Topic Naming Binary Covalent Compounds Do Now 1

Topic: Naming Binary Covalent Compounds Do Now: 1) 2) 3) 4)

Binary Covalent Compounds • Composed of two nonmetals –can be same or different

Two naming systems • Stock system: – learned for ionic compounds – “official” • Traditional: – what hear on news – what see on ingredient lists

a Both Traditional & Stock • Least electronegative of two elements goes first • REMINDER: – electronegativity values in table S

Stock System • 1 st element must have Roman Numeral • Have to figure out oxidation number

Stock System & Oxidation Numbers • molecular substances DON’T contain ions • use concept of oxidation number for electron bookkeeping – pretend all shared electrons go to atom with higher electronegativity value – oxidation number would be: • atom with higher electronegativity value has (-) oxidation number • atom with lower electronegativity value is (+)

7 Rules for Oxidation Numbers 1. # for free, uncombined element = 0 Na He O 2 N 2 S 8 Cl 2 P 2. # for monatomic ion = charge on ion Ca+2 = +2 Cl-1 = -1 Al+3 = +3 3. Fluorine is always -1 CF 4 +4 -1 CF 4

Hydrogen is nearly always +1, except 4. when bonded to a metal-then it’s -1 +1 -1 HCl +1 -1 Li. H +2 -1 Ca. H 2 5. Oxygen is nearly always -2 EXCEPT when its: -Bonded to fluorine, where O is +2 -In the peroxide ion, where O is -1 +2 -1 OF 2 +1 -1 H 2 O 2
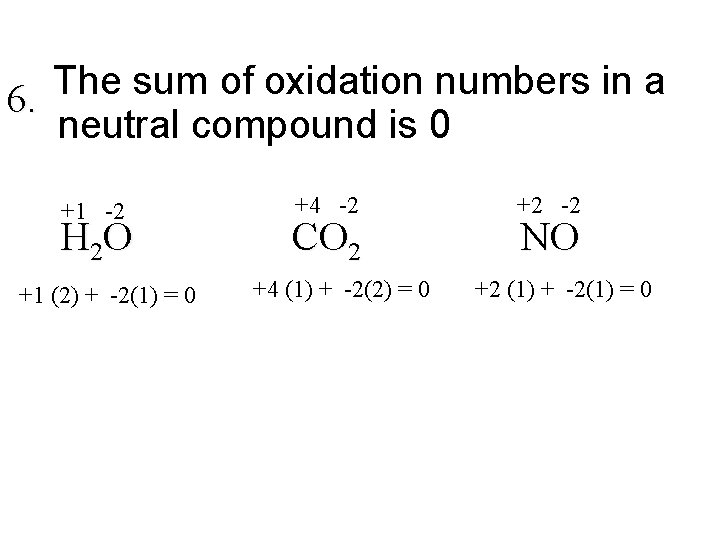
The sum of oxidation numbers in a 6. neutral compound is 0 +1 -2 H 2 O +1 (2) + -2(1) = 0 +4 -2 CO 2 +4 (1) + -2(2) = 0 +2 -2 NO +2 (1) + -2(1) = 0
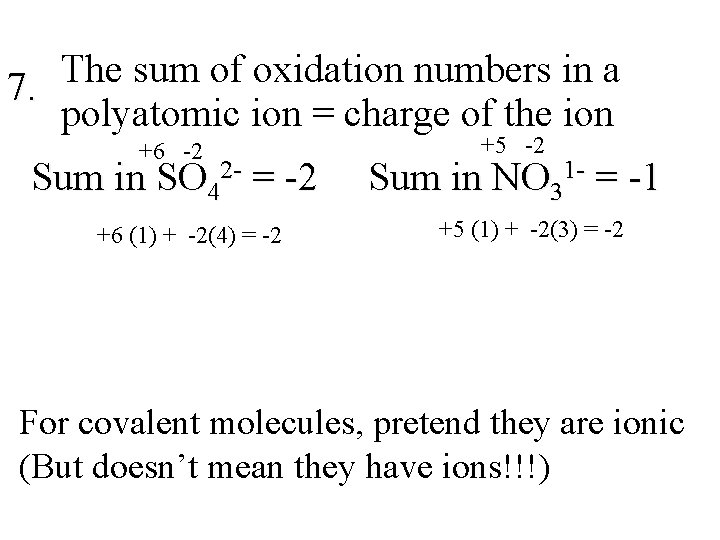
The sum of oxidation numbers in a 7. polyatomic ion = charge of the ion +6 -2 Sum in SO 42 - = -2 +6 (1) + -2(4) = -2 +5 -2 Sum in NO 31 - = -1 +5 (1) + -2(3) = -2 For covalent molecules, pretend they are ionic (But doesn’t mean they have ions!!!)

