Toothbrush Sterilization Technique Analysis Lukas Belechak Grade 11

Toothbrush Sterilization Technique Analysis Lukas Belechak Grade 11 Pittsburgh Central Catholic High School

A Body’s Defense • The body is normally able to defend itself against infections. • Intact skin and mucous membranes function as passive barriers • When barriers are challenged or breached, active mechanisms protect the body from disease – Enzymes – Antibodies – Digestive acids – White blood cells

A Need to Brush • Toothbrushes may even • Personal oral hygiene have bacteria on them • Effective plaque removal right out of the box as they are not required to • Oral cavity is home to be sold in a sterile hundreds of different package types of microorganisms • Microorganisms may establish themselves on the brush

Dangers of Bacteria • Clinical studies have indicated that billions of harmful bacteria live on a toothbrush bristle • Oral infections can spread through toothbrush contact • Toothbrushes are vulnerable to contamination from airborne bacteria • Microbes from toilet may be atomized and sprayed up to 20 feet with single flush. • Scientists commonly find E. coli on toothbrushes

E. Coli Bacteria • Escherichia coli • Main species of bacteria • Often found in the lower intestines of mammals and in fecal matter • Causes sickness and disease such as urinary tract infections • Commonly studied because of its ubiquity and abundance

Purpose • The purpose of this experiment was to evaluate common toothbrush sterilization techniques.

Hypotheses • Null: There will be no variation between sterilization techniques outside of chance. • Alternative: All sterilization techniques will significantly reduce survivorship of the E. coli.

Materials • • • LB Media and LB Agar plates (1% Tryptone, 1% Na. Cl, 0. 5% Yeast Extract; for plates 1. 5% Agar) E. coli B Oral-B® Advantage® Plus Toothbrushes Sterile Water Listerine® Antiseptic Mouthwash Peroxide Cold Filtered Water (0°C) Hot Filtered Water (25°C) Isopropyl Alcohol Sterile Dilution Fluid (SDF) • • • Sterile 15 m. L Polystyrene Conical Tubes 50 -200 µl Pipettor and tips Bunsen Burner Spreader bar Matches Ethanol Turn Table Vortex Incubator set to 37°C

Procedure 1. E. coli was grown overnight in sterile LB material. 2. A sample of the overnight culture was added to fresh media in a sidearm flask. 3. The culture was placed in a shaking water bath (37°C) until a density of 50 Klett spectrophotometer units was reached. This represents a cell density of approximately 107. 4. The culture was diluted in sterile dilution fluid to a concentration of 10 6 cells/m. L. 5. 10 m. L of the culture was added to six sterile 15 m. L Polystyrene Conical Tubes. 6. Identical toothbrushes were sterilized and equally cut into smaller segments. 7. One toothbrush segment was added to each of the six conical tubes containing the culture. 8. The tubes were vortexed and allowed to sit for ten minutes. 9. Meanwhile, six sterile 15 m. L Polystyrene Conical Tubes were filled with 10 m. L of one of the following liquids: peroxide, isopropyl alcohol, filtered water at 0°C, filtered water at 25°C, Listerine® Antiseptic Mouthwash, and E. coli at a concentration of 106 cells/m. L (control).

