Titrations Titration A known concentration of base or

Titrations

Titration A known concentration of base (or acid) is slowly added to a solution of acid (or base). Aqueous Equilibria

Titration A p. H meter or indicators are used to determine when the solution has reached the equivalence point, at which the stoichiometric amount of acid equals that of base. Aqueous Equilibria
Titration of a Strong Acid with a Strong Base Acid solution & indicator in the flask Aqueous Equilibria
Titration of a Strong Acid with a Strong Base solution in the buret Acid solution & indicator in the flask Aqueous Equilibria
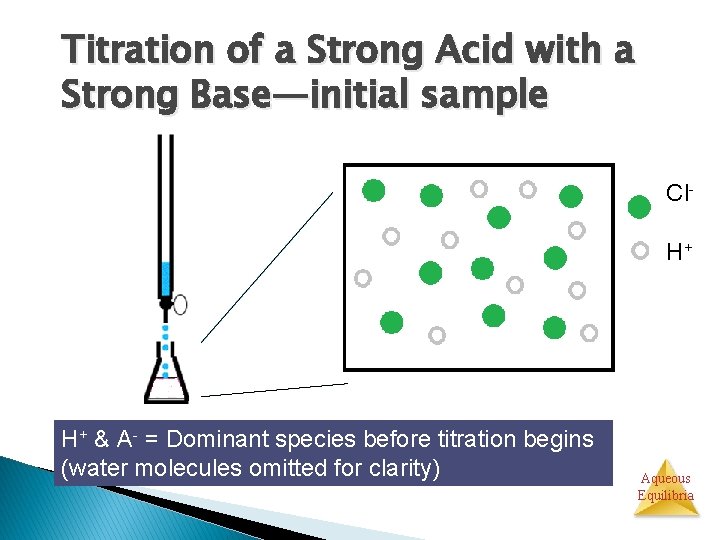
Titration of a Strong Acid with a Strong Base—initial sample Cl. H+ H+ & A- = Dominant species before titration begins (water molecules omitted for clarity) Aqueous Equilibria

Titration of a Strong Acid with a Strong Base From the start of the titration to near the equivalence point, the p. H goes up slowly. Aqueous Equilibria
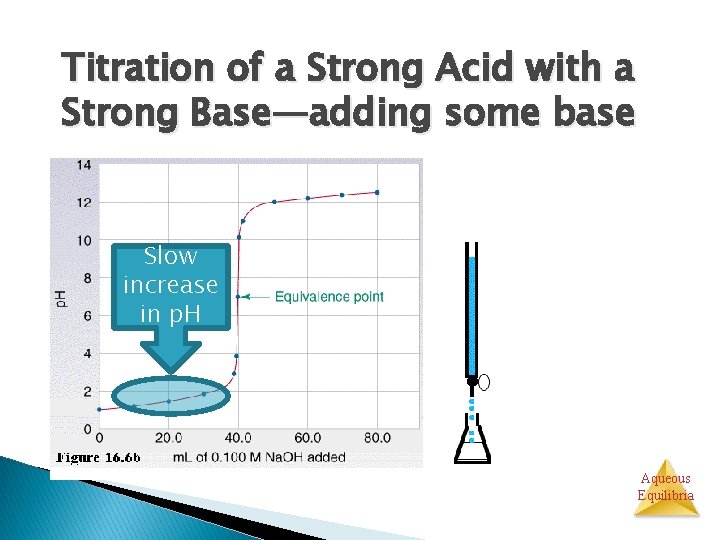
Titration of a Strong Acid with a Strong Base—adding some base Slow increase in p. H Aqueous Equilibria
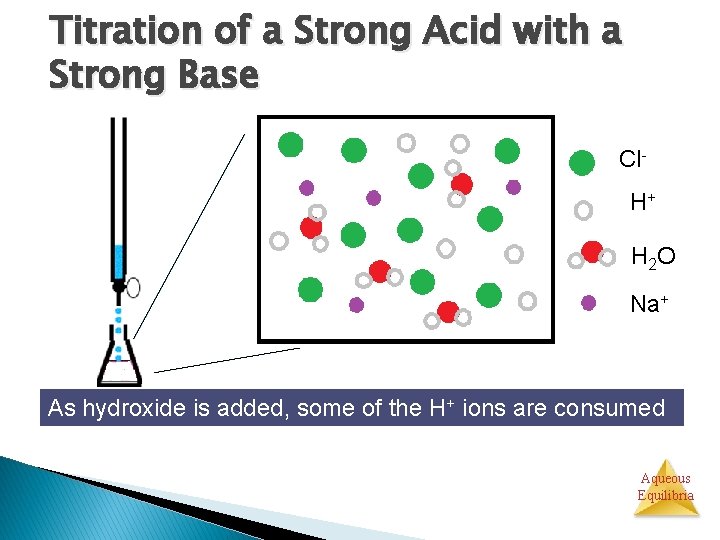
Titration of a Strong Acid with a Strong Base Cl. H+ H 2 O Na+ As hydroxide is added, some of the H+ ions are consumed Aqueous Equilibria

