Today We are here Cell mediated immunity BY

Today We are here Cell mediated immunity BY Dr. Dhafer Abdullah Farhan Ph. D Cancer research

List of abbreviations FAS fibroblast- associated cell surface TNF Tumor necrosis factor CD Cluster of differentiation CMI Cell Mediated Immunity Tc T cytotoxic cell ADCC AB-dependent cell-mediated cytotoxicity

Cell Mediated Immunity Historically, immunologists have divided adaptive immunity into namely: n CMI, which can be adoptively transferred only by viable T lymphocytes. n Humoral immunity, which can be adoptively transferred with serum containing antibodies. n

Cell Mediated Immunity Many microbes have developed mechanisms that enable them to survive and even replicate within phagocytes, so the innate immunity is unable to eradicate infections by such microbes. n In CMI against phagocytosed microbes, the specificity of the response is due to T cells – but the actual effector function is mediated by the phagocytes. n

Cell Mediated Immunity * Host defenses against extracellular infections are mediated by: - Antibody - Complement - Macrophages * Intercellular infections are mediates by CMI * CMI are responsible for: - Resistance to intracellular pathogens - Resistance to fungal and protozoal

Cell Mediated Immunity * CMI may play a role in some harmful conditions: - Hypersensitivity reactions type IV (contact dermatitis) - Graft rejection - Autoimmune diseases * Cell mediated cytotoxicity mediated by: - T-cytotoxic cells - Natural killer cells - Activated macrophages

Characters Of CMI Cellular immune response is mediated by: - Subpopulation of T-lymphocytes - Macrophages and their products
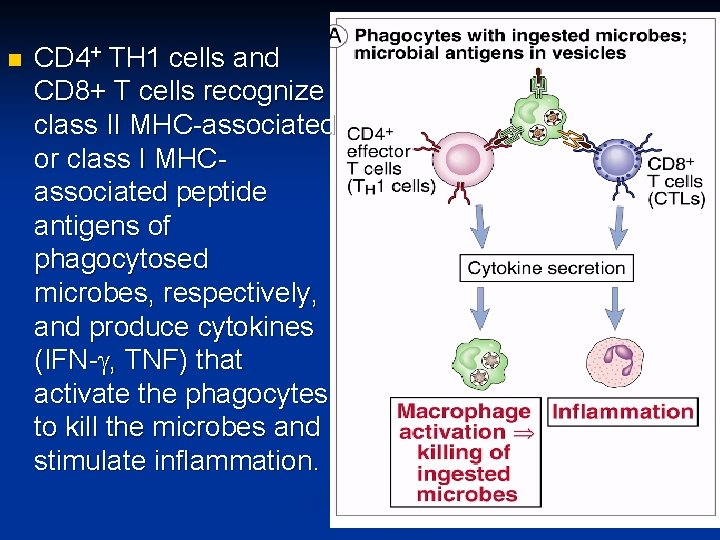
n CD 4+ TH 1 cells and CD 8+ T cells recognize class II MHC-associated or class I MHCassociated peptide antigens of phagocytosed microbes, respectively, and produce cytokines (IFN-g, TNF) that activate the phagocytes to kill the microbes and stimulate inflammation.
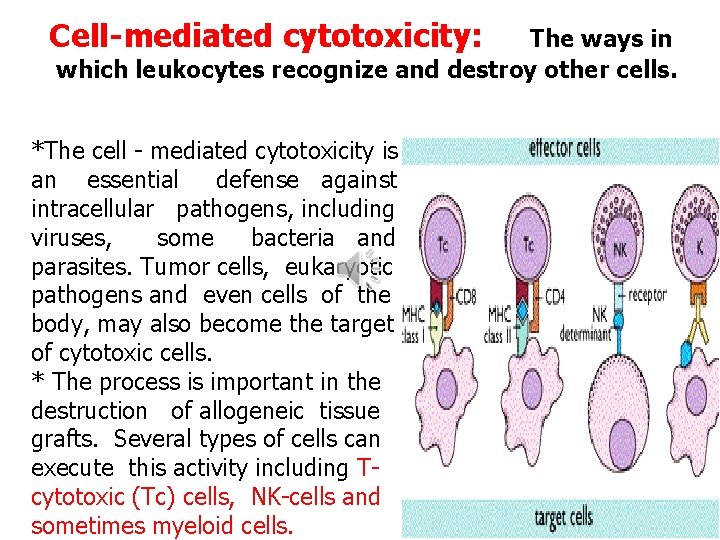
Cell-mediated cytotoxicity: The ways in which leukocytes recognize and destroy other cells. *The cell - mediated cytotoxicity is an essential defense against intracellular pathogens, including viruses, some bacteria and parasites. Tumor cells, eukaryotic pathogens and even cells of the body, may also become the target of cytotoxic cells. * The process is important in the destruction of allogeneic tissue grafts. Several types of cells can execute this activity including Tcytotoxic (Tc) cells, NK-cells and sometimes myeloid cells.

