TOPIC 4 BONDING 4 2 COVALENT BONDING Topic
TOPIC 4: BONDING 4. 2: COVALENT BONDING

Topic 4: Bonding 4. 2: Covalent bonding Essential Idea: Covalent compounds form by the sharing of electrons. Nature of Science: Looking for trends and discrepancies – compounds that contain non-metals have different properties from compounds that contain non-metals. (2. 5) Use theories to explain natural phenomena – Lewis introduced a class of compounds which share electrons. Pauling used the idea of electronegativity to explain unequal sharing of electrons (2. 2)

IB Topic 4: Bonding 4. 2: Covalent bonding Understandings: 1. A covalent bond is formed by the electrostatic attraction between a shared pair of electrons and the positively charged nuclei. 2. Single, double, and triple covalent bonds involve one, two, and three shared pairs of electrons, respectively. 3. Bond length decreases and bond strength increases as the number of shared electrons increases. 4. Bond polarity results from the difference in electronegativities of the bonded atoms.

IB Topic 4: Bonding 4. 2: Covalent bonding Applications and Skills: 1. Deduction of the polar nature of a covalent bond from electronegativity values

IB Topic 4: Bonding Ionic Bond: Transfer of electrons from metal to non-metal; bond formed by electrostatic attraction between oppositely charged ions. E. N. difference >1. 8 Covalent Bond: Sharing of electrons between non-metals

4. 2 U 1 Covalent Bond Covalent bonds occur between nonmetals since both want to gain electrons. They share electrons to achieve a noble gas (stable) configuration (8 valence e-).

