TOIPC Periodic Table Organization Do Now From your

TOIPC: Periodic Table Organization Do Now: From your activity yesterday, and p. 5 answer page 6 in your packet

Periodic Law • There is a periodic repetition of chemical and physical properties of elements when arranged by increasing atomic number

What is the periodic table? • compact way of organizing elements • contains a lot of information • allows us to make predictions about behaviour and properties of elements • Elements
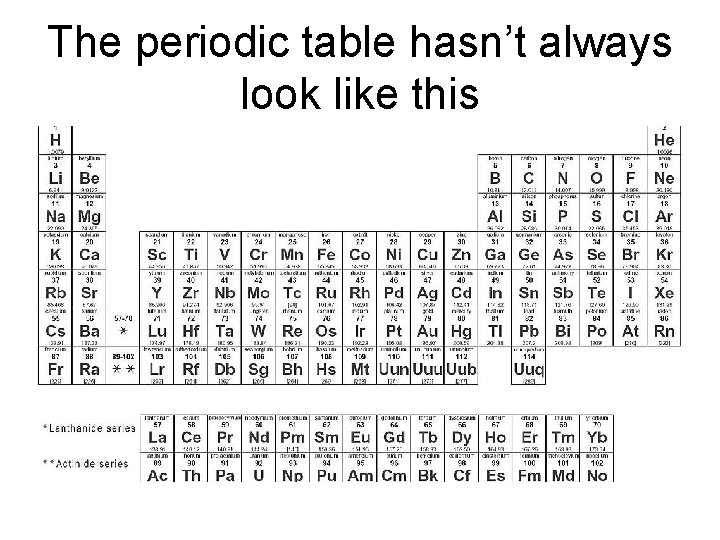
The periodic table hasn’t always look like this

History of the Periodic Table • End of the 1700’s – less than 30 elements known • Many elements discovered during 1800’s • Many experiments done to determine atomic masses
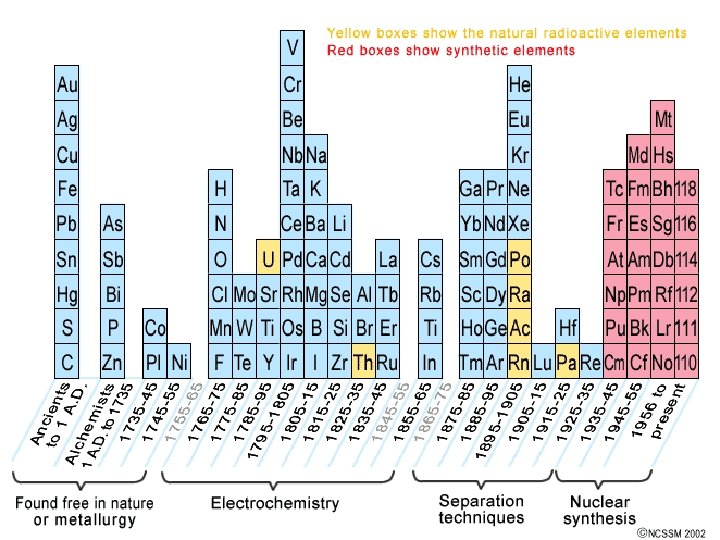

John Newlands • 1864: if elements arranged by atomic mass - properties repeat every 8 th element • Law of Octaves – did not work for all known elements • Key idea was correct: Properties of elements do repeat in periodic way

Mendeleev & Meyer Mendeleev produced 1 st accepted PT: 1869 • Elements ordered by ↑ atomic mass into columns with similar properties • Predicted existence & properties of undiscovered elements • Not totally correct – more accurate atomic mass calculations showed some elements weren’t in right place
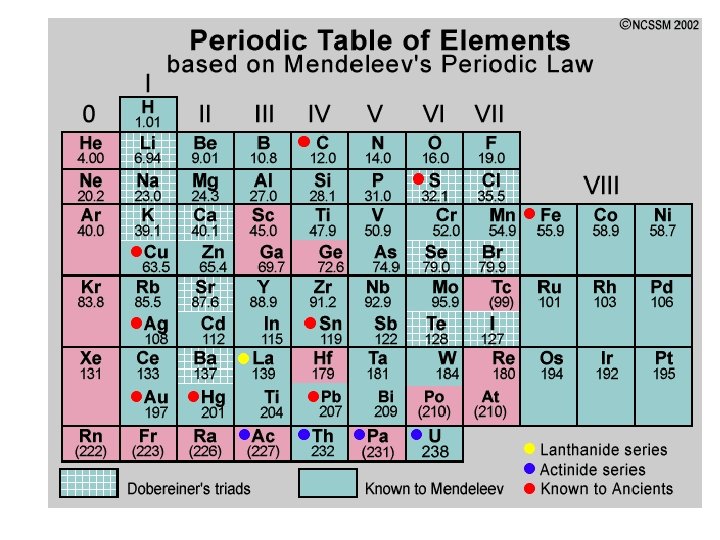

