Topic 2 Molecular Biology 2 2 Water Source

Topic 2 Molecular Biology 2. 2 Water (Source: Biology, Course Companion. 2014 Edition, Allot and Mindorff, Oxford University Press)

Understanding: Water molecules are polar and hydrogen bonds form between them. �Students: add questions here (at home) �Within water molecule = polar covalent bonds �Polar = 2 opposite electrical charges (like a battery) �Covalent = shares electrons �Unequal sharing between O and H’s (O is greedy!) �O is slightly – and H’s are slightly + �Between water molecules = hydrogen bonds
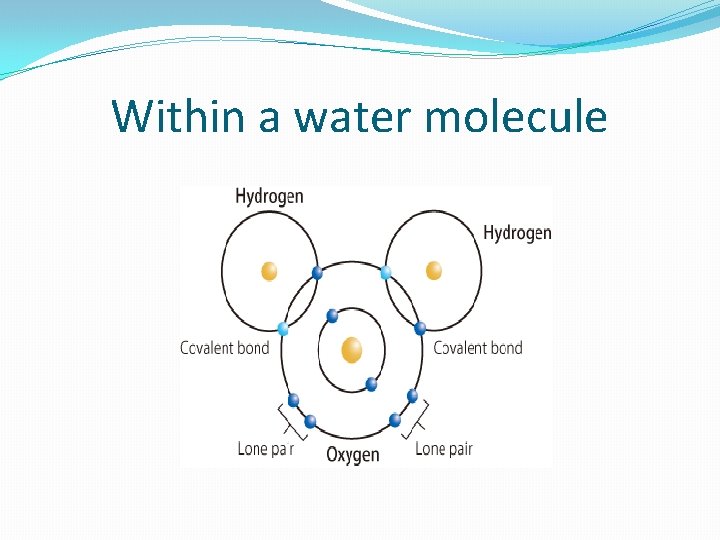
Within a water molecule
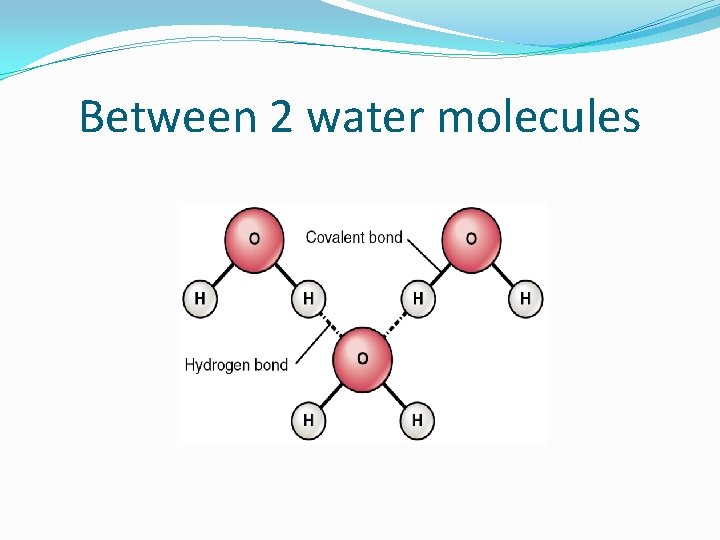
Between 2 water molecules

Nature of Science: Use theories to explain natural phenomena: theory that H bonds form b/n water molecules explain water’s properties. �H bonding b/n water molecules is a THEORY �Can’t prove b/c can’t see! �H bonds can explain properties of water �Theories assumed correct if: ~ evidence for it ~ can predict behavior ~ hasn’t been falsified ~ explains NATURAL phenomena
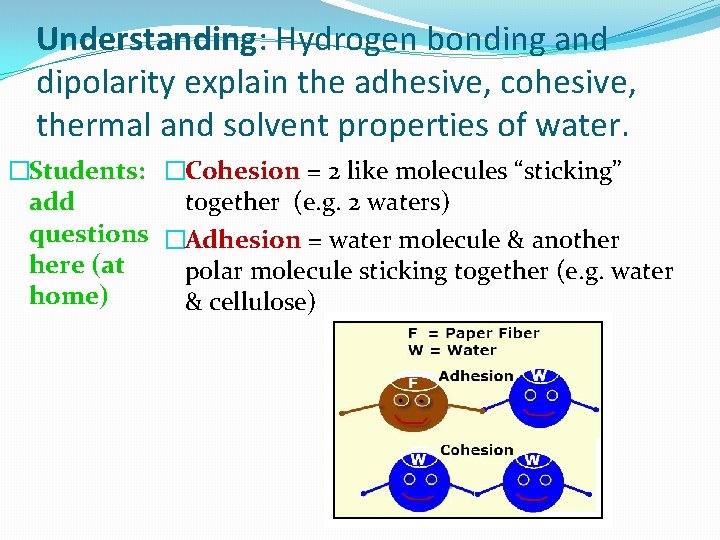
Understanding: Hydrogen bonding and dipolarity explain the adhesive, cohesive, thermal and solvent properties of water. �Students: �Cohesion = 2 like molecules “sticking” add together (e. g. 2 waters) questions �Adhesion = water molecule & another here (at polar molecule sticking together (e. g. water home) & cellulose)

