TOBACCO CESSATION Smoking Tobacco Cessation Many adult cigarette

TOBACCO CESSATION
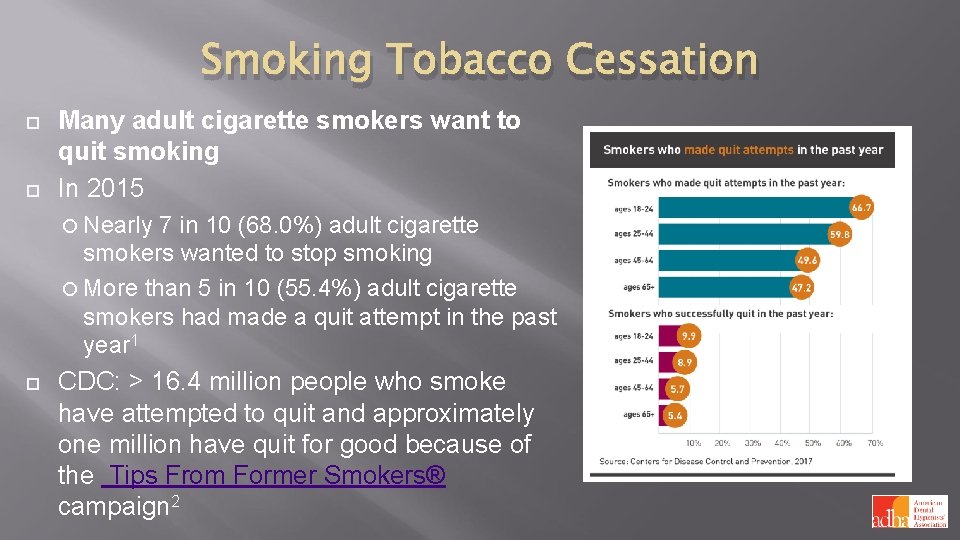
Smoking Tobacco Cessation Many adult cigarette smokers want to quit smoking In 2015 Nearly 7 in 10 (68. 0%) adult cigarette smokers wanted to stop smoking More than 5 in 10 (55. 4%) adult cigarette smokers had made a quit attempt in the past year 1 CDC: > 16. 4 million people who smoke have attempted to quit and approximately one million have quit for good because of the Tips From Former Smokers® campaign 2
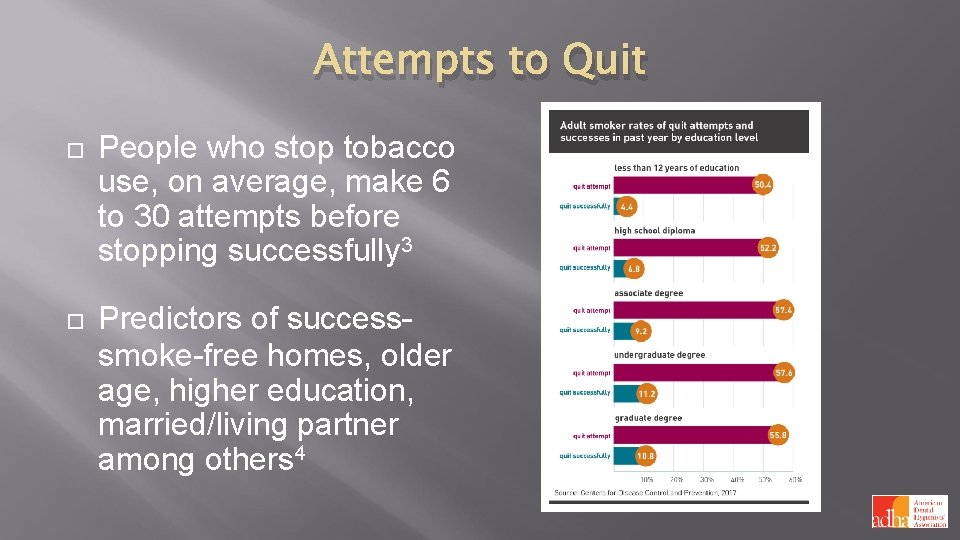
Attempts to Quit People who stop tobacco use, on average, make 6 to 30 attempts before stopping successfully 3 Predictors of success– smoke-free homes, older age, higher education, married/living partner among others 4

Two Categories for Treatment Behavioral Pharmacotherapy

Behavioral Interventions In-person behavioral support/counseling Telephone counseling (e. g. 1 -800 -QUIT-NOW) Self-help materials Behavioral interventions may increase rates of abstinence from 5 -11% to 7 -13%5 Recommended for all adults >/= 18 years of age 6 Non-pregnant adults: alone or in combination with pharmacotherapy improve likelihood of tobacco cessation Pregnant adults: improve achievement of tobacco smoking abstinence, increase infant birthweight, and reduce risk for preterm birth

Pharmacotherapy Interventions Multiple medications FDA approved Nicotine-replacement therapy (NRT) Bupropion HCl (Wellbutrin SR, Zyban) Varenicline (Chantix) Recommended for all non-pregnant adults >/= 18 years 6 Alone or in combination with behavioral interventions improve likelihood of tobacco cessation

Nicotine Replacement Therapy (NRT) Nicotine patch (OTC) Nicotine gum (OTC) Nicotine inhaler (Rx) Nicotine lozenge (OTC) Nicotine nasal spray (Rx) Using two types of NRT improves likelihood of tobacco cessation over just one 6 Addition of NRT to bupropion superior to bupropion alone 6
Nicotine Patch or Transdermal OTC Slow release over an extended period of time Dosage 5 -21 mg/day

