Topic 4 7 Types of Chemical Reactions References





















- Slides: 27

Topic 4. 7: Types of Chemical Reactions References: Text Ch 4. 2, 4. 3, 4. 4 Notes for Topic 4. 2: Net Ionic Equations Notes for Topic 4. 1: Introduction to Reactions

Objective: Identify a reaction as acid-base, oxidationreduction, or precipitation 1. Acid-base reactions involve a transfer of one or more protons between species. 2. Oxidation-reduction (redox) reactions involve the transfer of electrons between chemical species as indicated by the change of oxidation numbers. 3. In a redox reaction, electrons are transferred from the species that is oxidized to the species that is reduced 4. Oxidation numbers assigned to reactants and products identify the oxidized and reduced species. 5. Precipitation reactions involve mixing ions in aqueous solution to produce and insoluble or sparingly soluble compound. All sodium, potassium, ammonium, and nitrate salts are soluble in water (know for AP test).

4. 1 General Properties of Aqueous Solutions • ______ are homogeneous mixtures of two or more pure substances. • The substance present in the greatest quantity is the _____. • The other substances present are called _____. They are said to be dissolved in the solvent. • Solute + solvent = solution • Ex: salt dissolved in water 3

Electrolytes and Nonelectrolytes • An ________ is a substance that dissociates into ions when dissolved in water. • Electrolyte solutions conduct electricity. • A _________ (sugar, for example) may dissolve in water, but it does not dissociate into ions when it does so. • Nonelectrolyte solutions do not conduct electricity. 4

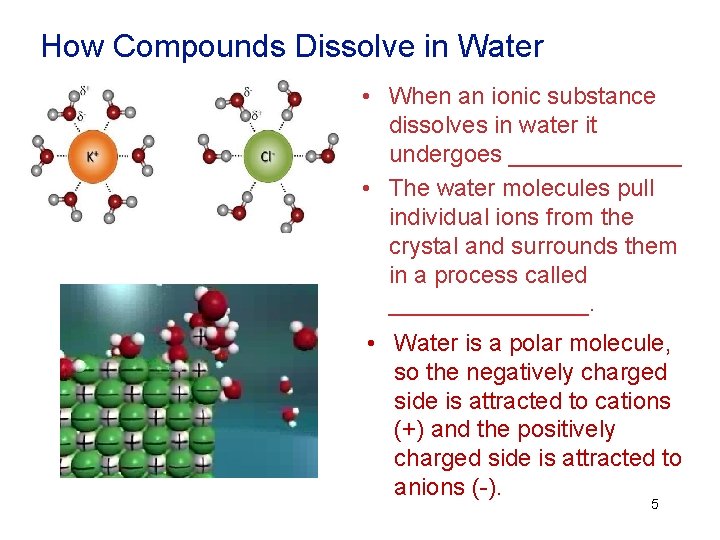
How Compounds Dissolve in Water • When an ionic substance dissolves in water it undergoes _______ • The water molecules pull individual ions from the crystal and surrounds them in a process called ________. • Water is a polar molecule, so the negatively charged side is attracted to cations (+) and the positively charged side is attracted to anions (-). 5

How Molecular Compounds Dissolve in Water • When a molecular compounds dissolves in water (such as methanol) the intact molecules disperse throughout the solution. • Acids are an exception. When they dissolve in water they ionize. 6

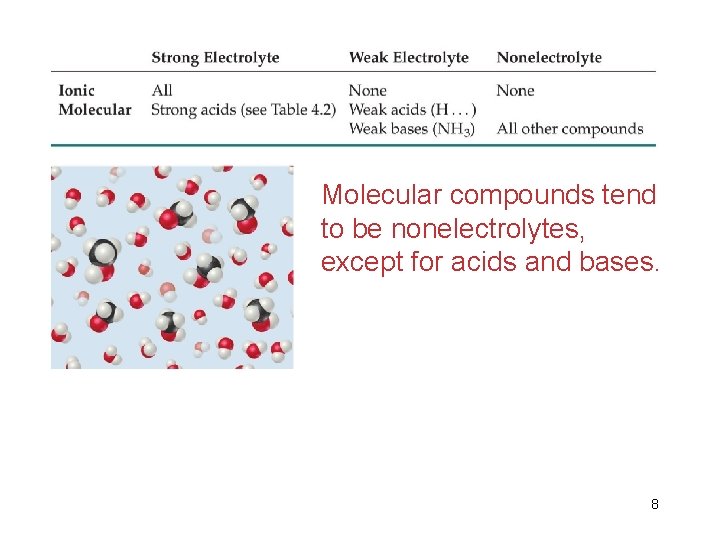
Strong and Weak Electrolytes • Soluble ionic compounds tend to be strong electrolytes because they completely or mostly dissolve in water. • Ex: • Weak electrolytes, such as weak acids, only partially dissolve in water. • Ex: 7

