Todays Agenda Bellwork Introduction to Stoichiometry Mole Ratio

Today’s Agenda • Bellwork • Introduction to Stoichiometry • Mole Ratio Notes • Mole Ratio Practice • Exit Ticket

Introduction to Stoichiometry and Mole Ratios

Learning Target • I can determine the mole ratio in a balanced equation. • I can use the mole ratio to convert between substances.

Stoichiometry • What is it? • Stoichiometry – Study of quantitative relationships between the amounts of reactants used and amounts of products formed by a chemical reaction. • Why is it useful? • Allows scientists to determine how much product can be made. • Allows scientists to know how much of the reactant they can use.

Mole Ratio • Mole Ratio is the KEY to stoichiometry! • What is it? • Mole Ratio – Ratio between the numbers of moles of any two of the substances in a balanced equation. • How do I find it? • The coefficients in the balanced equation will give you the mole ratio between substances.

Finding the Mole Ratio Ex 1 • Use the following equation to answer the questions. 2 K + Br 2 2 KBr 1. What is the mole ratio between potassium, K, and bromine, Br 2, in the reaction. 2. What is the mole ratio between bromine, Br 2, and potassium bromide, KBr.

Finding the Mole Ratio Ex 2 • Use the following equation to answer the questions. 2 KCl. O 3 + 2 KCl 3 O 2 1. What is the mole ratio between KCl and O 2? 2. What is the mole ratio between oxygen, O 2, and potassium chlorate, KCl. O 3? 3. What is the mole ratio between potassium chlorate, KCl. O 3, and potassium chloride, KCl?

Today’s Agenda • Bellwork • Review Homework • Steps to solving mole – mole stoichiometry problems • Practice Problems • Exit Ticket

Mole to Mole Stoichiometry

Today’s Objective • Learning Target • I can solve mole – mole stoichiometry problems

Steps to Solving Stoichiometry Problems • Step 1 • Write down what is given and what you want to find.

Steps to Solving Stoichiometry Problems • Step 1 • Write down what is given and what you want to find. • Step 2 • Determine the mole ratio for the substances in the problem

Steps to Solving Stoichiometry Problems • Step 1 • Write down what is given and what you want to find. • Step 2 • Determine the mole ratio for the substances in the problem • Step 3 • Determine where to start on the road map

Steps to Solving Stoichiometry Problems • Step 1 • Write down what is given and what you want to find. • Step 2 • Determine the mole ratio for the substances in the problem • Step 3 • Determine where to start on the road map • Step 4 • Follow the road map to stoichiometry success!!!

Using the Mole Ratio • Use the following equation for the Mole Ratio Moles of Unknown Moles of Given

Mole – Mole Stoich Ex 1 • Use the following equation to answer the questions: 2 HCl + Br 2 2 HBr + Cl 2 1. What is the mole ratio between Cl 2 and HCl?

Mole – Mole Stoich Ex 1 • Use the following equation to answer the questions: 2 HCl + Br 2 2 HBr + Cl 2 1. How many moles of chlorine will be produced if you start with 5. 0 moles of HCl?

Mole – Mole Stoich Ex 1 • Use the following equation to answer the questions: 2 HCl + Br 2 2 HBr + Cl 2 1. If you need 6. 0 moles of HBr, how many moles of Br 2 need to react?

Mole – Mole Stoich Ex 2 • Use the following equation to answer the questions: 4 NH 3 + 5 O 2 6 H 2 O + 4 NO 1. How many moles of H 2 O are produced if 2. 00 moles of O 2 are used? 2. How many moles of NO are produced in the reaction if 15 moles of H 2 O are also produced? 3. If you start with 3. 25 moles of NH 3, how many moles of H 2 O will be used?

Exit Ticket • Use the following equation to answer the questions 2 Ag 2 O 4 Ag + O 2 1. What is the mole ratio between silver oxide (Ag 2) and oxygen (O 2)? 2. How many moles of oxygen will be produced if you start with 4. 50 moles of Ag 2 O?

Today’s Agenda • Bellwork • Review Homework • Mass – Mole and Mole – Mass Stoichiometry • Practice Problems • Exit Ticket

Mole – Mass Stoich Ex 1 2 HCl + Br 2 2 HBr + Cl 2 1. How many grams of chlorine will be produced if you start with 5. 0 moles of HCl?

Mass– Mole Stoich Ex 1 2 HCl + Br 2 2 HBr + Cl 2 1. If you need 15. 0 grams of HBr, how many moles of Br 2 will react?
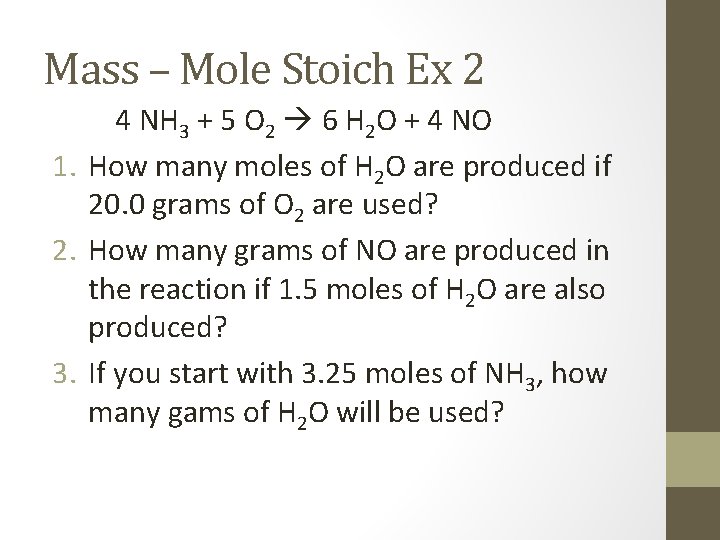
Mass – Mole Stoich Ex 2 4 NH 3 + 5 O 2 6 H 2 O + 4 NO 1. How many moles of H 2 O are produced if 20. 0 grams of O 2 are used? 2. How many grams of NO are produced in the reaction if 1. 5 moles of H 2 O are also produced? 3. If you start with 3. 25 moles of NH 3, how many gams of H 2 O will be used?

Exit Ticket 2 Ag 2 O 4 Ag + O 2 1. How many moles of Ag are made if you start with 10. 0 grams of Ag 2 O? 2. A chemist starts with 1. 25 moles of Ag 2 O, how many grams of O 2 will be made?
- Slides: 25