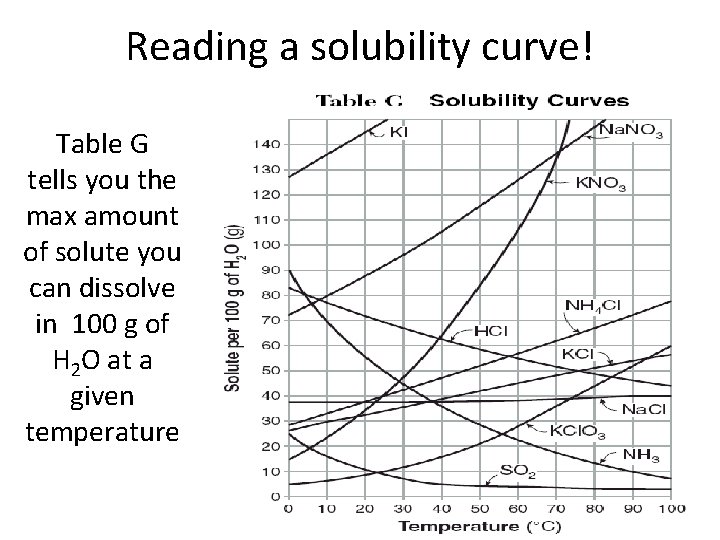
Reading a solubility curve! Table G tells you the max amount of solute you can dissolve in 100 g of H 2 O at a given temperature
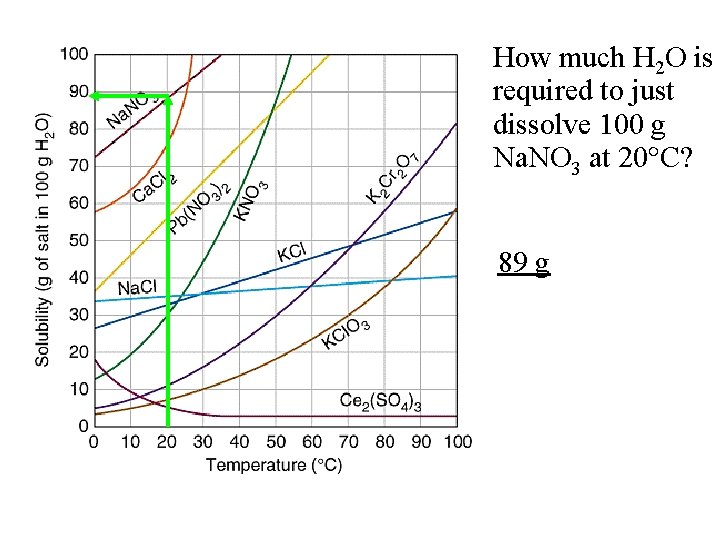
How much H 2 O is required to just dissolve 100 g Na. NO 3 at 20 C? 89 g
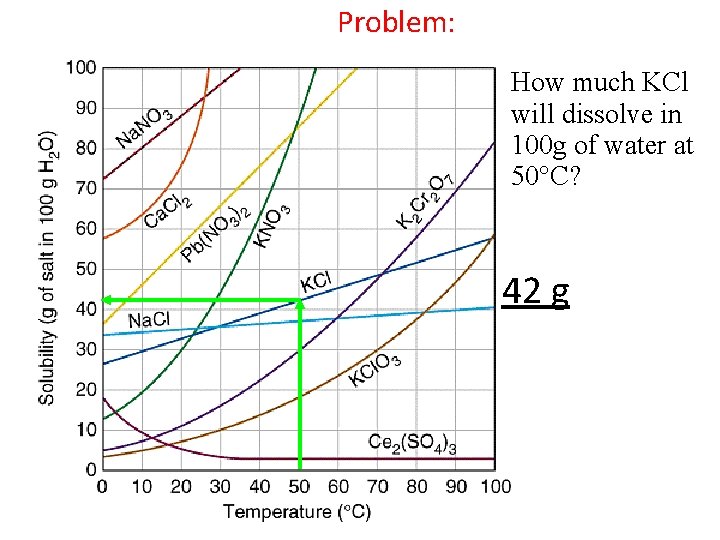
Problem: How much KCl will dissolve in 100 g of water at 50 C? 42 g
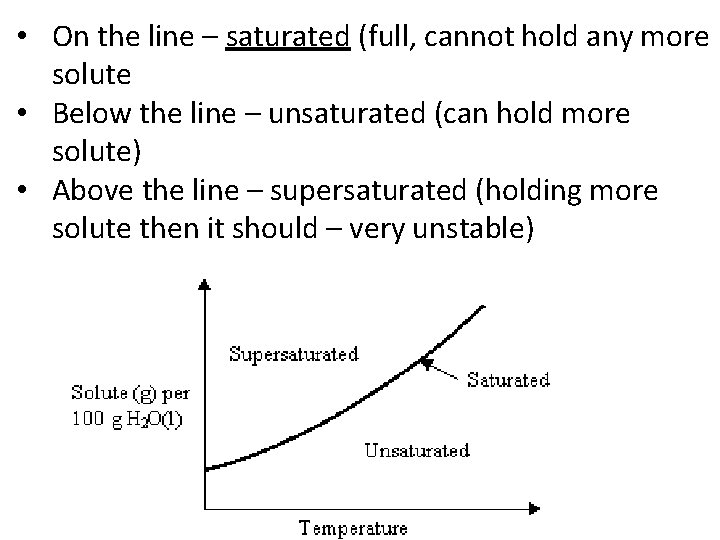
• On the line – saturated (full, cannot hold any more solute • Below the line – unsaturated (can hold more solute) • Above the line – supersaturated (holding more solute then it should – very unstable)

Unsaturated solution
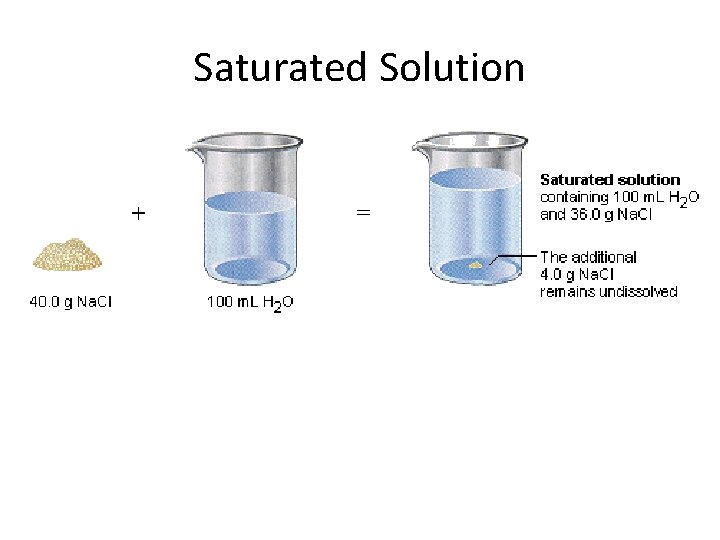
Saturated Solution

Supersaturated Solution (this picture is showing the addition of 100 g of glucose to 100 ml of water at 250 C) Note: at 250 C, only 91 g of glucose will dissolve in 100 ml of water Let’s see what happens
- Slides: 17