Remember 1860’s: • No subatomic particles yet discovered • Dalton’s billiard ball model of the atom

1913 – Henry Moseley • by 1913, protons & electrons discovered – Neutrons were predicted • Moseley determined atoms of each element contain unique # protons (= atomic number) • rearranged Mendeleev’s PT by atomic number instead of mass • problems with elements in wrong place disappeared
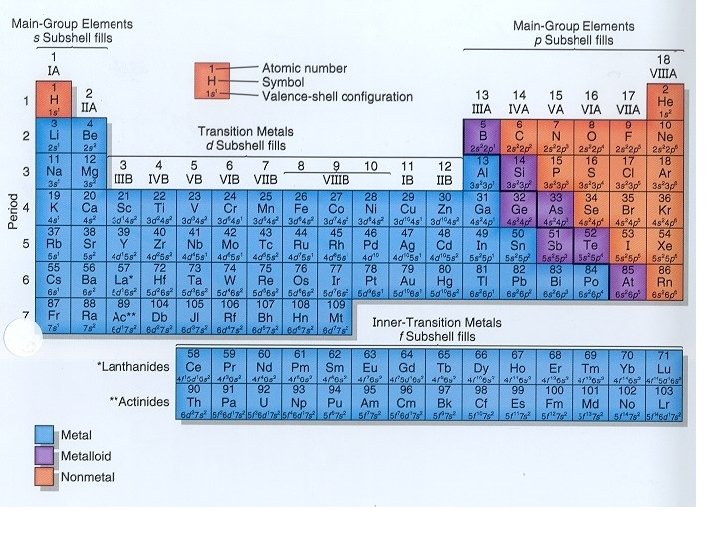
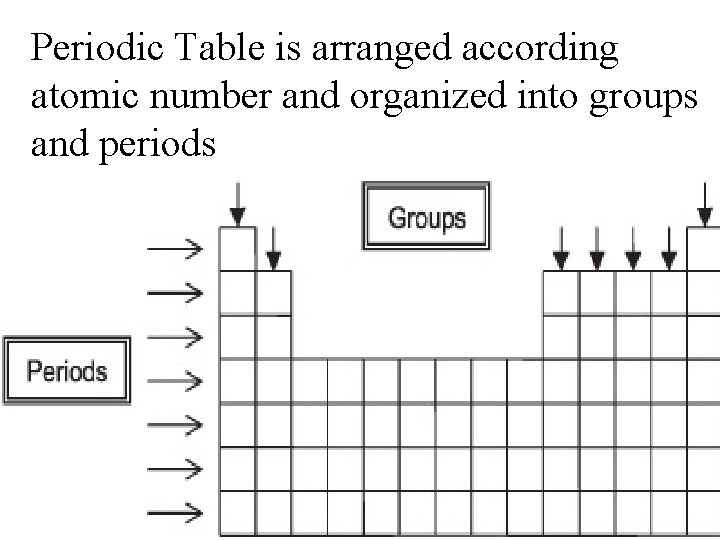
Periodic Table is arranged according atomic number and organized into groups and periods
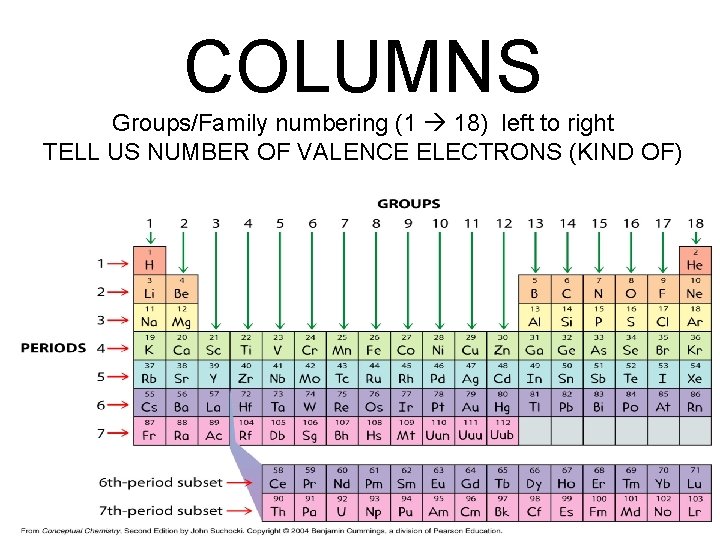
COLUMNS Groups/Family numbering (1 18) left to right TELL US NUMBER OF VALENCE ELECTRONS (KIND OF)

