TOPIC 1 BIOCHEMISTRY WHAT DOES IT MEAN FOR

TOPIC 1 BIOCHEMISTRY
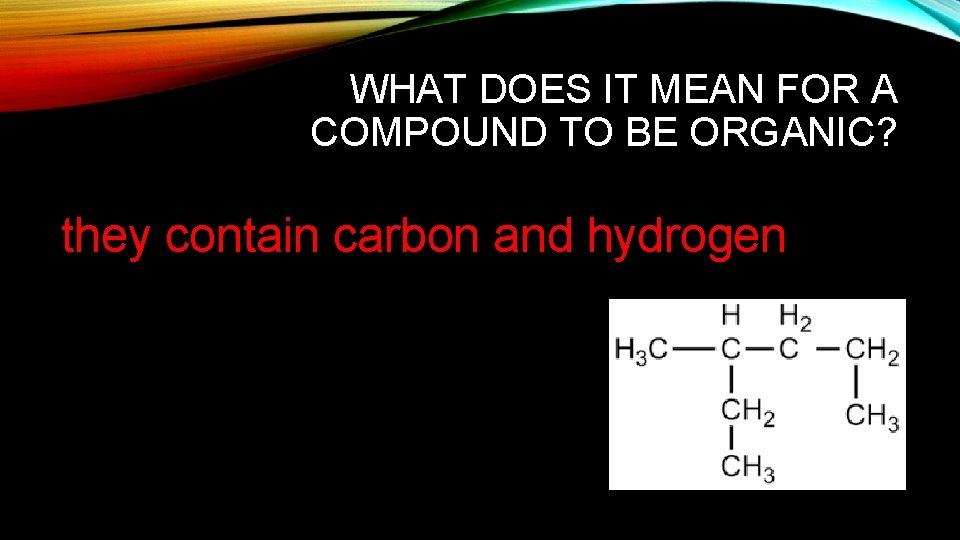
WHAT DOES IT MEAN FOR A COMPOUND TO BE ORGANIC? they contain carbon and hydrogen

Water is (circle one) polar / nonpolar because the electrons are not evenly shared.

WHAT ARE SOME PROPERTIES OF WATER THAT MAKE IT IMPORTANT TO LIFE ? polar molecule that forms hydrogen bonds; Cohesion, adhesion, high specific heat, density, universal solvent

WHAT IS THE DIFFERENCE BETWEEN A MONOMER AND POLYMER? monomers are building blocks of polymers
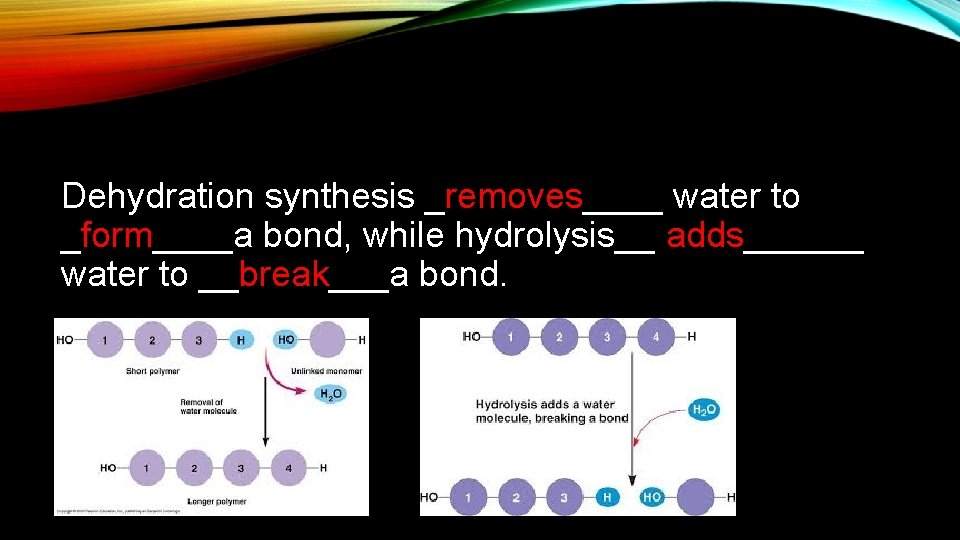
Dehydration synthesis _removes____ water to _form____a bond, while hydrolysis__ adds______ water to __break___a bond.