Molecular compounds tend to be nonelectrolytes, except for acids and bases. 8

4. 2 Precipitation Reactions • See Topics 4. 1 and 4. 2 9

4. 3 Acids, Bases, and Neutralization Reactions • Acids are: – Substances that increase the concentration of H+ (hydrogen ions) when dissolved in water (Arrhenius). – Proton donors (Brønsted–Lowry). – There are only seven strong acids: Hydrochloric (HCl), Hydrobromic (HBr), Hydroiodic (HI), Nitric (HNO 3), Sulfuric (H 2 SO 4), Chloric (HCl. O 3), Perchloric (HCl. O 4) 10

• Some acids are monoprotic, such as HCl and HNO 3. They yield just one H+ per molecule of acid: HCl(aq) H+(aq) + Cl-(aq) • Other acids are diprotic, such as H 2 SO 4. They yield two H+ per molecule of acid. The ionization of a diprotic acid occurs in two steps: H 2 SO 4(aq) H+(aq) + HSO 4 -(aq) H+(aq) + SO 42 -(aq) • Strong acids completely dissociate in water; weak acids only partially ionize. 11

• Bases are: – Substances that increase the concentration of OH− (hydroxide ions) when dissolved in water (Arrhenius). – Proton acceptors (Brønsted–Lowry). • The strong bases are the hydroxides of the following metals: – Alkali metals – Calcium – Strontium – Barium • Most other hydroxides are insoluble in water. • A common weak base is NH 3 (ammonia), which reacts with water to form OH- ions: NH 3(aq) + H 2 O(l) NH 4+(aq) + OH-(aq) 12

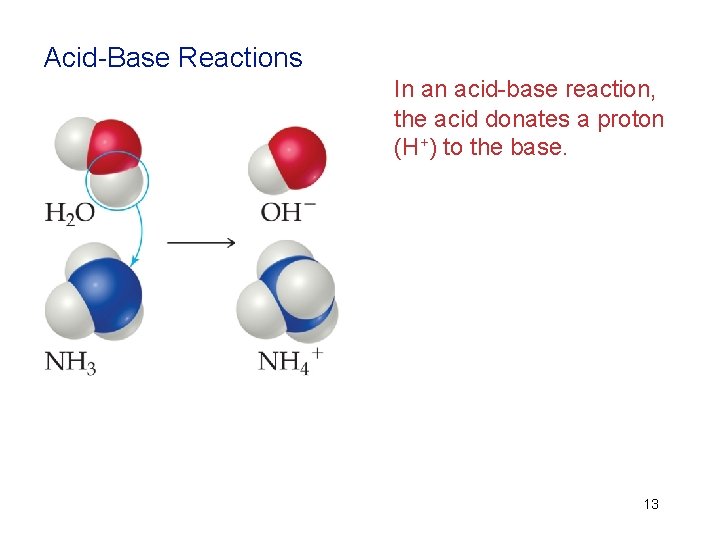
Acid-Base Reactions In an acid-base reaction, the acid donates a proton (H+) to the base. 13

Neutralization Reactions and Salts • Generally, when solutions of a strong acid and a strong base are combined, the products are a dissolved salt and water. HCl (aq) + Na. OH (aq) Na. Cl (aq) + H 2 O (l) • The complete ionic equation is H+ (aq) + Cl- (aq) + Na+ (aq) + OH-(aq) Na+ (aq) + Cl- (aq) + H 2 O (l) • The net ionic equation is H+ (aq) + OH- (aq) + H 2 O (l) 14

Observe the reaction between acetic acid and Ba(OH)2 15

Neutralization Reactions with Gas Formation • Some metathesis reactions do not give the product expected. • The expected product decomposes to give a gaseous product (CO 2 or SO 2). Ca. CO 3 (s) + HCl (aq) Ca. Cl 2 (aq) + CO 2 (g) + H 2 O (l) Na. HCO 3 (aq) + HBr (aq) Na. Br (aq) + CO 2 (g) + H 2 O (l) Sr. SO 3 (s) + 2 HI (aq) Sr. I 2 (aq) + SO 2 (g) + H 2 O (l) 16

4. 4 Oxidation-Reduction Reactions Oxidation and Reduction • An oxidation occurs when an atom or ion loses electrons. • A reduction occurs when an atom or ion gains electrons. 17

• One cannot occur without the other. When one substance is oxidized another is reduced, or vice-versa. • Oxidation-reduction reactions are referred to as redox reactions, for short. 18

Oxidation Numbers • To determine if an oxidation-reduction reaction has occurred, we assign an oxidation number to each element in a neutral compound or charged entity. • See rules for assigning oxidation numbers in text, page 139. You must remember these rules. • Oxidation numbers are written with the sign first, then the number. For example, O is -2, Ca is +2 • The oxidation numbers for a neutral compound must add up to zero. • The oxidation numbers for an ionic compound must add up to the charge on the ion. • Remember OIL RIG for what is oxidized and reduced: Oxidation is loss of electrons; reduction is gain of electrons 19

Assigning Oxidation Numbers 20

Oxidation of Metals by Acids and Salts • There are many kinds of redox reactions. – Ex: Combustion reactions where elemental oxygen is converted to compounds of oxygen. – Other types of redox reactions are discussed in Ch 20. • The reaction of a metal with either an acid or a metal salt is called a displacement reaction and conformed to the general formula: A + BX AX + B 21