… properties of water �Usefulness to organisms: Cohesion – Water can be pulled up to top of tall trees, against gravity Also, this guy! https: //www. youtube. com/watch? v=45 yabrnry. Xk

…properties of water �Thermal properties 1. High specific heat capacity = LARGE �Students amount of energy is needed to break H add bonds when raising temp of water (and to questions cool down, large amt of energy is lost) – so here! water temp remains stable while air and land temp rises and falls much quicker (Heat up pan of water – metal pan gets hotter quicker than the actual water does!) Usefulness to organisms: Water is a stable habitat for aquatic life!

… thermal properties of water: 2. High latent heat of vaporization = LARGE amount of heat needed to separate a water molecule from the others as it evaporates (goes from liquid to gas) Usefulness to organisms: Sweating cools us off because as it evaporates it has a cooling effect (heat taken away from our skin)!

…thermal properties of water 3. High boiling point = LARGE amounts of heat are needed to break H bonds and separate water molecules to become vapor Usefulness for organisms: Water is a great habitat for organisms because it stays liquid over a large range of temps (0° – 100°C)

… properties of water �Solvent properties = keeps other polar molecules and ions dissolved by forming shields around them �Usefulness to organisms: Many metabolic rxns can occur inside the cytoplasm of cells where molecules remain dissolved.

Understanding: Substances can be hydrophilic or hydrophobic Hydrophilic = water-loving = chemically attracted to water �Dissolves in water �Examples: 1. polar molecules (glucose) 2. ions (Na+ and Cl-) 3. molecules that water adheres to (cellulose)
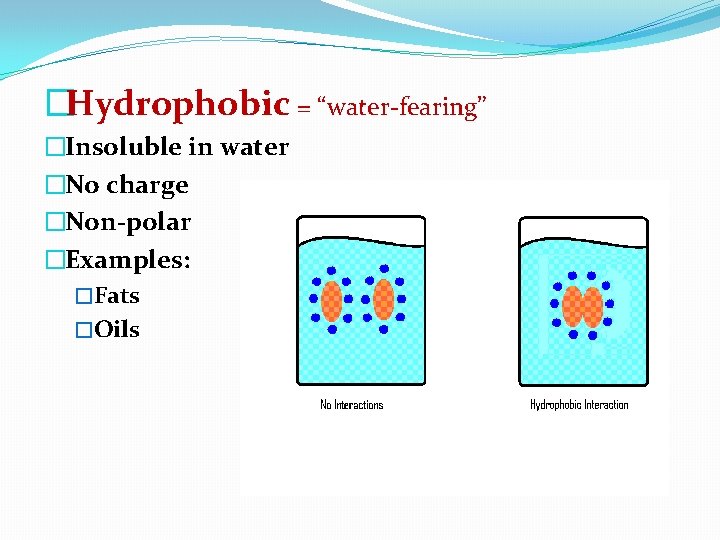
�Hydrophobic = “water-fearing” �Insoluble in water �No charge �Non-polar �Examples: �Fats �Oils

Applications: Comparison of thermal properties of water w/those of methane. �Methane (CH 4) = waste product of anaerobic respiration in some bacteria Property Formula Molecular mass Inner atomic bonding Polarity? can form H bond Density Specific heat capacity Latent heat of vaporization Melting point Boiling point temp range in which it is liquid Methane Water CH 4 H 2 O small covalent non-polar no yes less more lower higher 22°C 100°C This makes thermal properties different