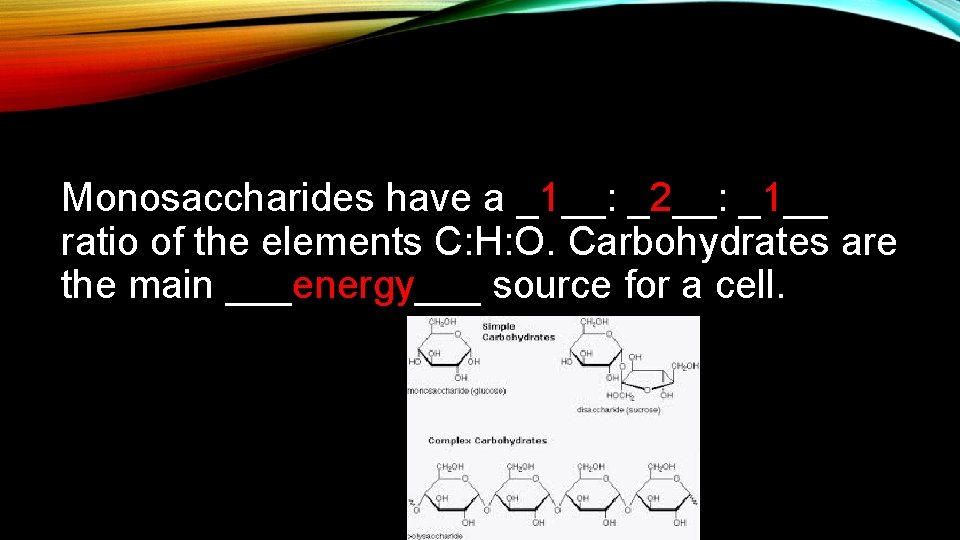
Monosaccharides have a _1__: _2__: _1__ ratio of the elements C: H: O. Carbohydrates are the main ___energy___ source for a cell.

WHAT ARE THE TWO PARTS OF A FAT? WHY ARE LIPIDS NOT TRUE POLYMERS? Composed of glycerol And 3 fatty acid chains …but lipids are not true polymers because they are not composed of repeating subunits
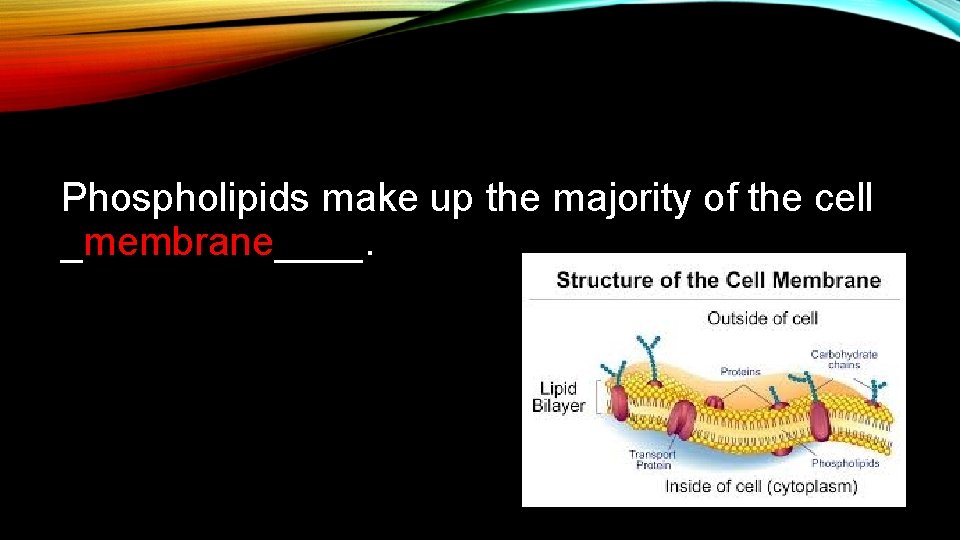
Phospholipids make up the majority of the cell _membrane____.
