Topic Chemistry Aim Describe the difference between elements

Topic: Chemistry Aim: Describe the difference between elements and compounds. Do Now: Bottom of Atoms reading notes HW: Ditto - Measuring Mass and Volume Part A (Properties Reading Notes due tomorrow)

Let’s review… 1. Identify the building blocks of matter. 2. Identify the subatomic particles that make up an atom. 3. Describe the location of each subatomic particle. 4. Describe the charge of each subatomic particle. 5. What is an atom’s atomic number indicate?
Oxygen atom + -
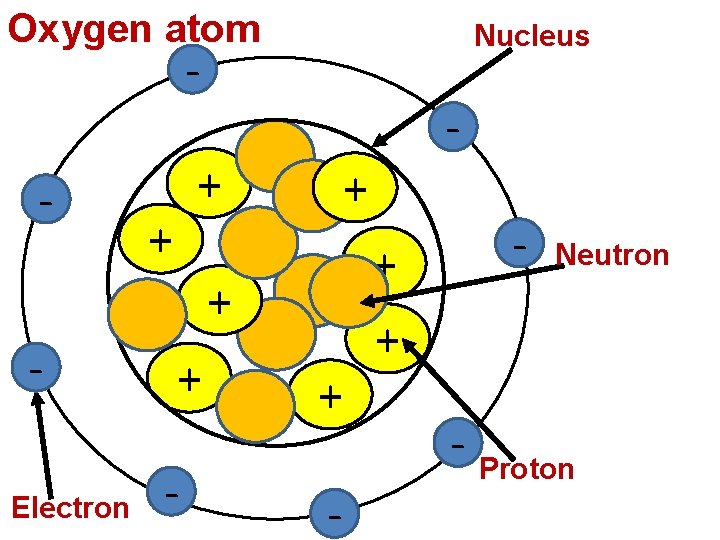
Oxygen atom Nucleus - - + + Electron + - - + + - Neutron - Proton
Elements • Made up of only ONE type of atom • CANNOT be broken down