4. 2 U 1 Covalent Bond The electrons in the bond are electrostatically attracted by both nuclei, so that it forms a directional bond between the two atoms.
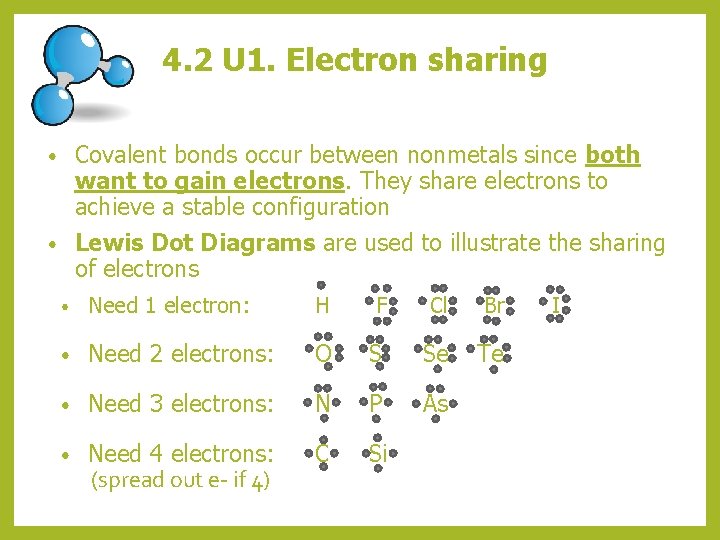
4. 2 U 1. Electron sharing Covalent bonds occur between nonmetals since both want to gain electrons. They share electrons to achieve a stable configuration • Lewis Dot Diagrams are used to illustrate the sharing of electrons • • Need 1 electron: H F Cl Br • Need 2 electrons: O S Se Te • Need 3 electrons: N P As • Need 4 electrons: (spread out e- if 4) C Si I
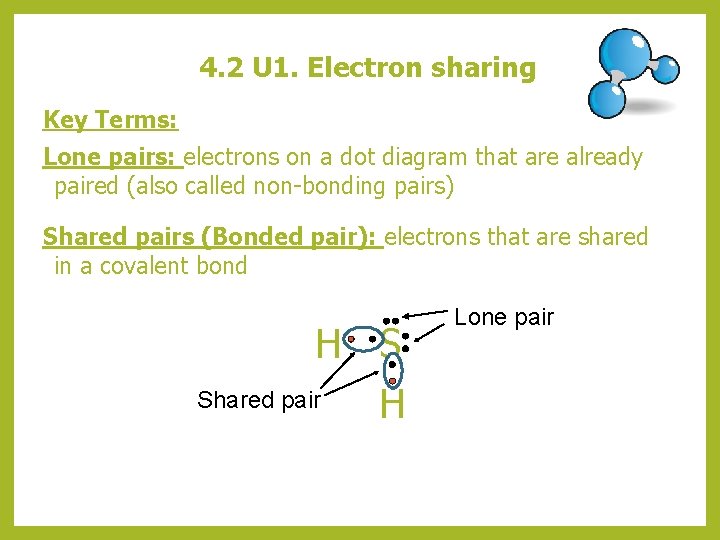
4. 2 U 1. Electron sharing Key Terms: Lone pairs: electrons on a dot diagram that are already paired (also called non-bonding pairs) Shared pairs (Bonded pair): electrons that are shared in a covalent bond H S Shared pair H Lone pair
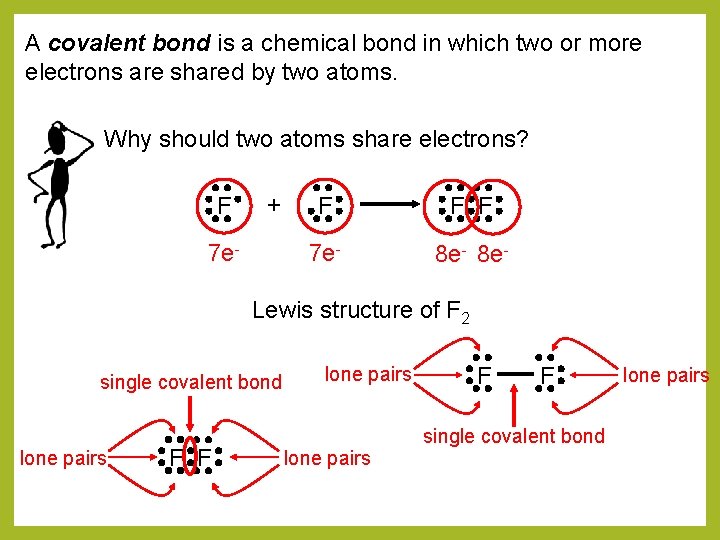
A covalent bond is a chemical bond in which two or more electrons are shared by two atoms. Why should two atoms share electrons? F + 7 e- F F F 7 e- 8 e- Lewis structure of F 2 single covalent bond lone pairs F F single covalent bond lone pairs
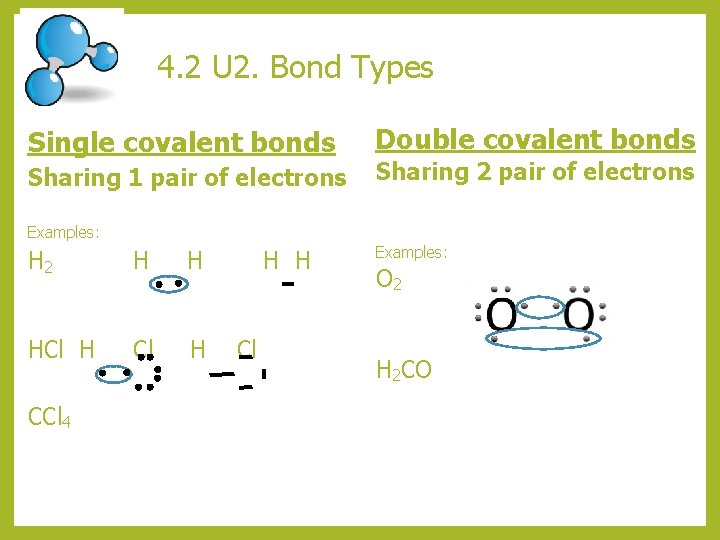
4. 2 U 2. Bond Types Single covalent bonds Sharing 1 pair of electrons Double covalent bonds Sharing 2 pair of electrons Examples: H 2 H H HCl H CCl 4 H H Cl Examples: O 2 H 2 CO