Titration of a Strong Acid with a Strong Base Just before and after the equivalence point, the p. H increases rapidly. Aqueous Equilibria

Titration of a Strong Acid with a Strong Base: Equivalence Point � moles acid initially present = moles base added � solution contains only water and the salt from the cation of the base and the anion of the acid. Aqueous Equilibria
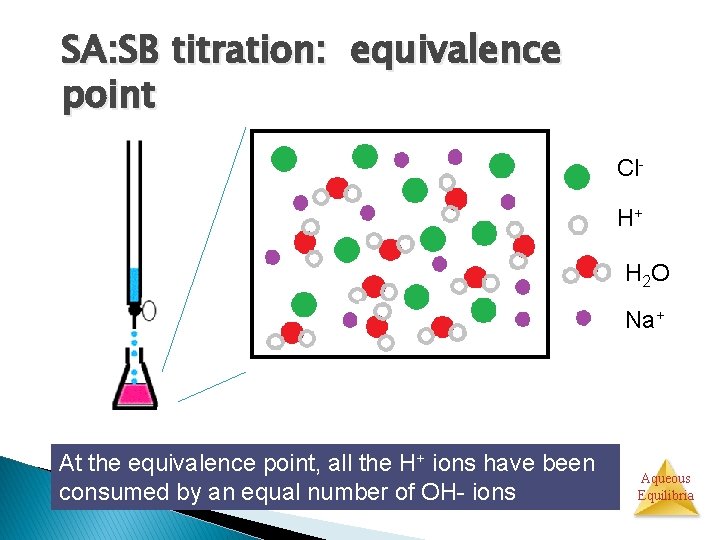
SA: SB titration: equivalence point Cl. H+ H 2 O Na+ At the equivalence point, all the H+ ions have been consumed by an equal number of OH- ions Aqueous Equilibria