26 29 79

28 47 6 17
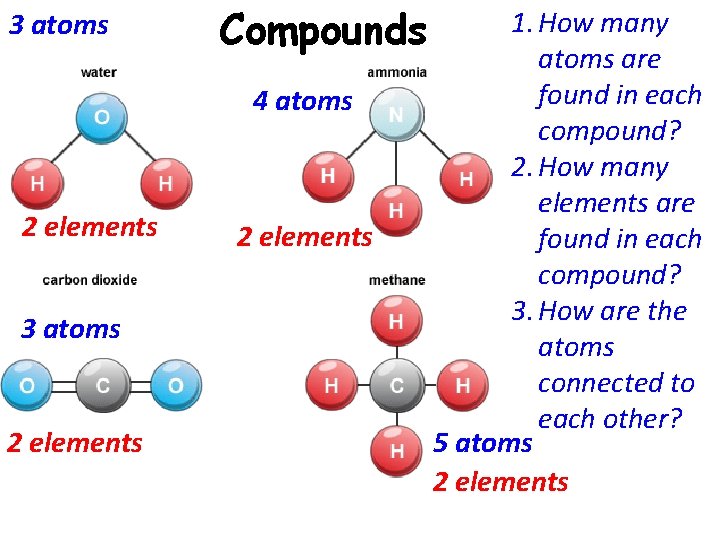
3 atoms 2 elements 1. How many Compounds atoms are found in each 4 atoms compound? 2. How many elements are 2 elements found in each compound? 3. How are the atoms connected to each other? 5 atoms 2 elements
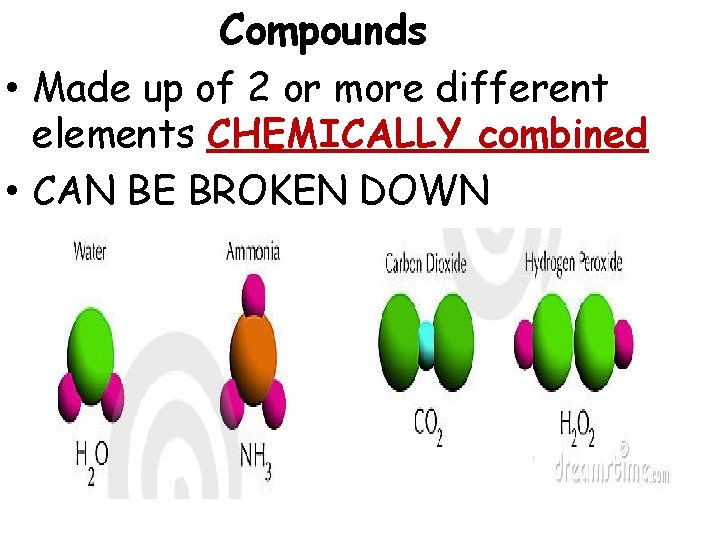
Compounds • Made up of 2 or more different elements CHEMICALLY combined • CAN BE BROKEN DOWN
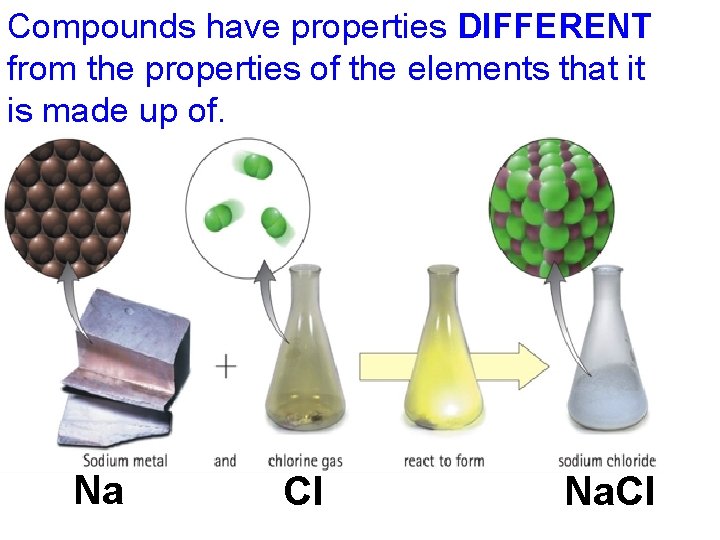
Compounds have properties DIFFERENT from the properties of the elements that it is made up of. • Compounds have properties DIFFERENT from the elements it is made up of Na Cl Na. Cl
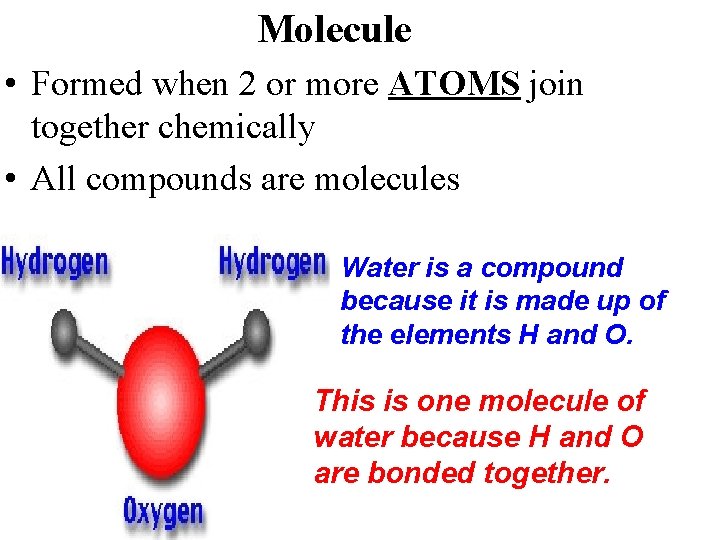
Molecule • Formed when 2 or more ATOMS join together chemically • All compounds are molecules Water is a compound because it is made up of the elements H and O. This is one molecule of water because H and O are bonded together.
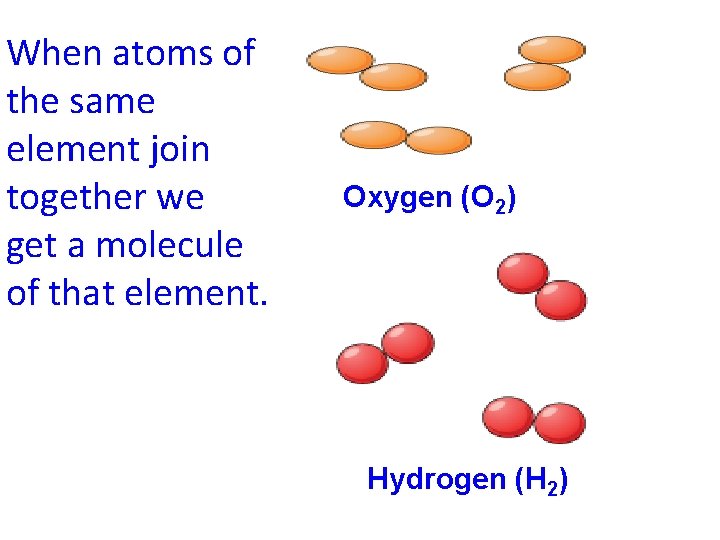
When atoms of the same element join together we get a molecule of that element. Oxygen (O 2) Hydrogen (H 2)
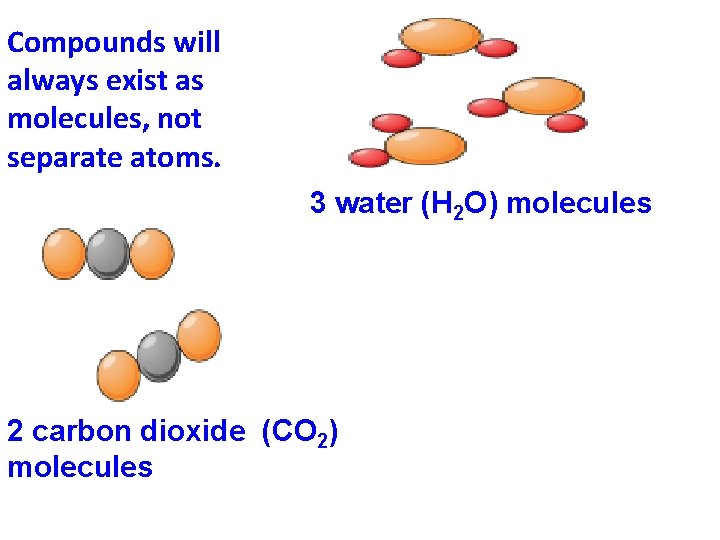
Compounds will always exist as molecules, not separate atoms. 3 water (H 2 O) molecules 2 carbon dioxide (CO 2) molecules

