Topic Alkanes Do Now Draw the possible bonds

Topic: Alkanes Do Now: Draw the possible bonds a single carbon atom can have if it has a bonding capacity of 4

Alkanes • Homologous series of saturated hydrocarbons • Release energy when burned • Cn. H 2 n+2 – all have this general formula CH 4 C 2 H 6 H H–C–H H H–C–C–H H H
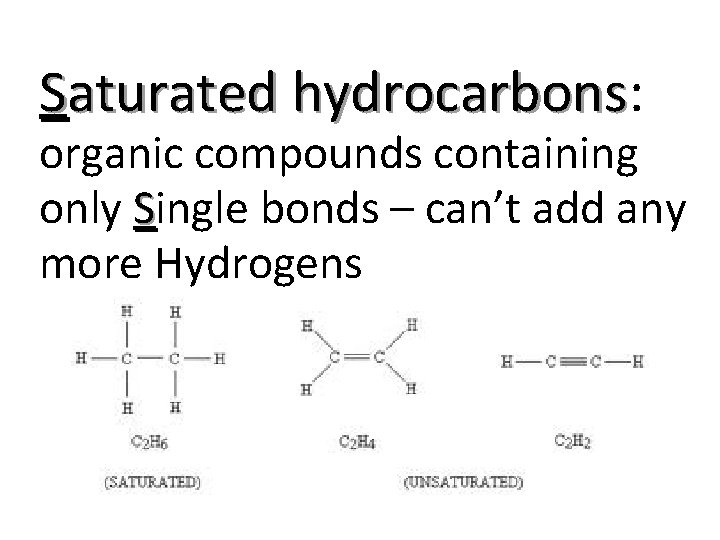
Saturated hydrocarbons: hydrocarbons organic compounds containing only Single bonds – can’t add any more Hydrogens
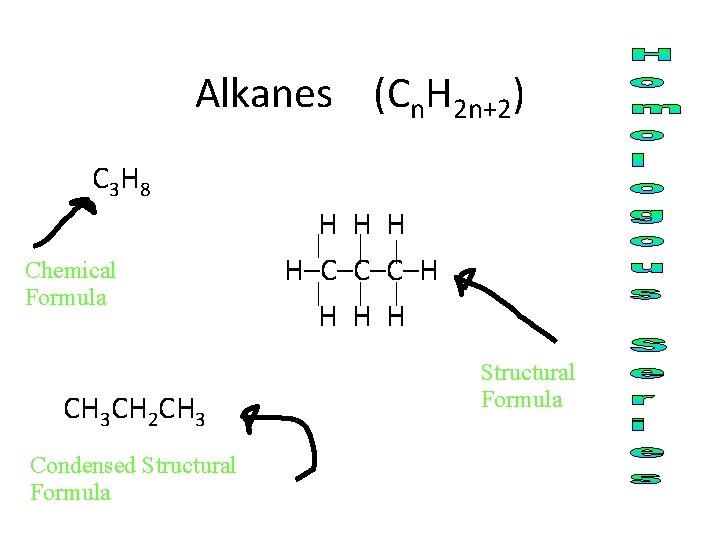
Alkanes (Cn. H 2 n+2) C 3 H 8 Chemical Formula CH 3 CH 2 CH 3 Condensed Structural Formula H H–C–C–C–H H Structural Formula
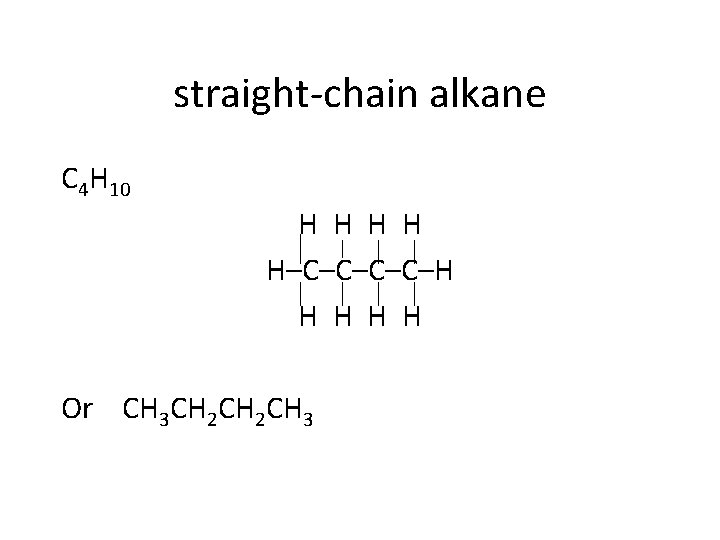
straight-chain alkane C 4 H 10 H H H–C–C–H H H Or CH 3 CH 2 CH 3