Valence Electrons – electrons in the outer shell • Chemical behaviour determined by # valence electrons • Elements with same # valence electrons will have similar chemical properties –Elements in same column have similar chemical properties
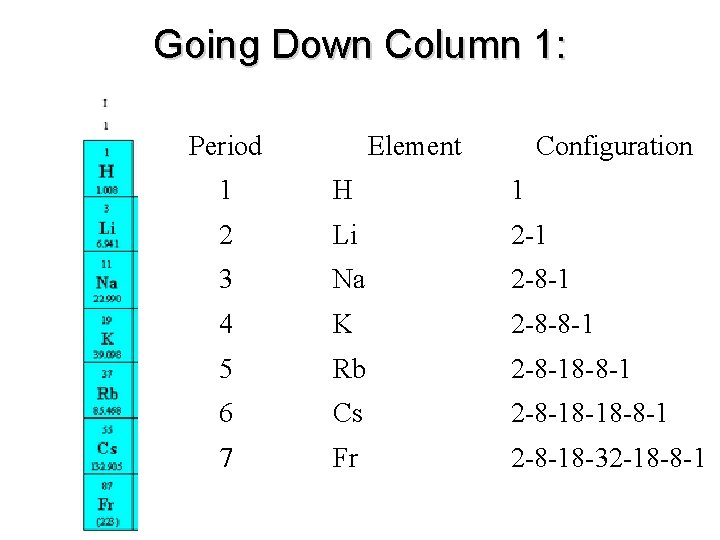
Going Down Column 1: Period Element Configuration 1 H 1 2 Li 2 -1 3 Na 2 -8 -1 4 K 2 -8 -8 -1 5 Rb 2 -8 -18 -8 -1 6 Cs 2 -8 -18 -18 -8 -1 7 Fr 2 -8 -18 -32 -18 -8 -1
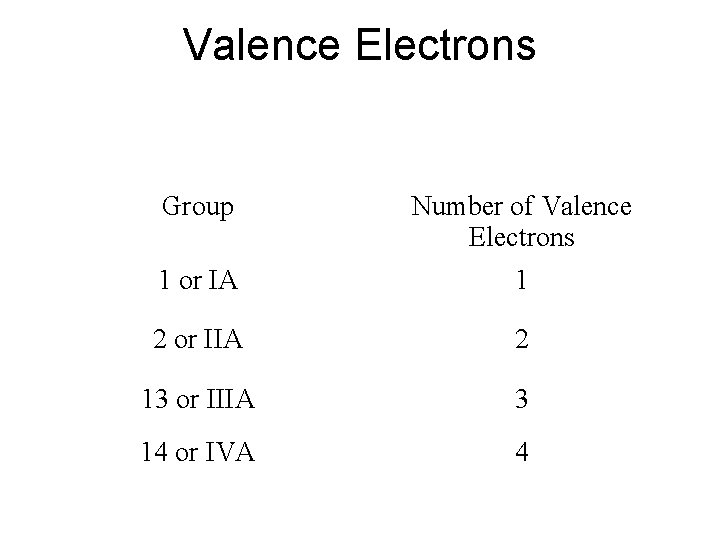
Valence Electrons Group Number of Valence Electrons 1 or IA 1 2 or IIA 2 13 or IIIA 3 14 or IVA 4

COLUMNS • Aka groups or families – #1 thru 18, Arabic numerals – (old way, Roman Numerals and letters) – - tell us number of valance electrons • (KIND OF) = works for 1, 2, 13, 14, 15, 16, 17, 18 Turn to packet page one, lets label the columns and valence electrons C O L U M N