SA: SB titration @ equivalence point Cl. H+ H 2 O Na+ Major species at equivalence point: water, sodium ions (cation from base), chloride ions (anion from acid) Aqueous Equilibria
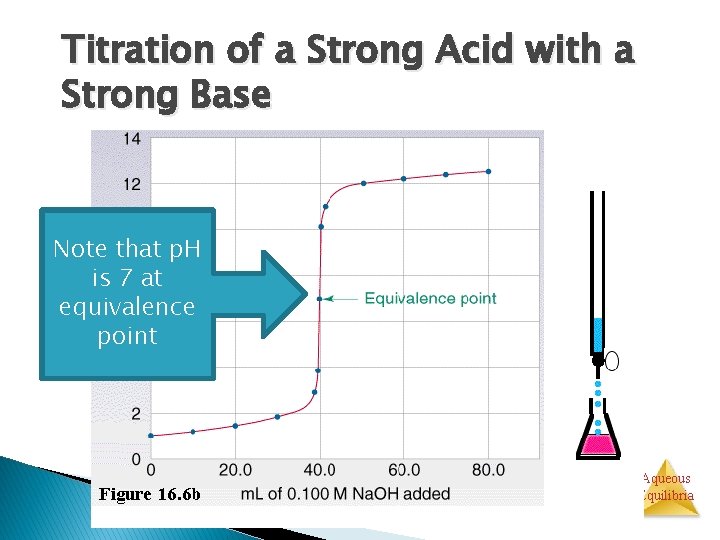
Titration of a Strong Acid with a Strong Base Cl. Note that p. H is 7 at equivalence point H+ H 2 O Na+ Aqueous Equilibria
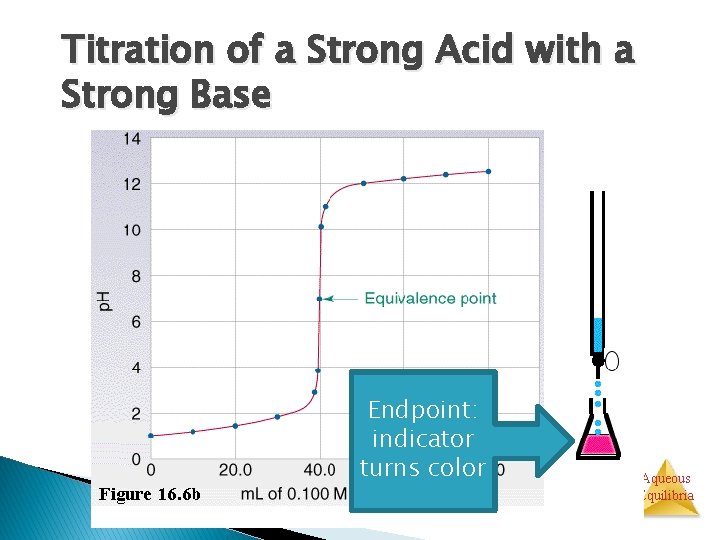
Titration of a Strong Acid with a Strong Base Cl. H+ H 2 O Na+ Endpoint: indicator turns color Aqueous Equilibria

Titration of a Strong Acid with a Strong Base As more base is added, the increase in p. H again levels off. Aqueous Equilibria

Titration of a Strong Acid with a Strong Base Excess hydroxide ions Aqueous Equilibria

SA: SB titration…beyond the Excess hydroxide ions (and equivalence point sodium ions, too), and leftover chloride ions Aqueous Water molecules omitted for clarity Equilibria

Titrating a weak acid with a strong base � Unlike in the previous case, the conjugate base of the acid affects the p. H when it is formed. Aqueous Equilibria
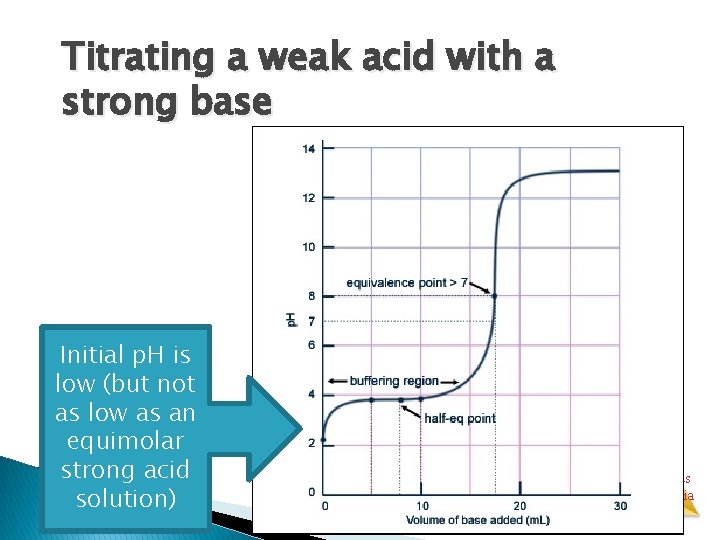
Titrating a weak acid with a strong base Initial p. H is low (but not as low as an equimolar strong acid solution) Aqueous Equilibria