Naming straight-chain Alkanes • Name: describes molecule so can draw it • all alkanes ane have the suffix –ane • prefix: tells # of C’s
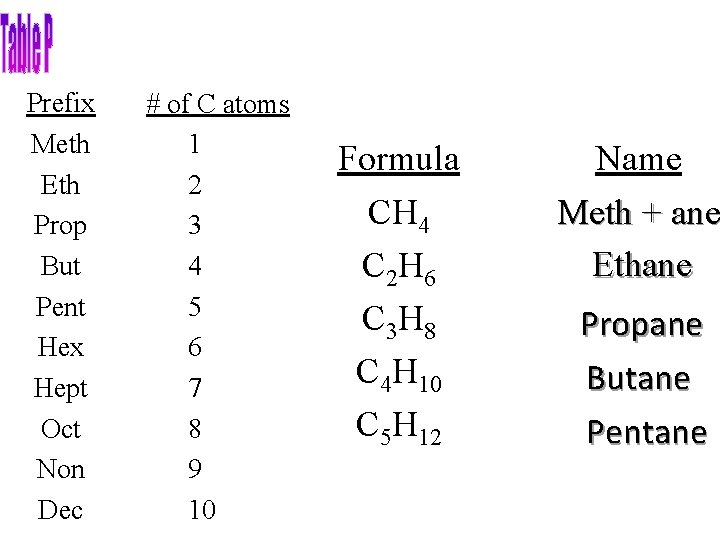
Prefix Meth Eth Prop But Pent Hex Hept Oct Non Dec # of C atoms 1 2 3 4 5 6 7 8 9 10 Formula CH 4 C 2 H 6 C 3 H 8 C 4 H 10 C 5 H 12 Name Meth + ane Ethane Propane Butane Pentane

Naming Branched-Chain Alkanes • goal of name is to describe molecule so you can draw it
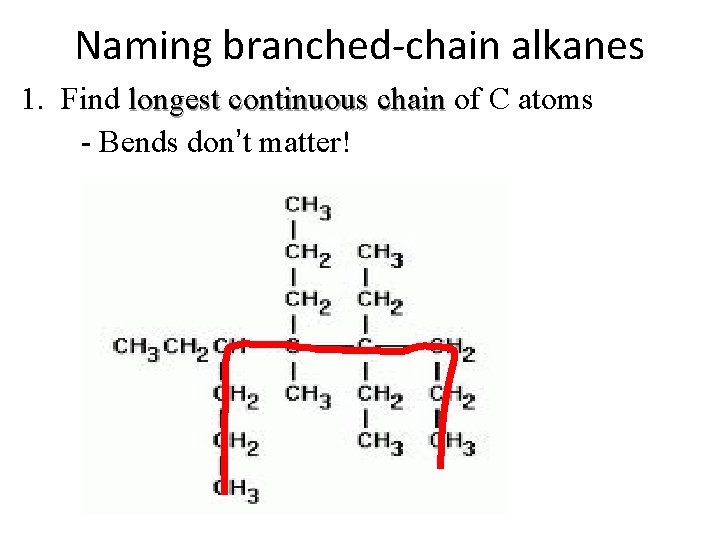
Naming branched-chain alkanes 1. Find longest continuous chain of C atoms - Bends don’t matter!
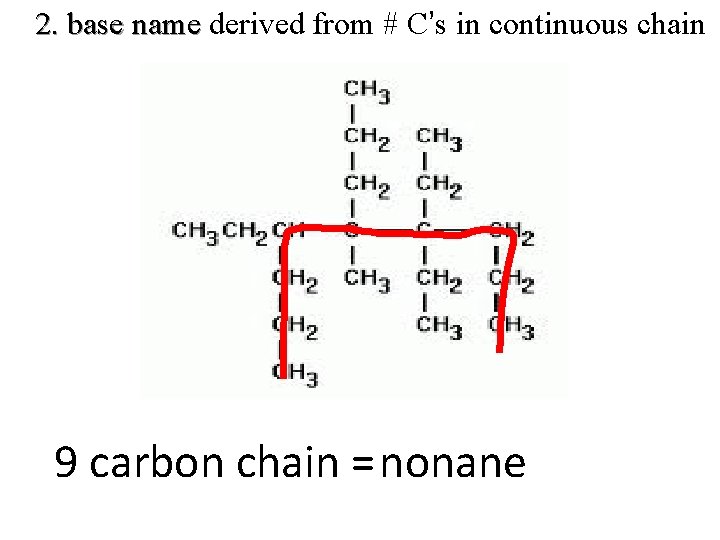
2. base name derived from # C’s in continuous chain 9 carbon chain = nonane
