Topic 3 Acidic Basic Neutral Substances p H

Topic 3: Acidic, Basic & Neutral Substances
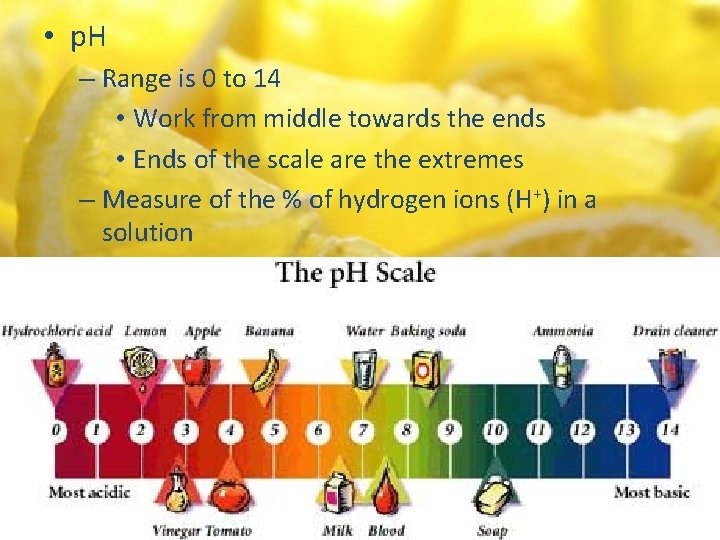
• p. H – Range is 0 to 14 • Work from middle towards the ends • Ends of the scale are the extremes – Measure of the % of hydrogen ions (H+) in a solution
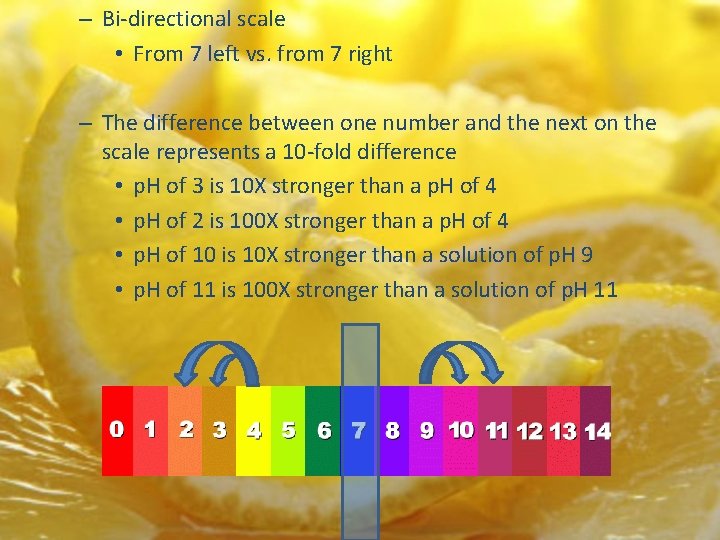
– Bi-directional scale • From 7 left vs. from 7 right – The difference between one number and the next on the scale represents a 10 -fold difference • p. H of 3 is 10 X stronger than a p. H of 4 • p. H of 2 is 100 X stronger than a p. H of 4 • p. H of 10 is 10 X stronger than a solution of p. H 9 • p. H of 11 is 100 X stronger than a solution of p. H 11

• Acid – Sharp / sour taste – Solution with a p. H lower than 7 • Base (Alkali) – Slippery / bitter – Solution with a p. H higher than 7 • Neutral – Neither an acid or base – p. H of 7. 0

– Measuring p. H • p. H meter – digital probe • Indicators – Chemical compound that will visually show the p. H of a solution

– Litmus paper » Red or blue colored paper that reacts with acids and bases

– Litmus paper » If the solution is acidic then the litmus paper turns/stays red » If the solution is basic then the litmus paper turns/stays blue » If the solution is neutral then red litmus stays red, blue litmus stays blue

• Which of the above liquids are acidic? Basic? Neutral?

• Neutralization – Reaction between acid + base that produces water + salt – After reaction, solution will have a neutral p. H – HCl (aq) + Na. OH (aq) → Na. Cl (s) + H 2 O (l) acid base salt water – Examples • Taking an antacid for an upset stomach – Antacid = mild base, stomach = acidic

• Acid Rain – Normal rainwater is naturally slightly acidic • Raindrops dissolve CO 2 from the air to form a very weak acid called carbonic acid • p. H of 5. 6 – Industrial processes and fuel combustion produce large quantities of CO 2, SO 2, and NO gases into the air (pollution) • p. H less than 5. 6 – These chemical dissolve in water to form acids • CO 2 → carbonic acids • SO 2 → sulfuric acids • NO → nitric acids


– Concerns • Can cause bodies of water to become acidic – Most organisms can only survive in a narrow p. H range – What would a graphical representation of this look like?


– Acidic lakes can be treated with lime (Ca. OH = calcium hydroxide) • Ca. OH = base • Results in neutralization • Ca(OH)2(aq) + H 2 SO 4(aq) → Ca. SO 4(aq) + H 2 O(l) base acid salt water

Laboratory Assignment: An Indication of p. H

• Online p. H Labs – http: //www. bgfl. org/bgfl/custom/resources_ftp/client_ftp/ks 3/science/acids/ – http: //sv. berkeley. edu/showcase/flash/juicebar. html – http: //www. miamisci. org/ph/phpanel. html
- Slides: 16