4. 2 U 2. Bond Types Triple covalent bonds Sharing 3 pair of electrons Examples: N 2 CH 2 N N
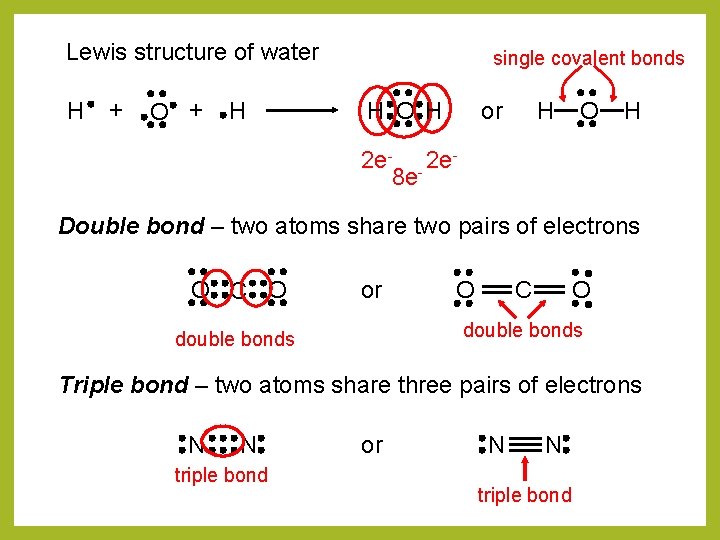
Lewis structure of water H + O + H single covalent bonds H O H or H O H 2 e- - 2 e 8 e Double bond – two atoms share two pairs of electrons O C O or O O C double bonds Triple bond – two atoms share three pairs of electrons N N triple bond or N N triple bond
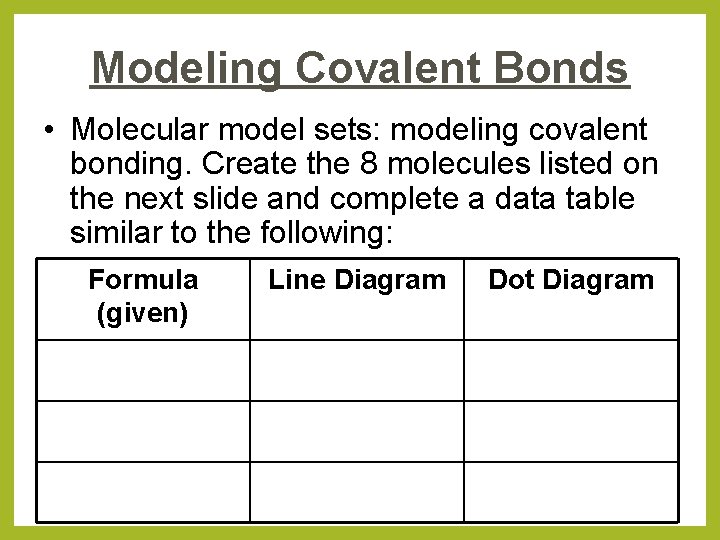
Modeling Covalent Bonds • Molecular model sets: modeling covalent bonding. Create the 8 molecules listed on the next slide and complete a data table similar to the following: Formula (given) Line Diagram Dot Diagram

Modeling Covalent Bonds • • • 1 bond = 2 shared electrons Red = oxygen Black = carbon White = hydrogen Blue = nitrogen (3 bonding sites + 1 “hat”) Green & Silver = halogens H 2 O ● H 2 C 2 H 4 ● O 2 CO 2 ● C 6 H 12 NH 3 ● N 2
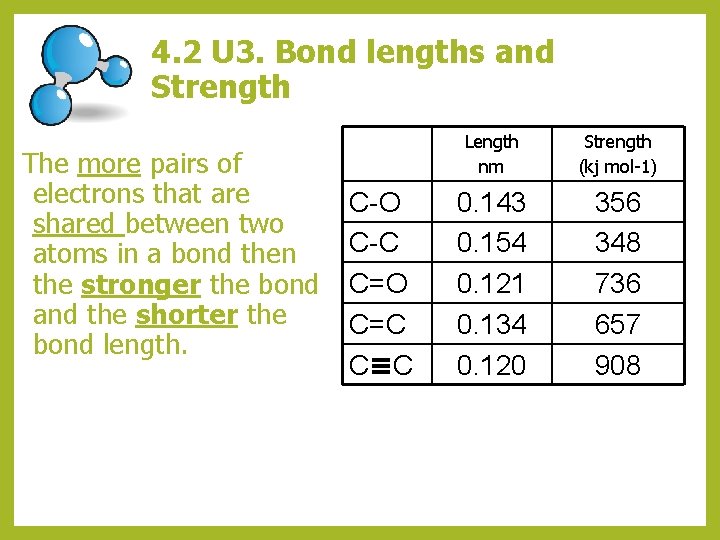
4. 2 U 3. Bond lengths and Strength The more pairs of electrons that are shared between two atoms in a bond then the stronger the bond and the shorter the bond length. C-O C-C C=O C=C C C Length nm Strength (kj mol-1) 0. 143 0. 154 0. 121 0. 134 0. 120 356 348 736 657 908