Nicotine Gum � � � OTC Transmucosal delivery released when chewed Content and dosage � 2 mg or 4 mg per piece � Recommended 9 -12 pieces per day � 2 mg strength no more than 30 pieces � 4 mg no more than 20 pieces � One per hour for 12 weeks � � � Can not eat or drink 15 minutes before using Used for quick relief from uncontrollable urges Instead of chewing gum entire hour, should chew until soft and then place gum between cheek and gingiva for a few minutes then reposition in different areas

Nicotine Inhaler � � � Prescription only Maximum limit of 16 cartridges a day, each cartridge delivers 4 mg or nicotine, which is around 80 inhalations Use for six months with gradual reduction in frequency over last 6 -12 weeks Do not eat or drink 15 minutes prior to usage Can not use with severe asthma

Nicotine Lozenge OTC Nicotine replacement therapy that comes in the form of a small, candy-like, sugar-free tablet in flavors like cinnamon, fruit, and mint. Nicotine lozenges should be used in the following dosages: Weeks 1– 6: One lozenge every 1 to 2 hours Weeks 7– 9: One lozenge every 2 to 4 hours Weeks 10– 12: One lozenge every 4 to 8 hours

Nicotine Nasal Spray � � � Prescription only Content: range from 2 -4 mg per dose 1 -2 sprays per hour 90% of all users report moderate to severe nasal irritation within first 2 -3 days May take 3+ months Can not use with severe asthma

Bupropion HCl (Wellbutrin SR, Zyban) Indications: smoking cessation in addition to major depressive disorder (MDD), attention deficit hyperactivity disorder Dosing: 150 mg once or twice a day for 7 -12 weeks Start 1 -2 weeks before quit date and use 2 -3 months after quit date Side effects: dry mouth and insomnia, worsening depression, mania Contraindication: seizure disorder Black box warning: increased risk for suicidality in children, adolescents, and young adults with MDD or other psychiatric disorders

Varenicline (Chantix) Indication: smoking cessation Dosing Starting: 0. 5 mg once a day for 3 days, then 0. 5 mg twice a day for 4 days, then 1 mg twice a day for 11 weeks (12 week total) Continuing: after initial 12 weeks, can extend to additional 12 weeks 1 mg twice a day Side effects: nausea, vomiting, abnormal dreams, sleep walking, suicidality Not to be combined with nicotine patches, gums, sprays or lozenges (increased risk nausea, vomiting and other side effects) Contraindication: age < 18 years

Considerations When Recommending Pharmacotherapies Health precautions Cost of medication Patient preference Previous patient experience when trying to quit History of depression or anxiety Concern with side effects

Engaging Patients in Cessation Discussion 5 A’s Intervention has been used as a tool to engage patients on smoking tobacco cessation in a clinical setting 7 1. Ask A 2. Advise A 3. Assess A 4. Assist A 5. Arrange A Patients who receive 5 A’s intervention have higher utilization of recommended counseling and medication therapies 8

Ask Do you use tobacco? How do you feel about it? Have you ever thought about quitting? Obtain pt confidence Present questions carefully

Advise To stop Motivate patient Personalize the message to patient’s lifestyle, needs, etc. Show willingness and concern Related oral findings Patient resistance: you have done your job and do not antagonize patient, and document all information given Be honest

Assess Determine willingness to make a quit attempt and use intervention appropriate their stage

Assist Set a quit date Alert family and friends and seek social support Remove tobacco products Learn from past experience Provide helpful hints for successful quitting Present education and motivational materials Anticipate problems Encourage use of pharmaceutical agents

Arrange Contact the patient on the quit date Praise the patient Follow up at recall In case of a relapse have plan or more encouragement What conditions lead to relapse? Encourage patient to try again Review type of pharmaceutical agents used

Content Sources 1. Wang TW, Asman K, Gentzke AS, Cullen KA, Holder-Hayes E, Reyes-Guzman C, Jamal A, Neff L, King BA. Tobacco product use among adults—United States, 2017. Morb Mortal Wkly Rep 2018; 67(44): 1225 -32. 2. Fast Facts: Centers for Disease Control and Prevention. https: //www. cdc. gov/tobacco/data_statistics/fact_sheets/fast_facts/index. htm. Date accessed: 16 Oct 2019. 3. Chaiton M, Diemert L, Cohen JE, Bondy SJ, Selby P, Philipneri A, Schwartz R. Estimating the number of quit attempts it takes to quit smoking successfully in a longitudinal cohort of smokers. BMJ Open 2016; 6: e 011045. doi: 10. 1136/bmjopen-2016 -011045 4. Chung-won L, Kahende J. Factors associated with successful smoking cessation in the United States, 2000. Am J Public Health 2007; 97: 1503 -1509. 5. Patnode CP, Henderson JT, Thompson JH, Senger CA, Fortmann SP, Whitlock EP. Behavioral counseling and pharmacotherapy interventions for tobacco cessation in adults, including pregnant women: a review of reviews for the U. S. Preventive Services Task Force. Ann Intern Med 2015; 163(8): 608 -621. 6. Siu AL. Behavioral and pharmacotherapy interventions for tobacco smoking cessation in adults, including pregnant women: U. S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2015; 163(8): 622 -634. 7. Five Major Steps to Intervention (The “ 5 A’s”): AHRQ. https: //www. ahrq. gov/sites/default/files/wysiwyg/professionals/clinicians-providers/guidelinesrecommendations/tobacco/5 steps. pdf; date accessed: 16 Oct 2019 8. Kruger J, O'Halloran A, Rosenthal AC, Babb SD, Fiore MC. Receipt of evidence-based brief cessation interventions by health professionals and use of cessation assisted treatments among current adult cigarette-only smokers: National Adult Tobacco Survey, 2009 -2010. BMC Public Health. 2016; 16: 141.
- Slides: 22