Topic 7 Equilibrium 7 Dynamic Equilibrium 1 Equilibrium

Topic 7 - Equilibrium 7 – Dynamic Equilibrium 1

Equilibrium In a ‘traditional’ reaction, all the reactants get turned into products (assuming no excess) In an equilibrium reaction this is not the case, and the reaction sort of ‘stops’ part way through ‘TRADITIONAL ’ REACTANTS EQUILIBRIUM 1 PRODUCTS EQUILIBRIUM 2 EQUILIBRIUM 3

Writing equilibrium equations Equilibrium reactions are written using a double arrow Each of the arrows only has a single-sided head REACTANTS PRODUCTS The unit will make a lot of reference to the rate of the: ‘forward reaction’ (reactants becoming products) ‘back reaction’ (products becoming reactants)
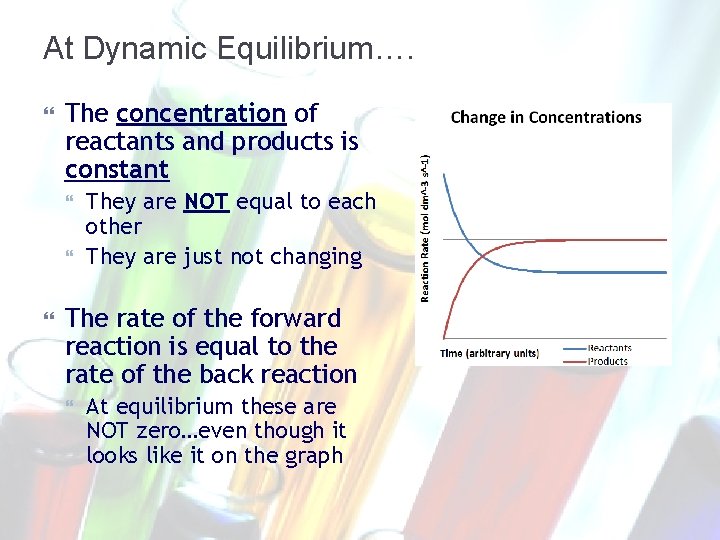
At Dynamic Equilibrium…. The concentration of reactants and products is constant They are NOT equal to each other They are just not changing The rate of the forward reaction is equal to the rate of the back reaction At equilibrium these are NOT zero…even though it looks like it on the graph

Why ‘Dynamic’ Equilibrium? The reaction hasn’t stopped, it is still going, but the rate of the forward and back reactions are equal, so there is no overall change. This is different to a static equilibrium (for example a seesaw) where there is no change at all.

Equilibrium Constant, Kc The equilibrium constant describes where the position of equilibrium lies: Minimum: 0, maximum: ∞, half-way: 1 Greater than 1: products favoured Less than 1: reactants favoured For the reaction: w. A + x. B ⇌ y. C + z. D

Dynamic Equilibrium Example: Calculate the value of the equilibrium constant, Kc, for the system shown, if 0. 1908 moles of CO 2, 0. 0908 moles of H 2, 0. 0092 moles of CO, and 0. 0092 moles of H 2 O vapor were present in a 2. 00 L reaction vessel were present at equilibrium. CO 2(g) + H 2(g) → CO(g) + H 2 O(g) Kc = [CO][H 2 O] [CO 2][H 2] [CO 2] = 0. 1908 mol CO 2/2. 00 L = 0. 0954 M [H 2] = 0. 0454 M CO] = 0. 0046 M [H 2 O] = 0. 0046 M Kc = [0. 0046] = 0. 0049 or 4. 9 x 10 3 [0. 0954][0. 0454]

Your Turn Example: Calculate the value of the equilibrium constant, Kc, for the reaction shown, if 1. 00 moles of NH 4 Cl, 0. 200 moles of NH 3, and 0. 400 moles of HCl vapor were present in a 5. 00 L reaction vessel were present at equilibrium. NH 4 Cl(g) → NH 3(g) + HCl(g)

Your Turn Example: Calculate the value of the equilibrium constant, Kc, for the reaction shown, if 1. 00 moles of NH 4 Cl, 0. 200 moles of NH 3, and 0. 400 moles of HCl vapor were present in a 5. 00 L reaction vessel were present at equilibrium. NH 4 Cl(g) → NH 3(g) + HCl(g) Solution: Kc = [NH 3][HCl] [NH 4 Cl] = 1. 00 mol NH 4 Cl/5. 00 L = 0. 200 M [NH 3] = 0. 0400 M [HCl] = 0. 0800 M Kc = [0. 0400][0. 0800] = 0. 0160 or 1. 60 x 10 2 [0. 200]

What if I Don’t Know the Equilibrium Concentrations? A mixture of 0. 100 mol of NO, 0. 050 mil of H 2, and 0. 10 mol of H 2 O is placed in a 1. 0 L vessel. The following equilibrium is established? 2 NO(g) + 2 H 2(g) N 2(g) + 2 H 2 O(g) At equilibrium [NO] = 0. 062 M. Calculate the equilibrium concentrations of H 2, N 2, and H 2 O.

