To explain 1 HNMR is based on four

To explain 1 H-NMR is based on four informations: ☺Integration: ratio of H atoms ☺Chemical shift: chemical environment ☺Spin-spin coupling: information about neighboring nuclei ☺Coupling constant: isomers (cis or trans)

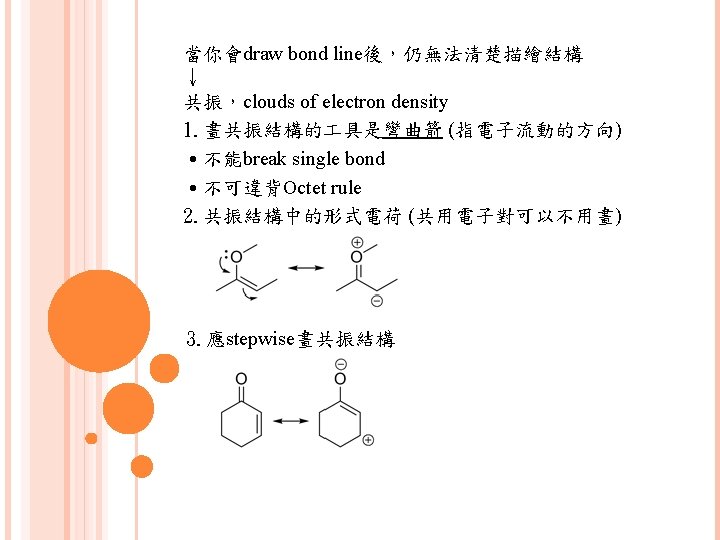
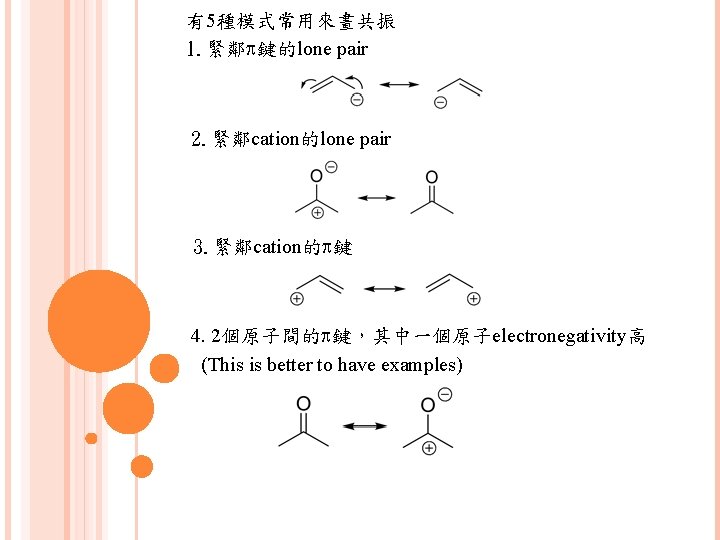
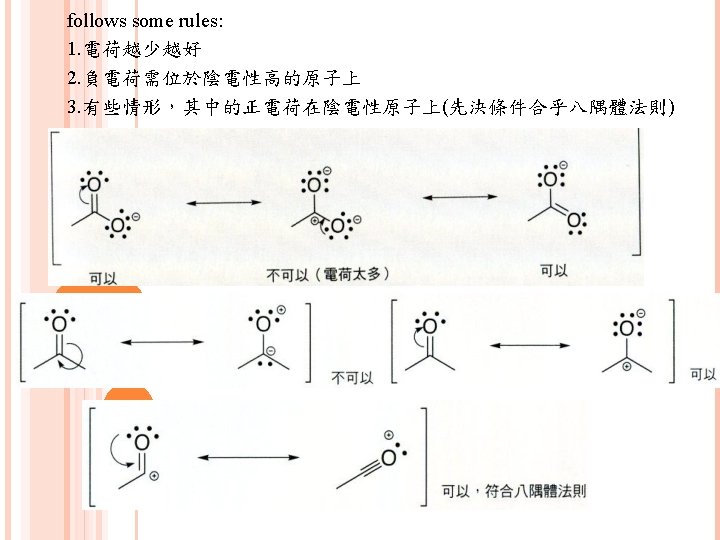
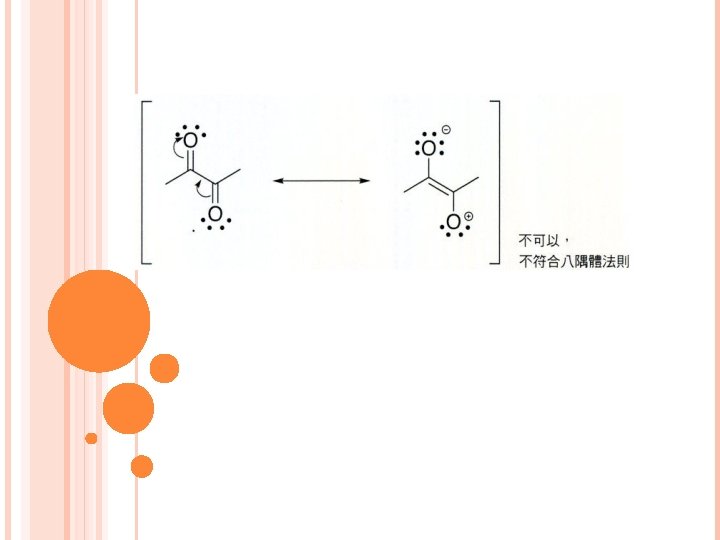
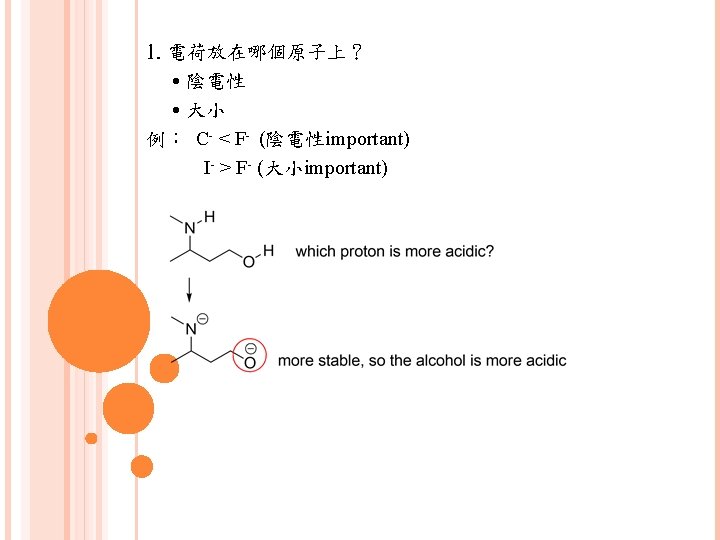
有5種模式常用來畫共振 1. 緊鄰p鍵的lone pair 2. 緊鄰cation的lone pair 3. 緊鄰cation的p鍵 4. 2個原子間的p鍵,其中一個原子electronegativity高 (This is better to have examples)