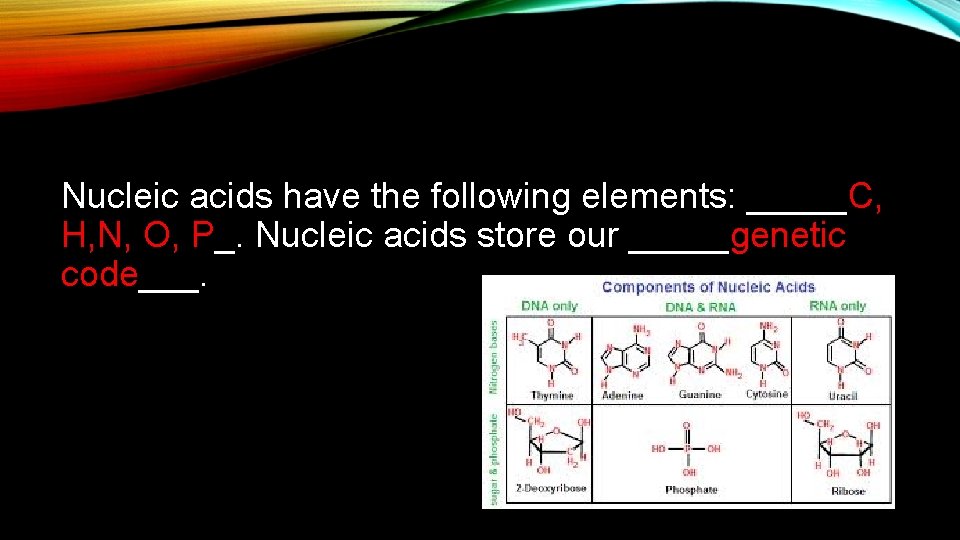
Nucleic acids have the following elements: _____C, H, N, O, P_. Nucleic acids store our _____genetic code___.
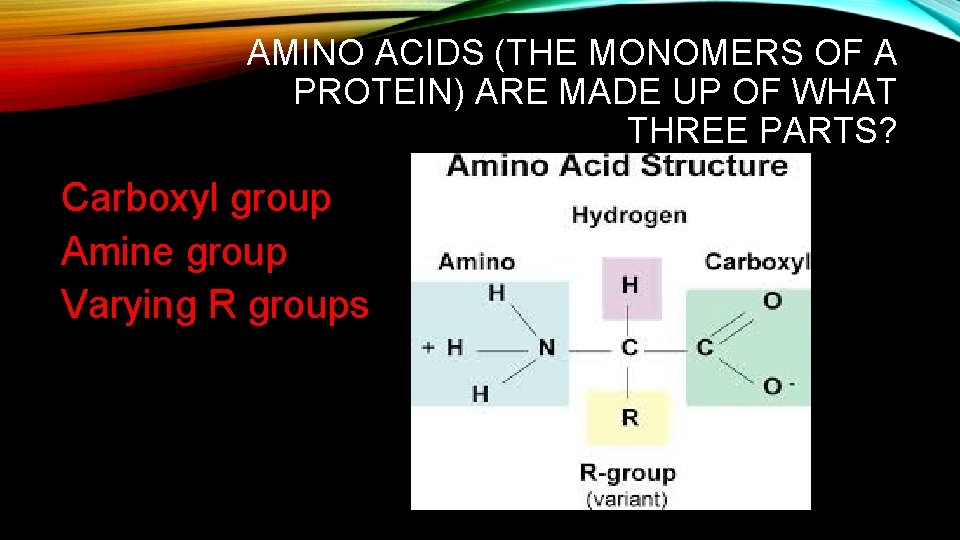
AMINO ACIDS (THE MONOMERS OF A PROTEIN) ARE MADE UP OF WHAT THREE PARTS? Carboxyl group Amine group Varying R groups
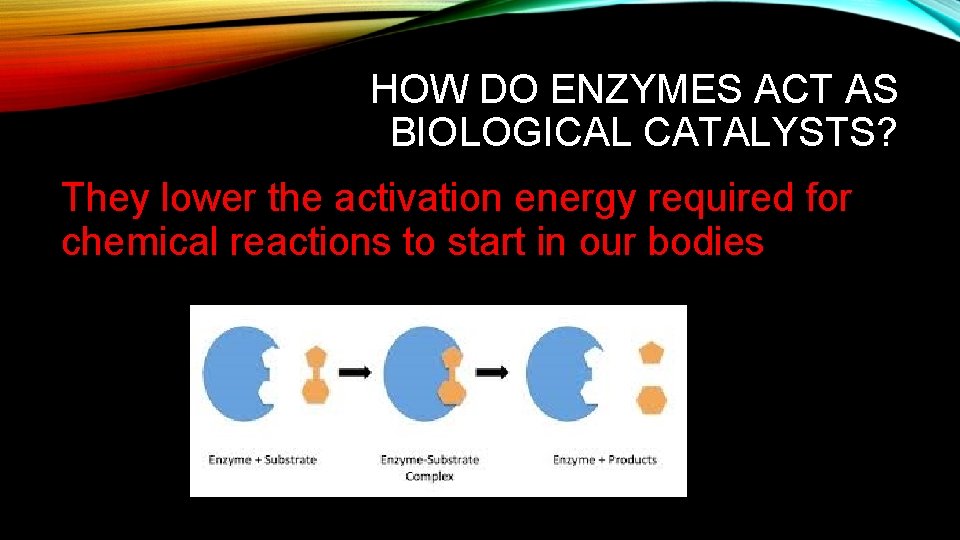
HOW DO ENZYMES ACT AS BIOLOGICAL CATALYSTS? They lower the activation energy required for chemical reactions to start in our bodies