Naming Binary Covalent Compounds Stock System
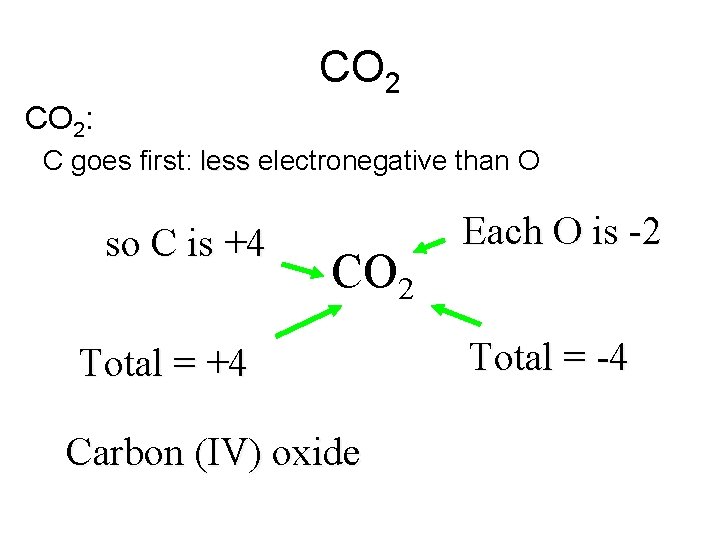
CO 2: C goes first: less electronegative than O so C is +4 CO 2 Total = +4 Carbon (IV) oxide Each O is -2 Total = -4
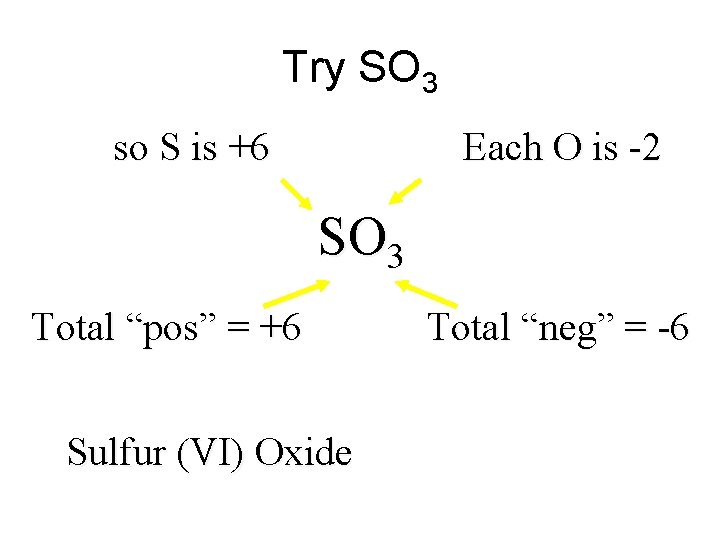
Try SO 3 so S is +6 Each O is -2 SO 3 Total “pos” = +6 Sulfur (VI) Oxide Total “neg” = -6
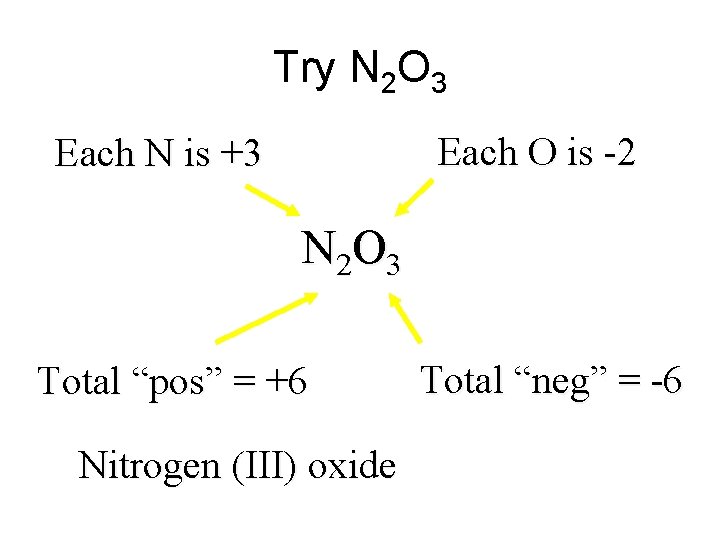
Try N 2 O 3 Each O is -2 Each N is +3 N 2 O 3 Total “pos” = +6 Nitrogen (III) oxide Total “neg” = -6
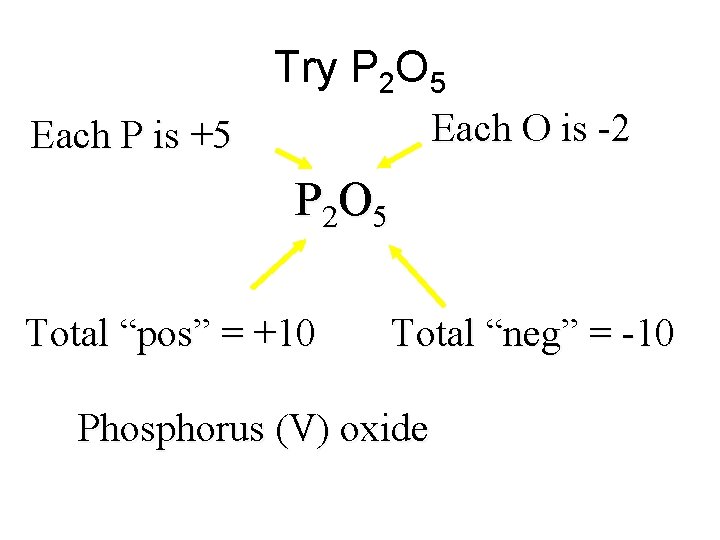
Try P 2 O 5 Each O is -2 Each P is +5 P 2 O 5 Total “pos” = +10 Total “neg” = -10 Phosphorus (V) oxide