Procedure – Method 1 10. After ten minutes, the toothbrushes were removed from the E. coli culture and one toothbrush was placed into one of the six different solutions. 11. Each tube was immediately vortexed and then allowed to sit for ten minutes. 12. After ten minutes, the solutions were once again vortexed. 13. 0. 1 m. L of each solution was taken from each tube and sterilely pipetted onto LB Agar plate (ten plates total for each solution). 14. The plates were incubated at 37°C for 24 hours. 15. After 24 hours, the resulting colonies were counted and recorded. All cells are assumed to have arisen from one cell.
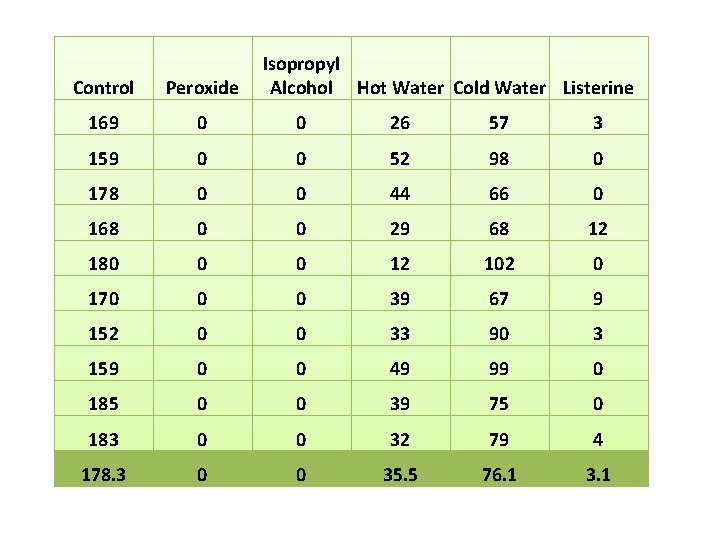
Isopropyl Alcohol Hot Water Cold Water Listerine Control Peroxide 169 0 0 26 57 3 159 0 0 52 98 0 178 0 0 44 66 0 168 0 0 29 68 12 180 0 0 12 102 0 170 0 0 39 67 9 152 0 0 33 90 3 159 0 0 49 99 0 185 0 0 39 75 0 183 0 0 32 79 4 178. 3 0 0 35. 5 76. 1 3. 1
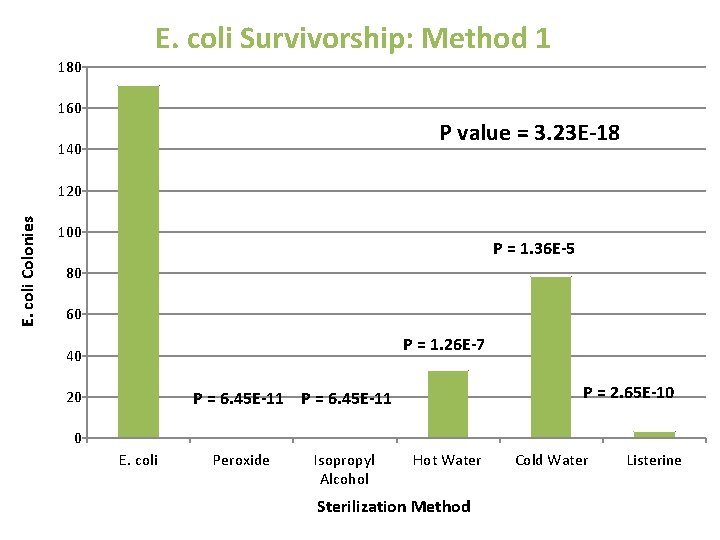
E. coli Survivorship: Method 1 180 160 P value = 3. 23 E-18 140 E. coli Colonies 120 100 P = 1. 36 E-5 80 60 P = 1. 26 E-7 40 20 P = 2. 65 E-10 P = 6. 45 E-11 0 E. coli Peroxide Isopropyl Alcohol Hot Water Sterilization Method Cold Water Listerine

Dunnett’s Test • Comparing Experimental Mean to Control Mean Solution T -Value T-Critical T value > T critical Peroxide 35. 408 3. 99 Significant Isopropyl Alcohol 35. 408 3. 99 Significant Hot Water 28. 358 3. 99 Significant Cold Water 20. 256 3. 99 Significant Listerine 34. 793 3. 99 Significant