Titrating a weak acid with a strong base—initial acid sample HA AH+ � Dominant species at beginning: HA molecules (5% dissociation not shown) � Water molecules and indicator omitted for clarity Aqueous Equilibria
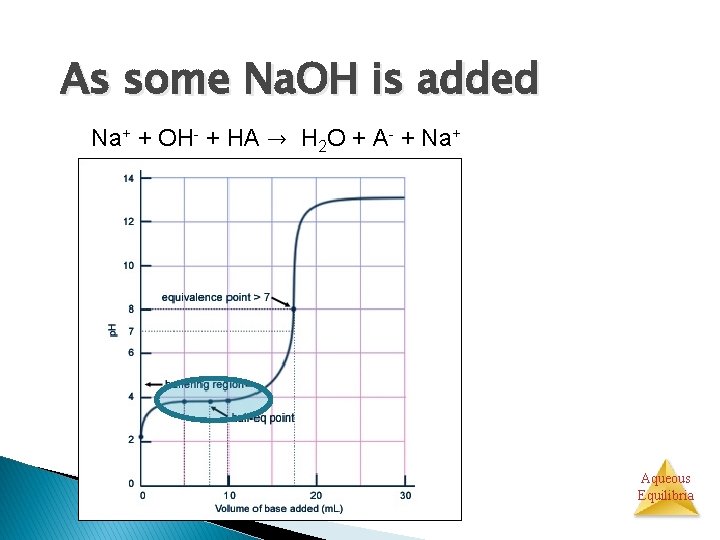
As some Na. OH is added Na+ + OH- + HA → H 2 O + A- + Na+ Aqueous Equilibria

As some Na. OH is added HA AH+ Na+ H 2 O � Both HA and A- are present � A buffer! ◦ HA: A- ratio is changing Aqueous Equilibria

As some Na. OH is added p. H increases slowly in the buffer region Aqueous Equilibria
![Halfway to the equivalence point [HA]=[A-] Aqueous Equilibria Halfway to the equivalence point [HA]=[A-] Aqueous Equilibria](http://slidetodoc.com/presentation_image_h2/0f668ff1df1c6687e37884d5c42ee14c/image-25.jpg)
Halfway to the equivalence point [HA]=[A-] Aqueous Equilibria

Halfway to the equivalence point p. H = p. Ka at the half-equivalence point Aqueous Equilibria

At the equivalence point Na+ + OH- + HA → H 2 O + A- + Na+ Initial moles of HA = moles of OH- added All of the moles of HA and OH- are consumed Aqueous Equilibria

At the equivalence point HA AH+ Na+ H 2 O Major species: A-, Also Na+ (cation from base), water Aqueous Equilibria

At the equivalence point p. H >7 at equivalence point Aqueous Equilibria
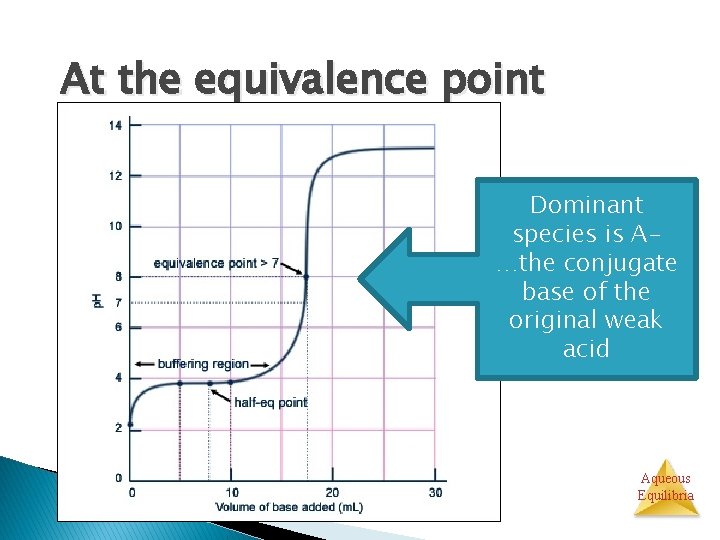
At the equivalence point Dominant species is A…the conjugate base of the original weak acid Aqueous Equilibria