4. 2 U 3. Bond lengths and Strength Bond Lengths Triple bond < Double Bond < Single Bond shortest shorter longer

4. 2 U 4. Electronegativity Difference Nonpolar covalent bonds have electronegativity difference of 0 Polar covalent bonds have electronegativity differences above 0 less than or equal to 1. 8 Ionic bonds have electronegativity differences greater than 1. 8
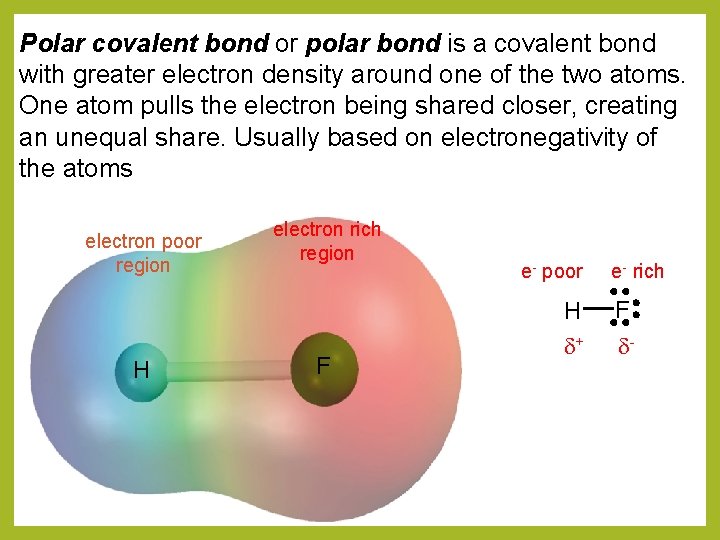
Polar covalent bond or polar bond is a covalent bond with greater electron density around one of the two atoms. One atom pulls the electron being shared closer, creating an unequal share. Usually based on electronegativity of the atoms electron poor region H electron rich region F e- poor H d+ e- rich F d-

Classification of bonds by difference in electronegativity Difference Bond Type ≤ 1. 8 Covalent > 1. 8 0 Ionic (Nonpolar) Pure Covalent 0. 1 to less than 1. 8 Polar Covalent Increasing difference in electronegativity Covalent Polar Covalent share e- Unequal sharing of e- Ionic transfer e-