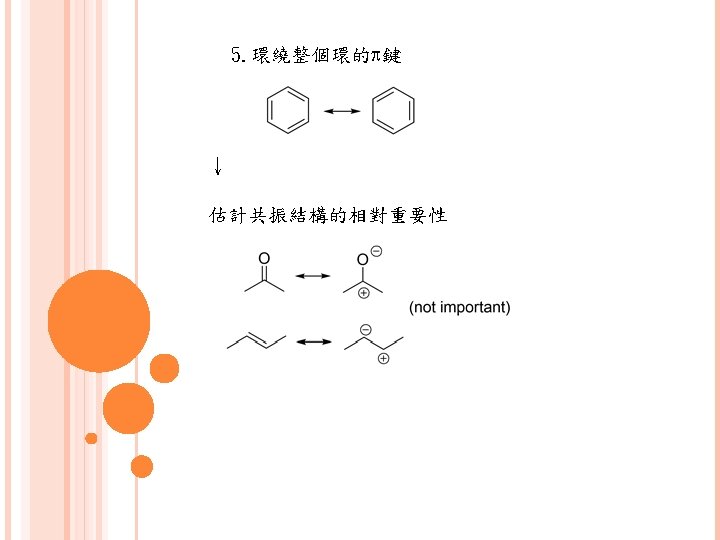

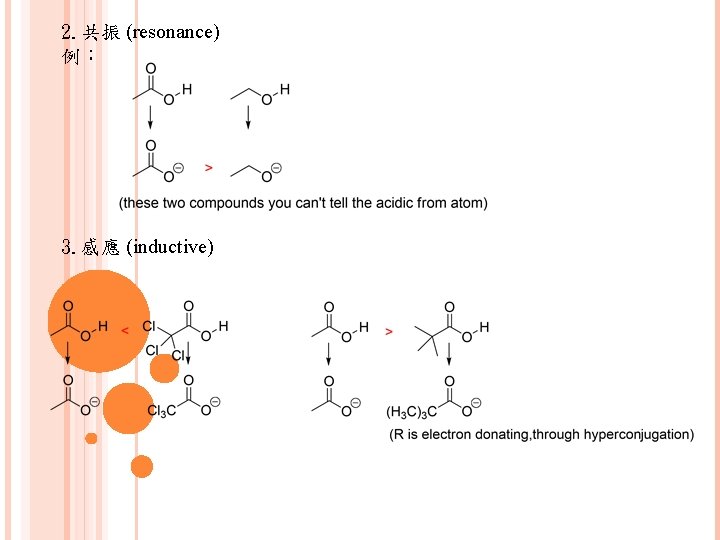
2. 共振 (resonance) 例: 3. 感應 (inductive)
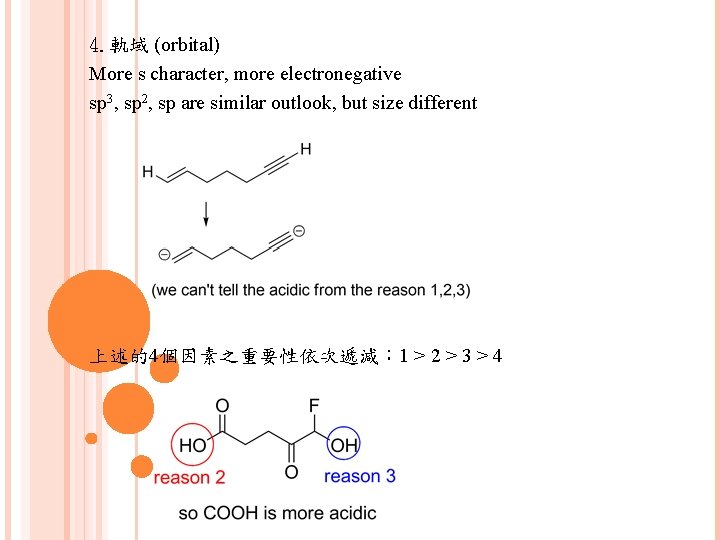
4. 軌域 (orbital) More s character, more electronegative sp 3, sp 2, sp are similar outlook, but size different 上述的4個因素之重要性依次遞減: 1 > 2 > 3 > 4


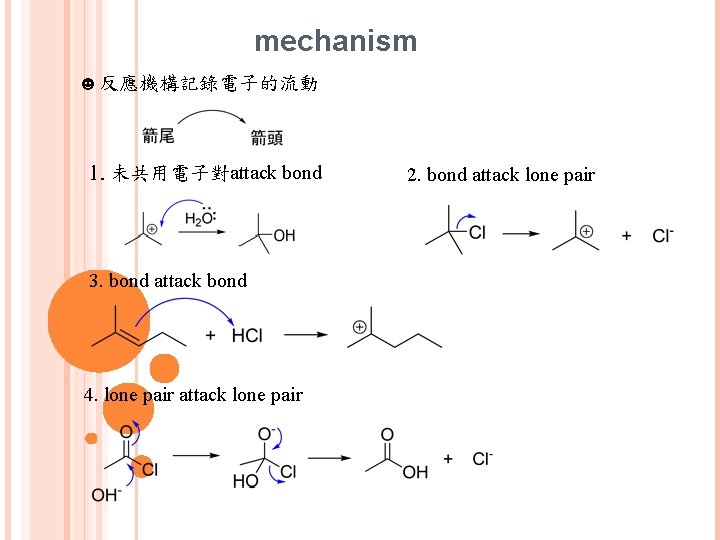
mechanism ☻反應機構記錄電子的流動 1. 未共用電子對attack bond 3. bond attack bond 4. lone pair attack lone pair 2. bond attack lone pair

☻ 鹼槓上了親核基 (basicity / nucleophilicity) basicity / nucleophilicity差別在功能上的不同 nucleophilicity: 反應速度 (快∕慢)(kinetic control) basicity: 