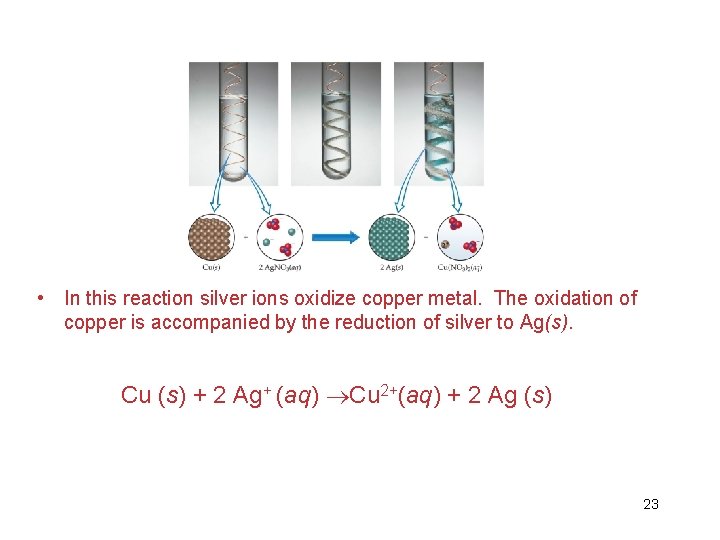
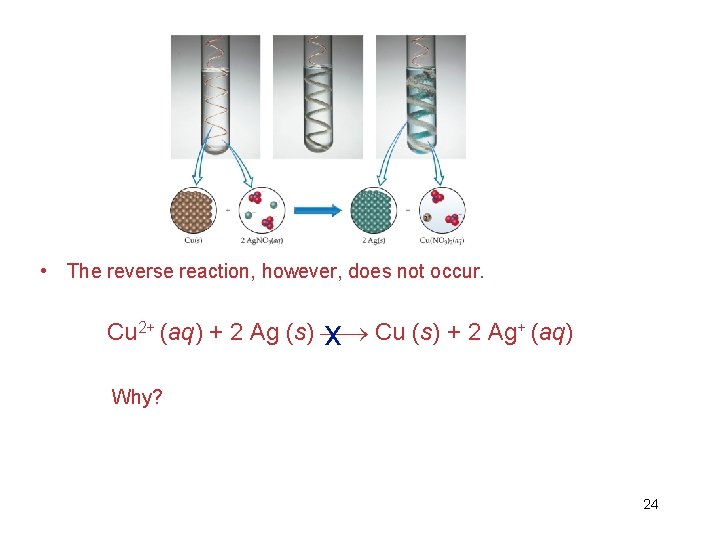
• In displacement reactions (single displacement reactions), ions in solution are displaced through oxidation of another element. • The ions that caused the oxidation are then reduced. • In the case of copper reacting with silver ions in solution, the copper metal is placed in solution and silver metal is formed as a solid. See figure 4. 14. 22

• In this reaction silver ions oxidize copper metal. The oxidation of copper is accompanied by the reduction of silver to Ag(s). Cu (s) + 2 Ag+ (aq) Cu 2+(aq) + 2 Ag (s) 23

• The reverse reaction, however, does not occur. Cu 2+ (aq) + 2 Ag (s) x Cu (s) + 2 Ag+ (aq) Why? 24

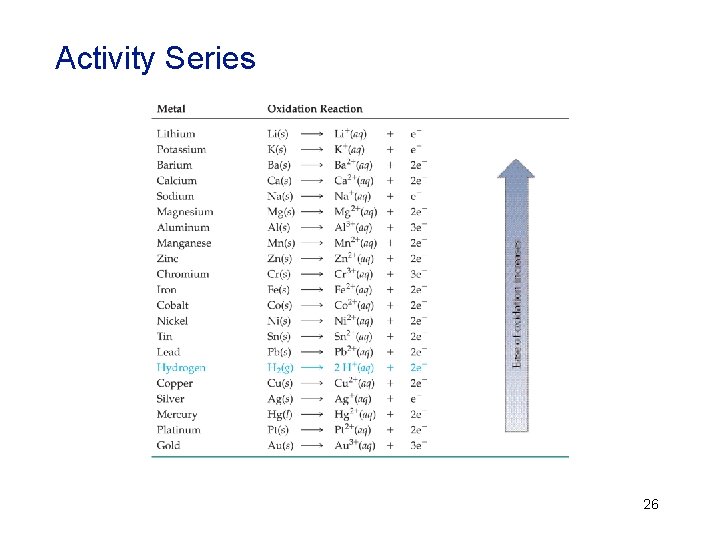
• The reactivity of metals is due to the difference in stability of their electron configurations as atoms and as ions. All metals will form positive ions when they react. • Reference the activity series table on the following slide or in the text. In it you will see that copper lies above silver in ease of oxidation. Since copper is already oxidized, as Cu+, silver cannot replace it since it is not as easily oxidized. • Metals in the activity series replace, or oxidize, metals below them in the series. 25

Activity Series 26

Oxidation-Reduction equations • Iron is oxidized by a solution of gold nitrate Fe + Au(NO 3)3 Fe(NO 3)3 + Au 27