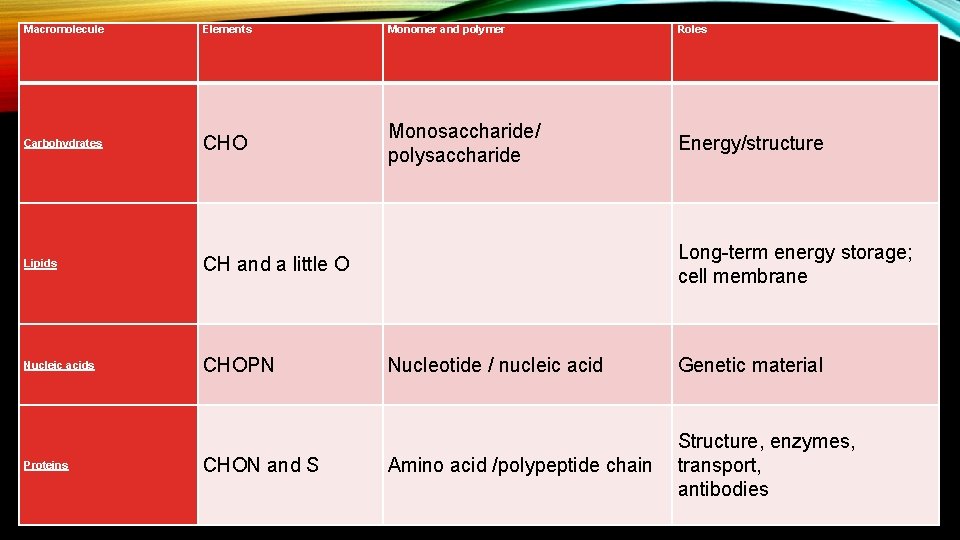
Macromolecule Elements Monomer and polymer Roles Carbohydrates CHO Monosaccharide/ polysaccharide Energy/structure Lipids CH and a little O Nucleic acids CHOPN Proteins CHON and S Long-term energy storage; cell membrane Nucleotide / nucleic acid Genetic material Amino acid /polypeptide chain Structure, enzymes, transport, antibodies
- Slides: 13