指平衡的位置(thermodynamic control) ☻ 反應機構內包含了區域選擇性 (regiochemistry) Elimination: Zaitsev product; Hoffmann product Addition: Markovnikov product; anti-Markovnikov product ☻ 反應機構內包含了立體化學 (stereoselective) Syn-addition and anti-addition ☻ 反應機構清單

․Second-Row Nucleophiles in the SN 2 Reaction Basicity- measure of a compound shared its lone pair 可經由equilibrium constant (acid dissociation constant Ka)strong / weak Nucleophilicity - measure of a compound is able to attack an electron-deficient atom可經 由rate constant (K) 來得知 good / poor RULE 1 (Second-row) nucleophiles: • Are approximately the same size. • The stronger the base, the better the nucleophile. stronger base, better nucleophile OH– CH 3 O– H 2 N– CH 3 CH 2 NH– weaker base, poorer nucleophile > > H 2 O CH 3 OH NH 3 CH 3 CH 2 NH 2
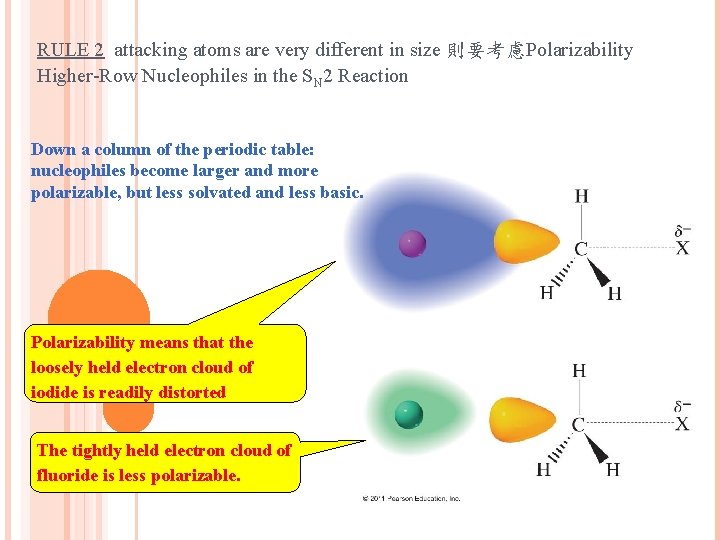
RULE 2 attacking atoms are very different in size 則要考慮Polarizability Higher-Row Nucleophiles in the SN 2 Reaction Down a column of the periodic table: nucleophiles become larger and more polarizable, but less solvated and less basic. Polarizability means that the loosely held electron cloud of iodide is readily distorted The tightly held electron cloud of fluoride is less polarizable.