Names of Families (AKA group A elements) • Group 1 = Alkali Metals • Group 2 = Alkaline Earth Metals • Groups 3 -12: Transition metals • Group 17 = Halogens • Group 18 = Noble Gases
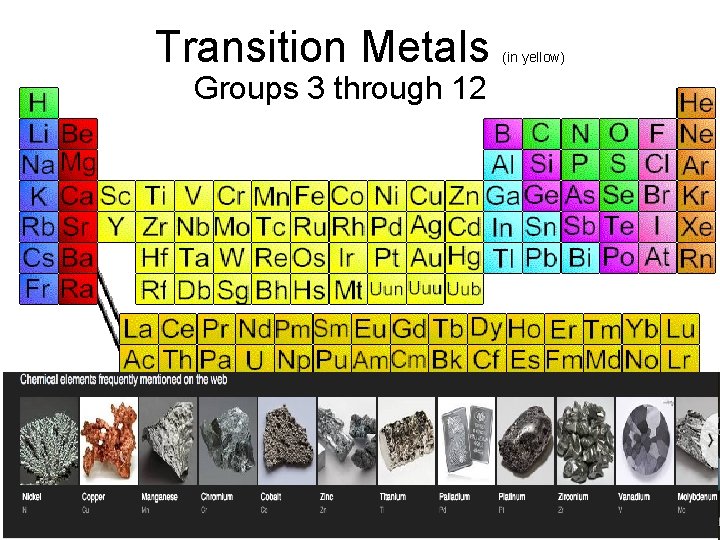
Transition Metals • Groups 3 through 12 (in yellow)

form brightly colored salts

Glenn Seaborg: 1950’s f-Block Metals • Lanthanide (rare Earth metals) – can be found naturally on Earth, only 1 is radioactive • Actinide– all are radioactive, some are made in the lab. INCLUDES Uranium Turn to packet page one, lets label the f-block metals
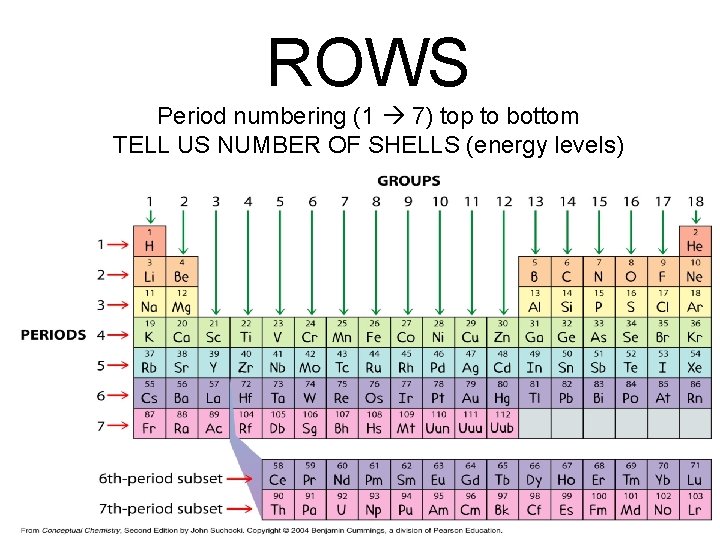
ROWS Period numbering (1 7) top to bottom TELL US NUMBER OF SHELLS (energy levels)
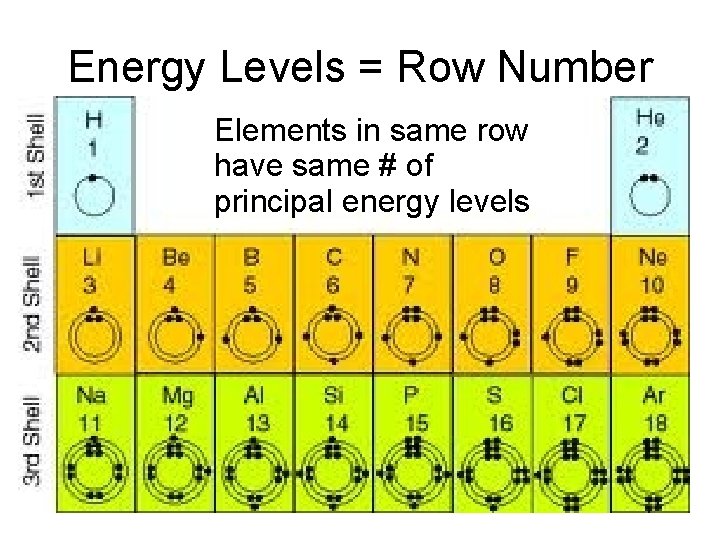
Energy Levels = Row Number • Elements in same row have same # of principal energy levels
ROWS • Rows are called series or periods – #1 thru 7 - TELL US HOW MANY ENERGY LEVELS ROW Turn to packet page one, lets label the periods
- Slides: 27