Titration of a Weak Acid with a Strong Base With weaker acids, the initial p. H is higher and p. H changes near the equivalence point are more subtle. Aqueous Equilibria
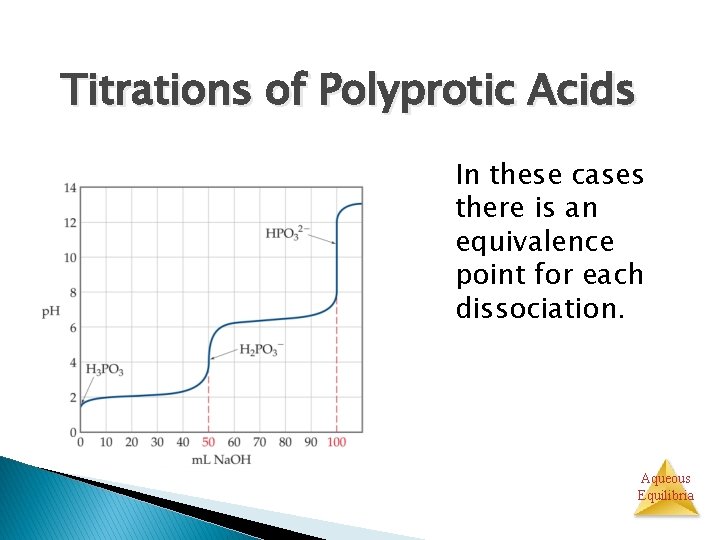
Titrations of Polyprotic Acids In these cases there is an equivalence point for each dissociation. Aqueous Equilibria
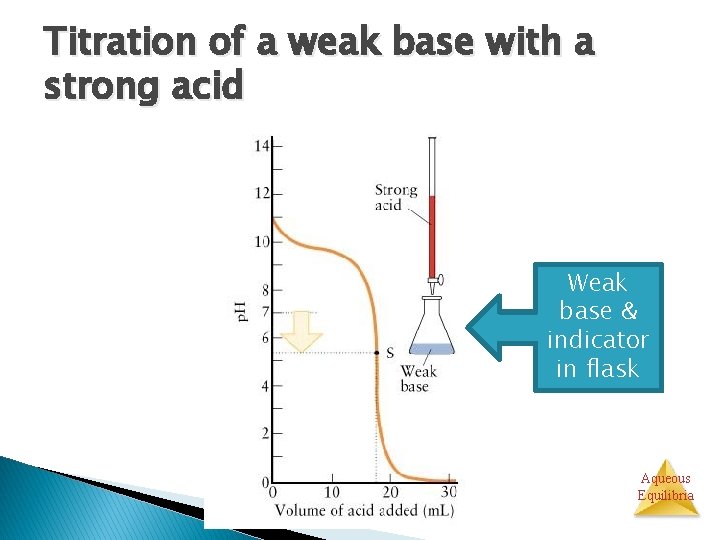
Titration of a weak base with a strong acid Weak base & indicator in flask Aqueous Equilibria
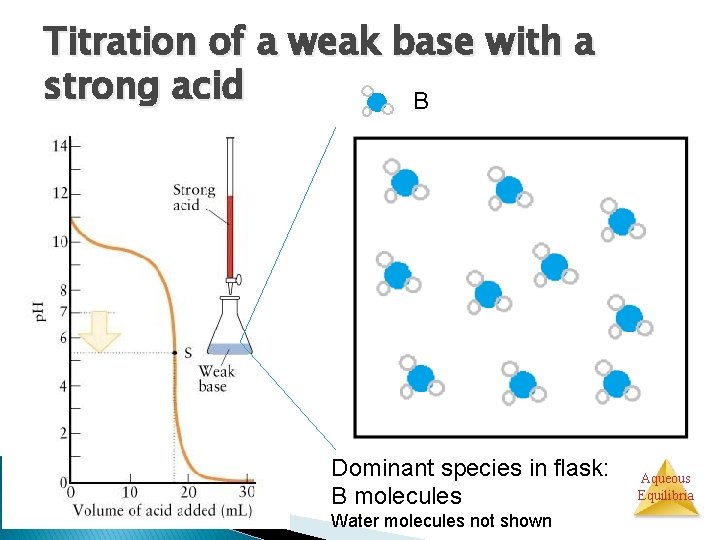
Titration of a weak base with a strong acid B Dominant species in flask: B molecules Water molecules not shown Aqueous Equilibria