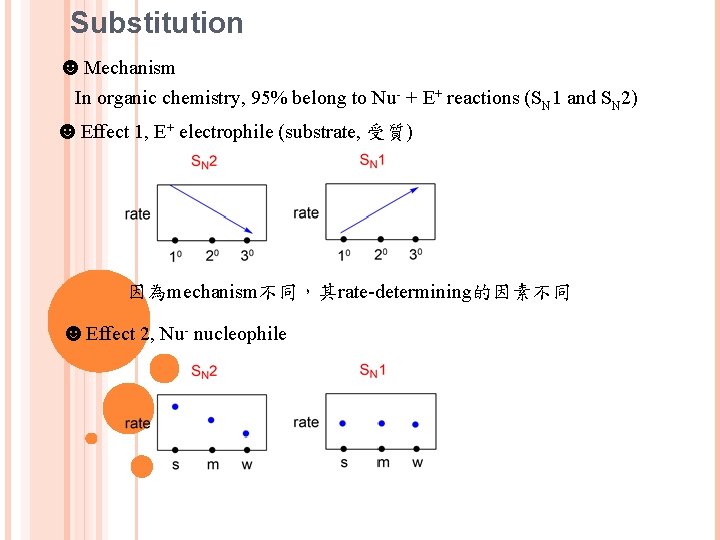
Substitution ☻Mechanism In organic chemistry, 95% belong to Nu- + E+ reactions (SN 1 and SN 2) ☻Effect 1, E+ electrophile (substrate, 受質) 因為mechanism不同,其rate-determining的因素不同 ☻Effect 2, Nu- nucleophile

☻Effect 3, Leaving group 若受質substrate 20 , 而Nu-溫和,則SN 1? SN 2? ․Extremely good leaving group: (中性) ․Good leaving group, stable anion: ․Bad leaving group: RO-, R 2 N-
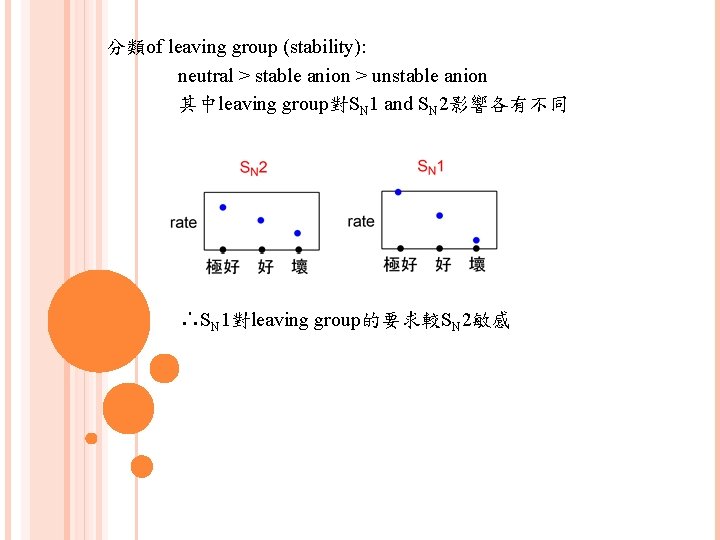
分類of leaving group (stability): neutral > stable anion > unstable anion 其中leaving group對SN 1 and SN 2影響各有不同 ∴SN 1對leaving group的要求較SN 2敏感
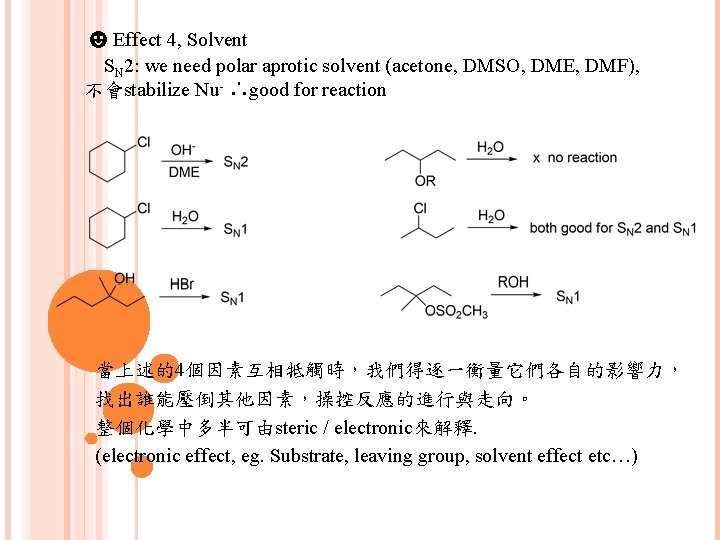
☻Effect 4, Solvent SN 2: we need polar aprotic solvent (acetone, DMSO, DME, DMF), 不會stabilize Nu- ∴good for reaction 當上述的4個因素互相牴觸時,我們得逐一衡量它們各自的影響力, 找出誰能壓倒其他因素,操控反應的進行與走向。 整個化學中多半可由steric / electronic來解釋. (electronic effect, eg. Substrate, leaving group, solvent effect etc…)