Procedure – Method 2 16. After ten minutes, the toothbrushes were removed from the E. coli culture and one toothbrush was placed into one of the six different solutions. 17. Each tube was immediately vortexed and then allowed to sit for ten minutes. 18. After ten minutes, the solutions were once again vortexed. 19. 1 m. L of the each solution was pipetted into a sterile 15 m. L Polystyrene Conical Tube containing 9 m. L of SDF to create a 1/10 dilution of the Method 1 solutions. 20. The solution was immediately vortexed. 21. 0. 1 m. L of each solution was taken from each tube and sterilely pipetted onto LB Agar plate (ten plates total for each solution). 22. The plates were incubated at 37°C for 24 hours. 23. After 24 hours, the resulting colonies were counted and recorded. All cells were assumed to have arisen from one cell.
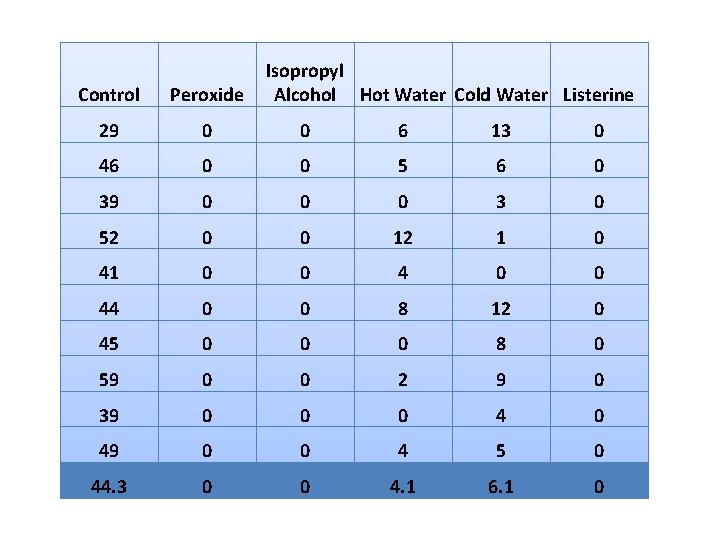
Isopropyl Alcohol Hot Water Cold Water Listerine Control Peroxide 29 0 0 6 13 0 46 0 0 5 6 0 39 0 0 0 3 0 52 0 0 12 1 0 41 0 0 44 0 0 8 12 0 45 0 0 0 8 0 59 0 0 2 9 0 39 0 0 0 49 0 0 4 5 0 44. 3 0 0 4. 1 6. 1 0
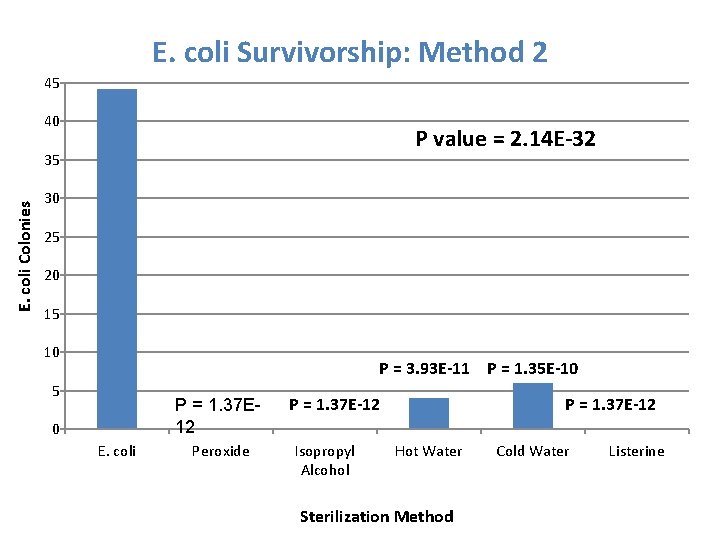
E. coli Survivorship: Method 2 45 40 P value = 2. 14 E-32 E. coli Colonies 35 30 25 20 15 10 P = 3. 93 E-11 P = 1. 35 E-10 5 P = 1. 37 E 12 0 E. coli Peroxide P = 1. 37 E-12 Isopropyl Alcohol P = 1. 37 E-12 Hot Water Sterilization Method Cold Water Listerine
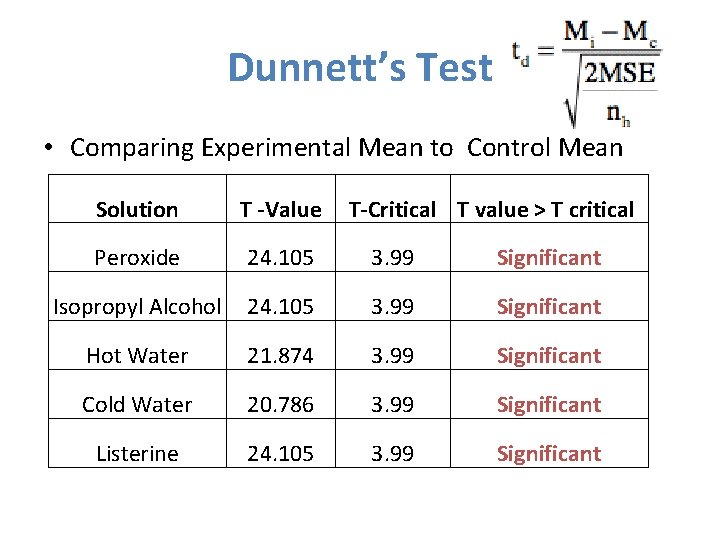
Dunnett’s Test • Comparing Experimental Mean to Control Mean Solution T -Value T-Critical T value > T critical Peroxide 24. 105 3. 99 Significant Isopropyl Alcohol 24. 105 3. 99 Significant Hot Water 21. 874 3. 99 Significant Cold Water 20. 786 3. 99 Significant Listerine 24. 105 3. 99 Significant

Conclusions • The Null Hypothesis can be rejected. • The statistical analysis strongly supports the hypothesis that all sterilization methods would reduce the survivorship of E. coli. • The Dunnett’s Test gives 99% confidence that the Null can be rejected. • The alternative hypothesis that all sterilization techniques will significantly reduce survivorship of the E. coli can be accepted.

Limitations and Extensions • Amount of liquid toothbrush bristles absorbed could not be standardized. • Work with a team of students in order assure all toothbrushes were immersed in the cell culture and variables for the same amount of time. • More trials could be performed. • More sanitization methods could be performed.

Sources Cited http: //en. wikipedia. org/wiki/E. _coli http: //davidmlane. com/hyperstat/B 112114. html http: //www. dentalgentlecare. com/fun_dental_facts. htm http: //www. ada. org/prof/resources/positions/statements/to othbrush. asp • http: //medind. nic. in/jao/t 02/i 3/jaot 02 i 3 p 81 g. pdf • http: //serendip. brynmawr. edu/exchange/node/1839 • http: //www. dentalarticles. com/mouth. html • •
- Slides: 20