Ex: A glass of water is made up of millions of water molecules that are exactly the same
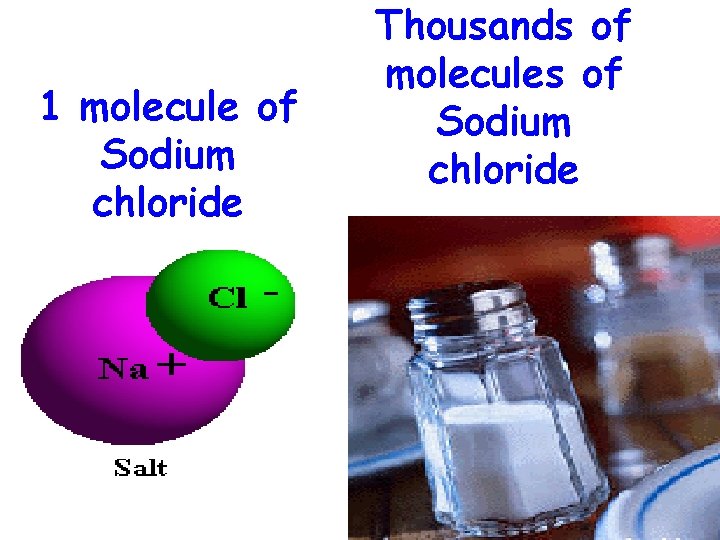
1 molecule of Sodium chloride Thousands of molecules of Sodium chloride

Let’s summarize: 1. Describe an element. 2. Describe a compound. 3. Describe the difference between an element and a compound. 4. Are all compounds molecules? Support your answer. 5. Identify a molecule that is not a compound.

Review: An object has a mass of 10 g and a volume of 2 cm 3. Identify the density of the object. D = M/V 10 g/2 cm 3 = 5 g/cm 3

Identify the instrument used to measure mass. 1. Thermometer 2. Graduated cylinder 3. Triple beam balance 4. Metric ruler

The SI units used when measuring mass are 1. milliliters 2. centimeters cubed 3. meters 4. grams

Identify the instrument used to measure temperature. 1. Thermometer 2. Graduated cylinder 3. Triple beam balance 4. Metric ruler

Identify the SI units used to measure temperature. 1. meter 2. degrees Celsius 3. grams 4. Degrees Fahrenheit

Identify the SI units used to measure the density of a liquid. 1. m. L 3 2. g/cm 3 3. cm 4. g/m. L
- Slides: 24