Molecular (Covalent) Naming for two nonmetals • Prefix System (binary compounds) 1. Less electronegative atom comes first. 2. Add prefixes to indicate # of atoms. Omit mono- prefix on the 1 st element. Mono- is REQUIRED on the 2 nd element. 3. Change the ending of the 2 nd element to -ide.
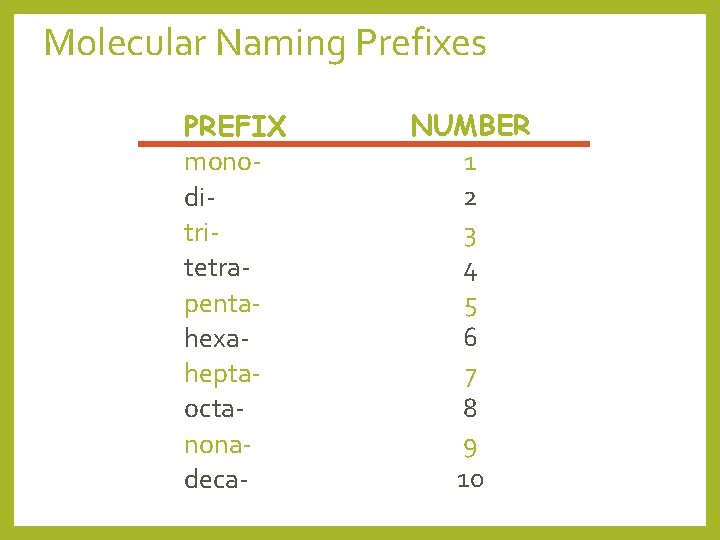
Molecular Naming Prefixes PREFIX monoditritetrapentahexaheptaoctanonadeca- NUMBER 1 2 3 4 5 6 7 8 9 10
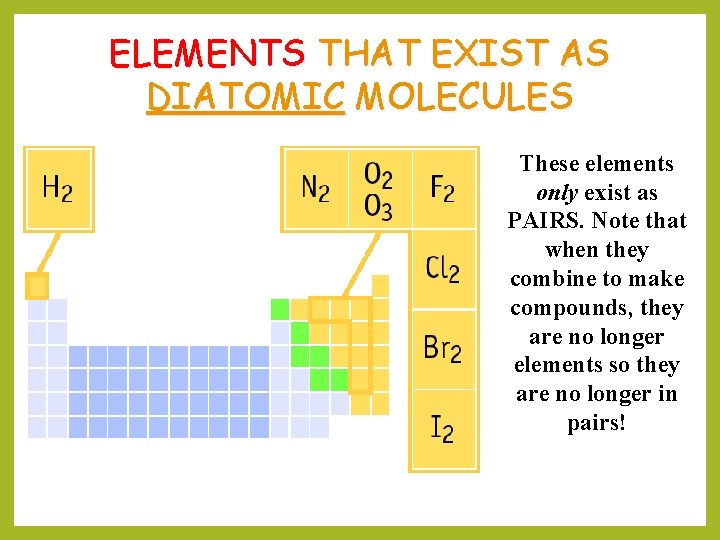
ELEMENTS THAT EXIST AS DIATOMIC MOLECULES These elements only exist as PAIRS. Note that when they combine to make compounds, they are no longer elements so they are no longer in pairs!

Molecular Naming: Examples • CCl 4 • carbon tetrachloride • N 2 O • dinitrogen monoxide • SF 6 • sulfur hexafluoride

Molecular Formula Examples • arsenic trichloride • As. Cl 3 • dinitrogen pentoxide • N 2 O 5 • tetraphosphorus decoxide • P 4 O 10

Practice 1 Fill in the blanks to complete the following names of covalent compounds. CO carbon ______oxide CO 2 carbon ________ PCl 3 phosphorus _______chloride CCl 4 carbon ____chloride N 2 O _____nitrogen _____oxide

Practice 1 Fill in the blanks to complete the following names of covalent compounds. CO carbon monoxide CO 2 carbon dioxide PCl 3 phosphorus trichloride CCl 4 carbon tetrachloride N 2 O dinitrogen monoxide

Practice 2 1. P 2 O 5 a) phosphorus oxide b) phosphorus pentoxide c) diphosphorus pentoxide 2. Cl 2 O 7 a) dichlorine heptoxide b) dichlorine oxide c) chlorine heptoxide 3. Cl 2 a) chlorine b) dichlorine c) dichloride
Learning Check 1. P 2 O 5 a) phosphorus oxide b) phosphorus pentoxide c) diphosphorus pentoxide 2. Cl 2 O 7 a) dichlorine heptoxide b) dichlorine oxide c) chlorine heptoxide 3. Cl 2 a) chlorine b) dichlorine c) dichloride

Covalent Bond Physical Properties Covalent Compounds: • MP: low • BP: low • Volatility: may be volatile • Conductivity: do not conduct electricity • Solubility: typically insoluble in water • Form molecules
- Slides: 30