Tc-cells and NK-cells act in a complementary way to protect the body. Tc-cells recognize specific Ag (e. g. viral peptides on infected cells) presented by MHC-I most Tc are CD 8+ve, but about 10% of MHC-restricted cytotoxic T-cells are CD 4+ve & recognize Ag presented on MHC-II. NK-cells recognize cells which fail to express MHCI molecules, they can also bind to Ab already attached to Ag an a target cell, using their Fc-R (CD 16), in a what is known as AB-dependent cell -mediated cytotoxicity (ADCC). Several viruses have evolved mechanisms to avoid recognition by Tc cell. They reduce the expression of MHC molecules or even produce proteins which divert MHC molecules out of the endoplasmic reticulum. So NKcells specifically recognize cells which have lost their MHC - I molecules.
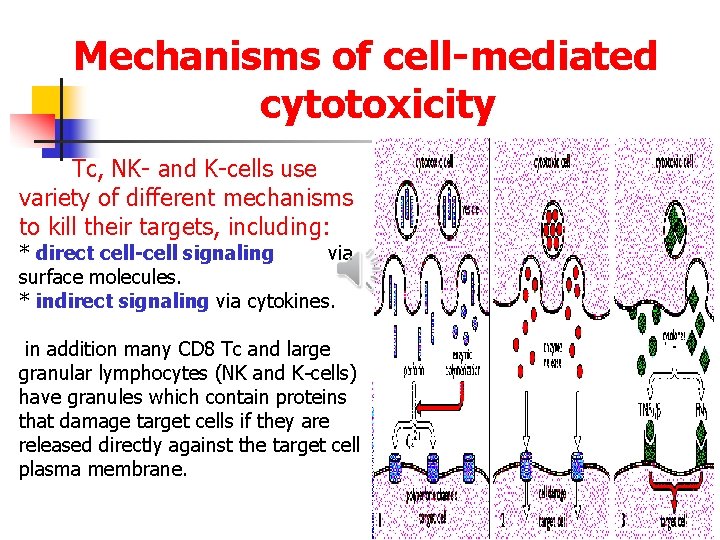
Mechanisms of cell-mediated cytotoxicity Tc, NK- and K-cells use variety of different mechanisms to kill their targets, including: * direct cell-cell signaling via surface molecules. * indirect signaling via cytokines. in addition many CD 8 Tc and large granular lymphocytes (NK and K-cells) have granules which contain proteins that damage target cells if they are released directly against the target cell plasma membrane.

• a cytotoxicity which may be signaled via Fas or a TNF-R on a target cell. • Tc cells signal to their targets using members of TNF-R group of molecules. These include Fas (CD 95) and the type-1 TNF-R (TNFR-1) which are widely distributed in the body. Ligation of Fas-induces trimerization of the Fas-molecule on the cell surface, which causes them to associate with a transducing molecule which recrutes and activates caspases 8 and 10. • For the cytotoxic lymphocytes which lack granules the Faspathway is thought to be the principle means of signaling to the target.

** Most CD 8 TC and NK-cells (and macrophages) have vesicles containing TNF and lymphotoxin which can be released onto a target cells. TNF acts in a very similar way to the Fas-ligand. It causes trimerization of the TNFR-1 so that the receptor associates with adaptor proteins which recruite caspases. Both TNFR-1 and Fas containing intracytoplasmic domains (deathdomains) which are found on a number of proteins involved in cell survival.

*** The specific granules of NK-cells and Tc contain several proteins, including perforin and granzymes (granules associated enzymes). After binding to its target, the Tc cells directs its granules towards the membrane a joining the target. Then, in a Ca-dependent phase, the granule contents are discharge into the cleft between the two cells. Perforin is a pore-forming proteins and in the presence of Ca , the perforin bind to the target cell membrane and polymerize to form transmembrane channels. Tc-protected from autodestruction with this protein by a proteoglycan (chondroitin-sulphate A) which may bind to and inactivate the perforin, thus Tc-survives and can continue kill further targets. Granzymes are collection of serine estrases (enzymes) which are also released upon granule exocytosis and become active after release. Some of these enzyme trigger apoptosis and DNA-degradation. Both perforin and granzymes act synergistically; the granzymes enter the target cells via pore created by perforin.

To summarize the lymphoid cytotoxic effectors; CD 8 Tc use both Fas- L and granule release to kill their targets, CD 4 Tc-cells use principally Fas. L, and NK-cells use primarily their granules. TNF may contribute to the cytotoxic damage produced by any of these cells.

A number of non-lymphoid cells may be cytotoxic to other cells, *cytotoxicity may be triggered specifically to a target by ADCC or may involve a range of non-specific toxic mediators. Macrophage and neutrophile express Fc-g. R 1&2 which allow them to engage their targets by ADCC. ` ** Macrophages and neutrophiles granules which contain neutrophil defensins, lysosomal enzymes and cytostatic proteins. If the phagocyte fails to internalize its target, then these mediators may be released into the extracellular environment and contribute to localized cell damage ((frustrated phagocytosis)), the effect of these mediators tends to produce necrosis and inflammation. *** Activated macrophage secrete TNF which can induce apoptosis in a similar way to lymphoid cyotoxic effectors.

Eosinophils are characterized by their granules. Eosinophils are only weakly phagocytic, they ingest some bacteria following activation but are less efficient than neutrophils at intracellular killing. Their major function appears to be the secretion of various toxic granules. Constituents. They are therefore effective for the extracellular killing of microorganisms, particularly large parasites such as schistosoma. The components of the eosinophil granule include major basic protein (MBP), eosinophil peroxidase (EPO) and eosinophil cationic protein (ECP). Other molecules produced by eosinophils are lysophospholipase and eosinophil -derived neurotoxin (EDN), which is also a ribonuclease but with strong neurotoxic activity. Thus, killing may involve contact-dependent degranulation or may simply require deposition of toxins within the local tissue.

THANK YOU
- Slides: 18