Application: Use of water as a coolant in sweat �Hypothalamus = body’s thermostat – receives signal from skin & blood that body is overheating, & stimulates sweat glands �Sweat glands in skin secrete sweat, which travels through ducts to surface (~2 liters/hour) �Adrenalin cause body to overheat in intense or stressful activity �Evaporation of sweat has a cooling effect: heat needed for evaporation of water in sweat is taken from skin, reducing its temp �Ions dissolved in the water (Na+) are left behind on skin surface �Dogs and birds pant instead of sweat, which also involves evaporation of water as a cooling effect �Plants cool through evaporation of water from leaves (“transpiration”)

Application: Methods of transport of glucose, amino acids, cholesterol, fats, O 2 & Na. Cl in blood in relation to their solubility in H 2 O �Blood plasma (liquid part of blood) carries: �Na. Cl – soluble in water (dissolves to form Na+ and Clions) �A. A. - soluble in water; have + and – charges; solubility depends on R group, which can be either hydrophilic or hydrophobic �Glucose – soluble in water due to being polar �O 2 – non-polar; soluble due to small molecule, but solubility varies with water temp (higher temp hold less O 2); hemoglobin in RBC’s binds O to increase transport in blood �Fats – non-polar & large, therefore insoluble in water (hydrophobic); carried in blood inside lipoproteins �Cholesterol – mostly hydrophobic, therefore insoluble in water; carried in lipoproteins with fats
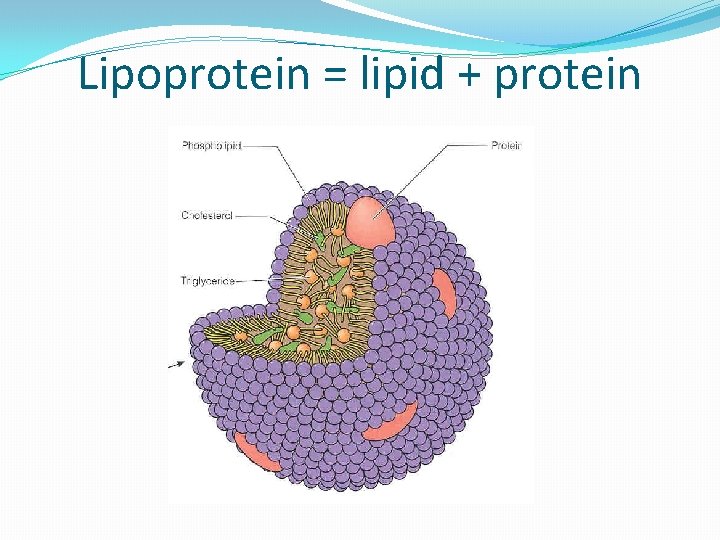
Lipoprotein = lipid + protein
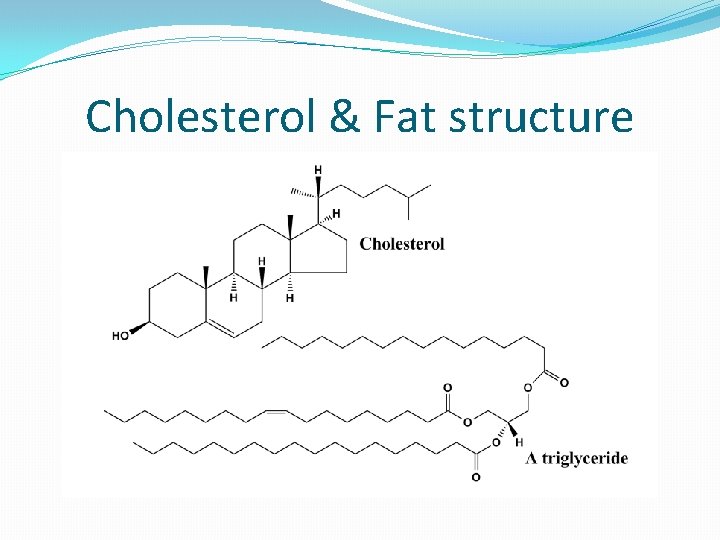
Cholesterol & Fat structure
- Slides: 18