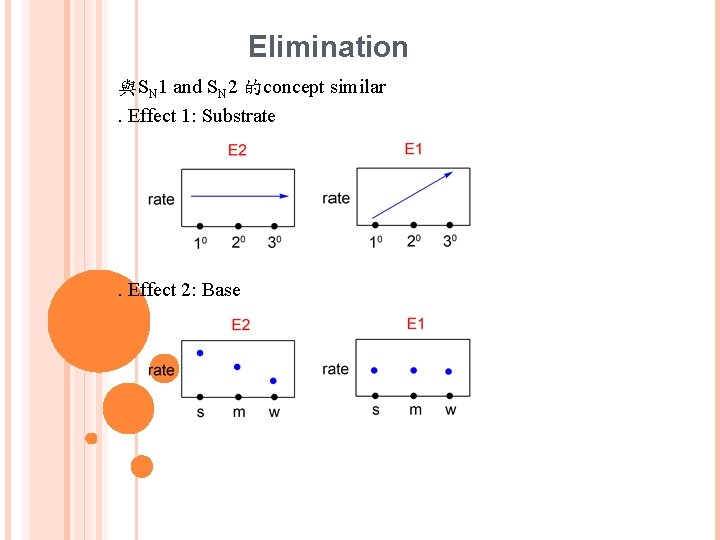
Elimination 與SN 1 and SN 2 的concept similar ․Effect 1: Substrate ․Effect 2: Base
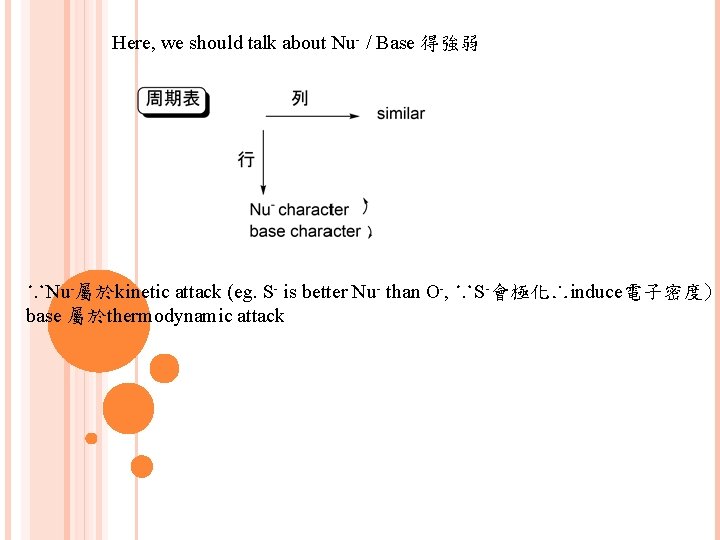
Here, we should talk about Nu- / Base 得強弱 ∵Nu-屬於kinetic attack (eg. S- is better Nu- than O-, ∵S-會極化∴induce電子密度) base 屬於thermodynamic attack

․Effect 3: Leaving group E 1 is more sensitive to the tendency of the leaving group ․Effect 4: Solvent effect polar aprotic solvent (acetone, DMSO, DME, DMF) ☻Regiochemistry, Stereochemistry For the regiochemistry, There are two different products, one is Zaitsev product, the other is Hoffmann product For stereochemistry, you need to know the elimination mechanism are syn-elimination or anti-elimination. You can use Newman project technique.

Addition reaction 加成反應要考慮Markovnikov addition or Anti-Markovnikov reaction ↓ regiochemistry, stereochemistry 先畫反應機構,要同時考慮regiochemistry and stereochemistry 這樣就會寫到right answer

- Slides: 29