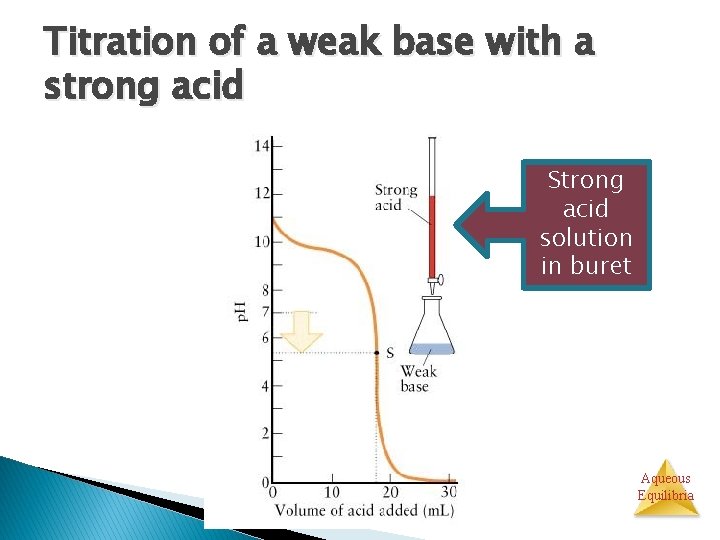
Titration of a weak base with a strong acid Strong acid solution in buret Aqueous Equilibria
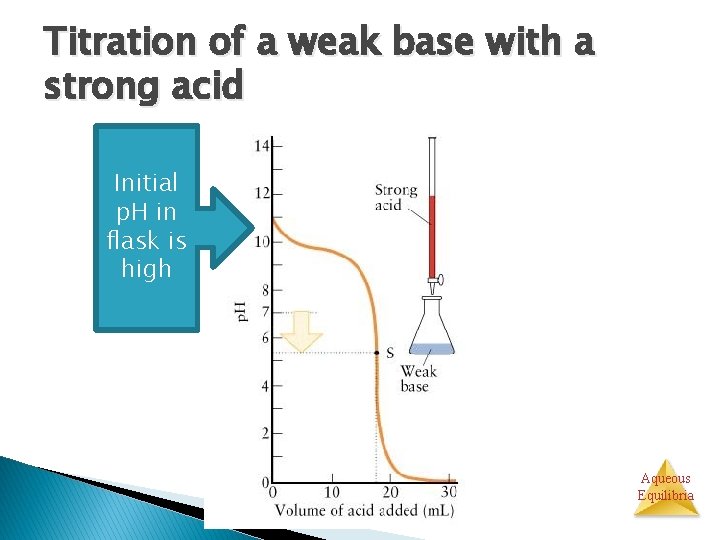
Titration of a weak base with a strong acid Initial p. H in flask is high Aqueous Equilibria
![Titration of a weak base with a strong acid—half titration point [B] = [HB+] Titration of a weak base with a strong acid—half titration point [B] = [HB+]](http://slidetodoc.com/presentation_image_h2/0f668ff1df1c6687e37884d5c42ee14c/image-37.jpg)
Titration of a weak base with a strong acid—half titration point [B] = [HB+] Aqueous Equilibria