Naming Binary Covalent Compounds Traditional Naming System

Traditional Naming 1. less electronegative element named 1 st 2. stem 2 nd element plus “-ide” 3. prefixes tell how many of each element Exception: NEVER start a name with “mono-”
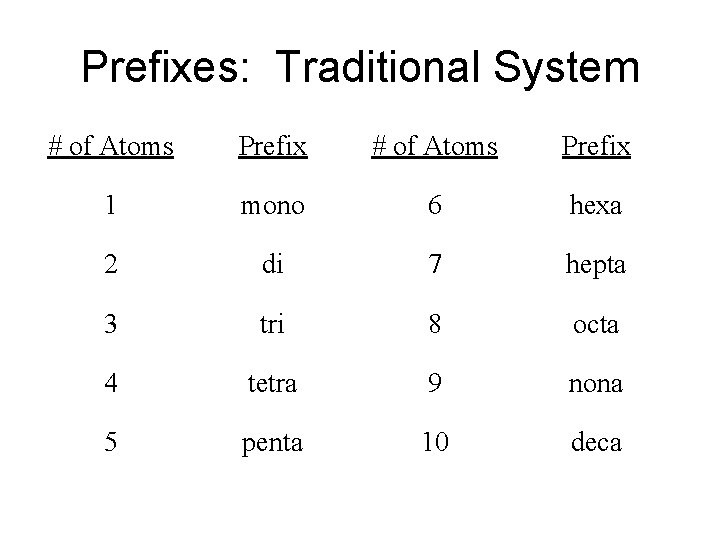
Prefixes: Traditional System # of Atoms Prefix 1 mono 6 hexa 2 di 7 hepta 3 tri 8 octa 4 tetra 9 nona 5 penta 10 deca

Stems: just a reminder H = hydr C = carb N = nitr O = ox F = fluor Si = silic P= phosph S = sulf Cl = chlor As = arsen Se = selen Br = brom Te = tellur I = iod

Traditional Naming H 2 O dihydrogen monoxide NH 3 nitrogen trihydride N 2 H 4 dinitrogen tetrahydride NO nitrogen monoxide NO 2 nitrogen dioxide N 2 O dinitrogen monoxide
- Slides: 20