Calculating Equilibrium A mixture of 0. 100 mol of NO, 0. 050 mil of H 2, and 0. 10 mol of H 2 O is placed in a 1. 0 L vessel. The following equilibrium is established? 2 NO(g) + 2 H 2(g) N 2(g) + 2 H 2 O(g) At equilibrium [NO] = 0. 062 M. Calculate the equilibrium concentrations of H 2, N 2, and H 2 O. 2 NO Initial Change Equilibrium 2 H 2 N 2 2 H 2 O

Calculating Equilibrium A mixture of 0. 100 mol of NO, 0. 050 mil of H 2, and 0. 10 mol of H 2 O is placed in a 1. 0 L vessel. The following equilibrium is established? 2 NO(g) + 2 H 2(g) N 2(g) + 2 H 2 O(g) At equilibrium [NO] = 0. 062 M. Calculate the equilibrium concentrations of H 2, N 2, and H 2 O. Initial 2 NO 2 H 2 N 2 2 H 2 O 0. 100 M 0. 050 M 0 0. 10 M Change Equilibrium 0. 062 M

Calculating Equilibrium A mixture of 0. 100 mol of NO, 0. 050 mil of H 2, and 0. 10 mol of H 2 O is placed in a 1. 0 L vessel. The following equilibrium is established? 2 NO(g) + 2 H 2(g) N 2(g) + 2 H 2 O(g) At equilibrium [NO] = 0. 062 M. Calculate the equilibrium concentrations of H 2, N 2, and H 2 O. 2 NO 2 H 2 N 2 2 H 2 O Initial 0. 100 M 0. 050 M 0 0. 10 M Change 0. 038 M Equilibrium 0. 062 M

Calculating Equilibrium A mixture of 0. 200 mol of NO, 0. 100 mil of H 2, and 0. 20 mol of H 2 O is placed in a 2. 0 L vessel. The following equilibrium is established? 2 NO(g) + 2 H 2(g) N 2(g) + 2 H 2 O(g) At equilibrium [NO] = 0. 062 M. Calculate the equilibrium concentrations of H 2, N 2, and H 2 O. 2 NO 2 H 2 N 2 2 H 2 O Initial 0. 100 M 0. 050 M 0 0. 10 M Change 0. 038 M + 0. 019 M + 0. 038 M Equilibrium 0. 062 M 0. 019 M 0. 138 M

Your Turn Given the reaction: H 2 + I 2 2 HI Calculate all three equilibrium concentrations when [H 2] = [I 2] = 0. 200 M initially and =. 0200 M at equilibrium. H 2 Initial Change Equilibrium I 2 2 HI 0

Calculating Equilibrium Given the reaction: 2 HI H 2 + I 2 For the system, if we start with 0. 010 mol/L of H 2 and I 2 and 0. 096 mol/L of HI, what are their concentrations at equilibrium given that Keq = 0. 016? 2 HI Initial Change Equilibrium I 2 H 2

How are Kc and Kp related? Start with ideal gas law PV = n. RT Rearranging gives P = n (RT) V Substituting for molar concentrations into Kc results in pressure-based formula Δn = moles of gas in products moles of gas in reactants Kp = Kc(RT) Kp = Kc when n = 0 n

Homework Open the simulation here: https: //phet. colorado. edu/en/simulation/reactions-and-rates Using the ‘Investigate the conditions under which a significant equilibrium is more and less likely to form You will need to use the ‘Rate Experiments’ tab and select ‘Design you own’ for the reaction. Under ‘chart options’, select ‘bar chart’. You should change: Enthalpy level of reactants Enthalpy level of products Activation energy Temperature Concentrations of reactants and products You should find a systematic way to record your findings, and should try to reach some kind of conclusion. Complete

Key Points: At dynamic equilibrium: Concentrations of reactants and products are constant The rate of the forward and backward reactions are equal The position of an equilibrium is given by Kc > 1 means products are favoured Kc < 1 means reactants are favoured
- Slides: 19