Titration of a weak base with a strong acid At half equivalence point, p. OH = p. Kb p. H = p. Ka of HB+ Aqueous Equilibria

Titration of a weak base with a strong acid Dominant species: HB+ It will react with water… ICE problem to calculate p. H Aqueous Equilibria
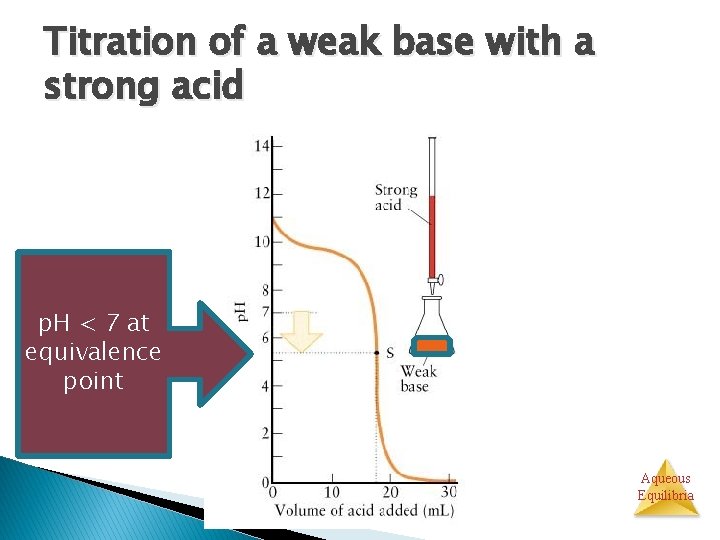
Titration of a weak base with a strong acid p. H < 7 at equivalence point Aqueous Equilibria
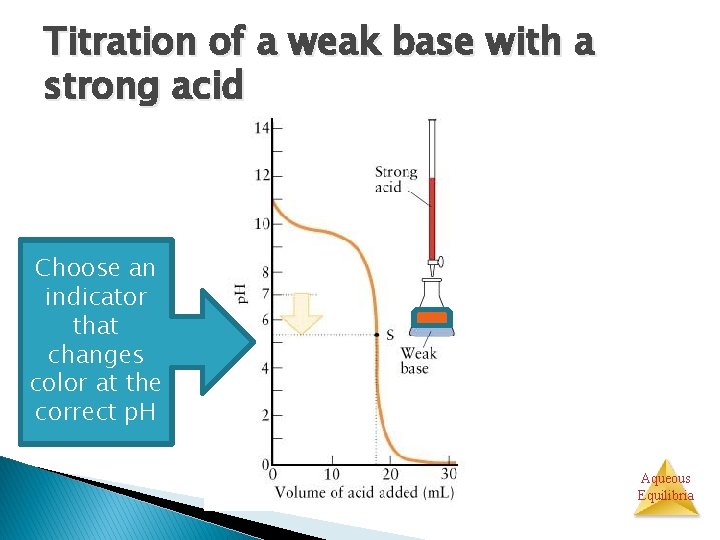
Titration of a weak base with a strong acid Choose an indicator that changes color at the correct p. H Aqueous Equilibria
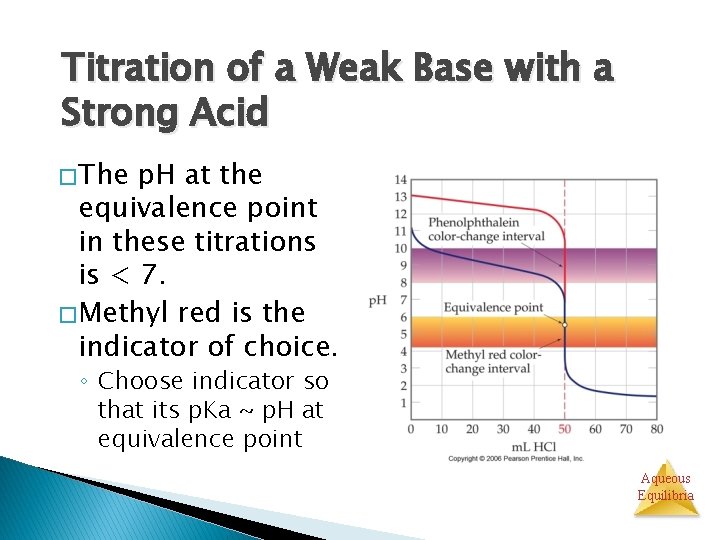
Titration of a Weak Base with a Strong Acid � The p. H at the equivalence point in these titrations is < 7. � Methyl red is the indicator of choice. ◦ Choose indicator so that its p. Ka ~ p. H at equivalence point Aqueous Equilibria
- Slides: 42