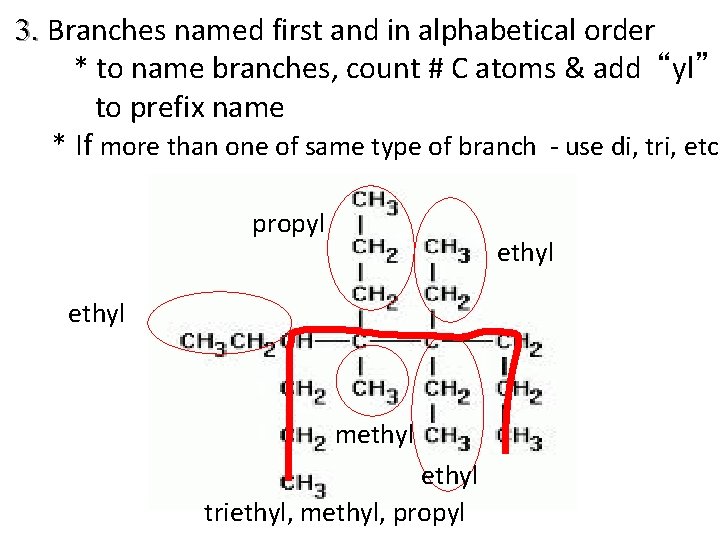
3. Branches named first and in alphabetical order * to name branches, count # C atoms & add “yl” to prefix name * If more than one of same type of branch - use di, tri, etc. propyl ethyl methyl triethyl, methyl, propyl
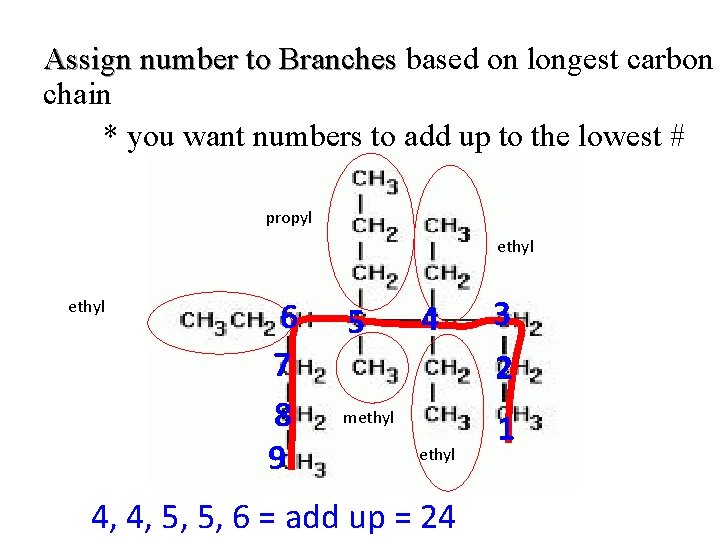
Assign number to Branches based on longest carbon chain * you want numbers to add up to the lowest # propyl ethyl 6 7 8 9 5 4 methyl 4, 4, 5, 5, 6 = add up = 24 3 2 1
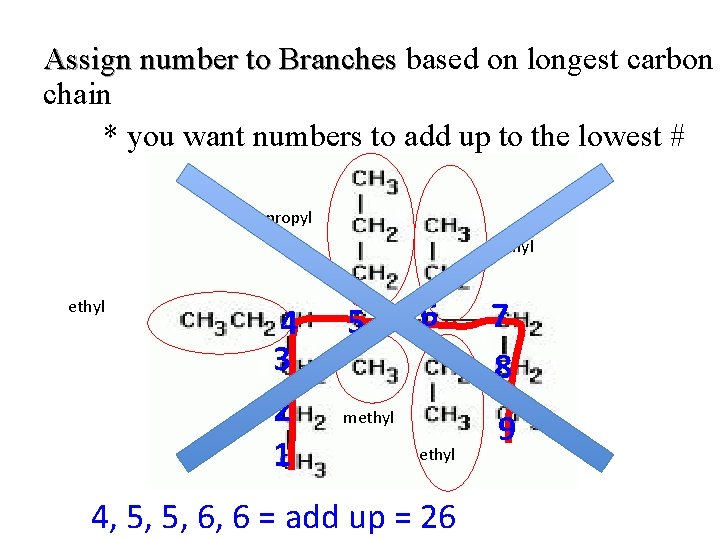
Assign number to Branches based on longest carbon chain * you want numbers to add up to the lowest # propyl ethyl 4 3 2 1 5 6 methyl 4, 5, 5, 6, 6 = add up = 26 7 8 9
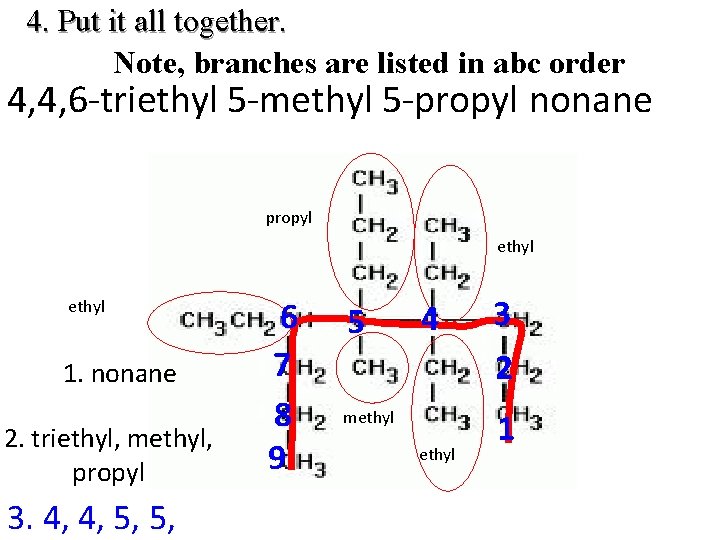
4. Put it all together. Note, branches are listed in abc order 4, 4, 6 -triethyl 5 -methyl 5 -propyl nonane propyl ethyl 1. nonane 2. triethyl, methyl, propyl 3. 4, 4, 5, 5, 6 7 8 9 5 4 methyl 3 2 1
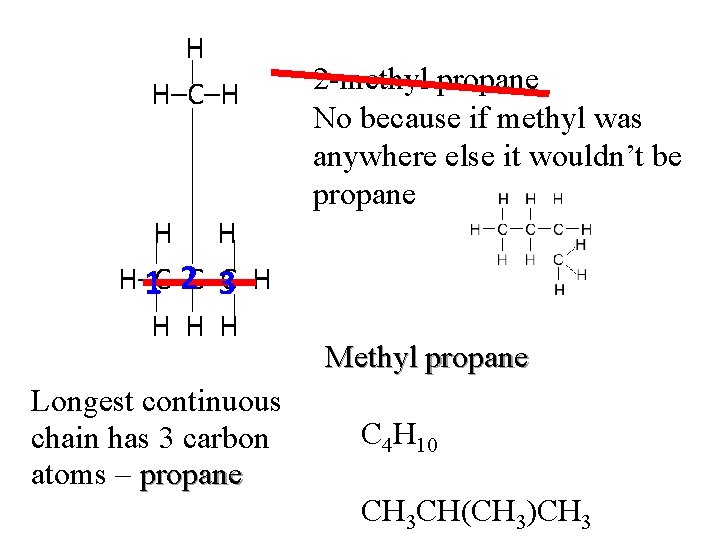
H H–C–H H H H–C–C–C–H 1 2 3 H H H Longest continuous chain has 3 carbon atoms – propane 2 -methyl propane No because if methyl was anywhere else it wouldn’t be propane Methyl propane C 4 H 10 CH 3 CH(CH 3)CH 3
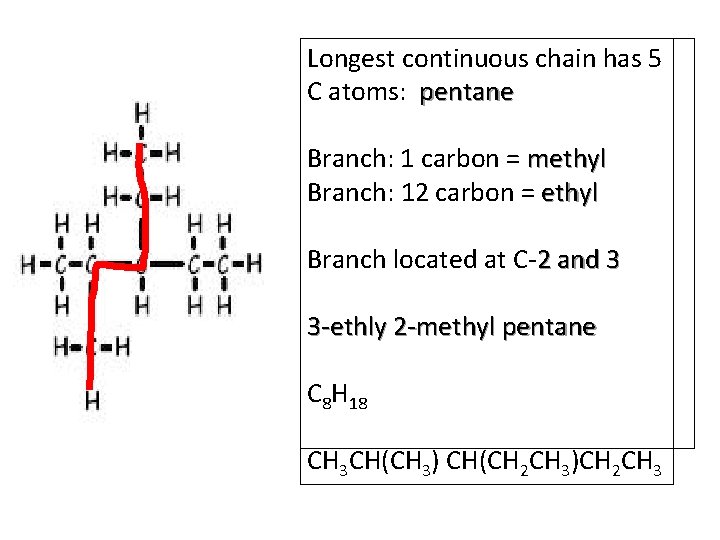
Longest continuous chain has 5 C atoms: pentane H H–C–H 6 H–C–H HH H 5 H–C–C–H H 4 H–C–H 3 2 H 1 Branch: 1 carbon = methyl Branch: 12 carbon = ethyl Branch located at C-2 and 3 3 -ethly 2 -methyl pentane C 8 H 18 CH 3 CH(CH 3) CH(CH 2 CH 3)CH 2 CH 3
- Slides: 16