Titrations Strong Acid with Strong Base Starting p

Titrations
![Strong Acid with Strong Base • Starting p. H = -log[FAcid] • Just before Strong Acid with Strong Base • Starting p. H = -log[FAcid] • Just before](http://slidetodoc.com/presentation_image/3eaf37e94b47c27b86099f1a3628e9eb/image-2.jpg)
Strong Acid with Strong Base • Starting p. H = -log[FAcid] • Just before the Equivalence Point [H+] = (Vacid·Facid-Vbase·Fbase)/(Vsol+Vbase) • Very Sharp Equivalence Point p. H = 7. 00 • Excess base p. H = 14 – (-log[OH-]) [OH-] = Fbase·{(Vbase-V*)/(Vbase+Vsol)]}
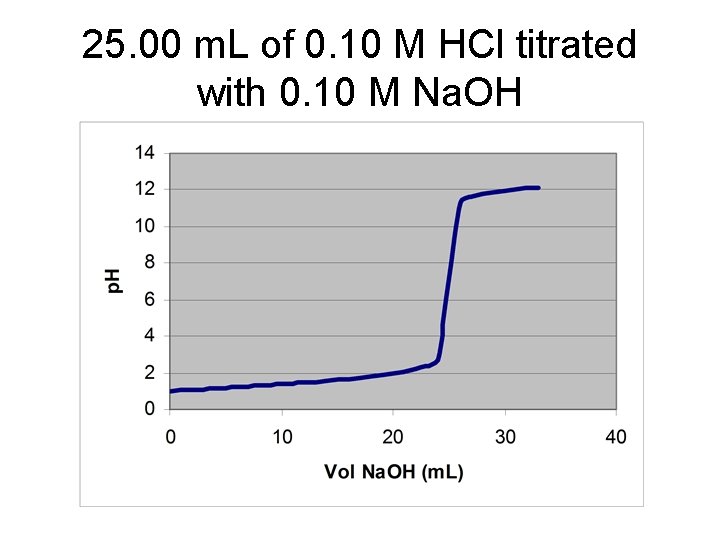
25. 00 m. L of 0. 10 M HCl titrated with 0. 10 M Na. OH
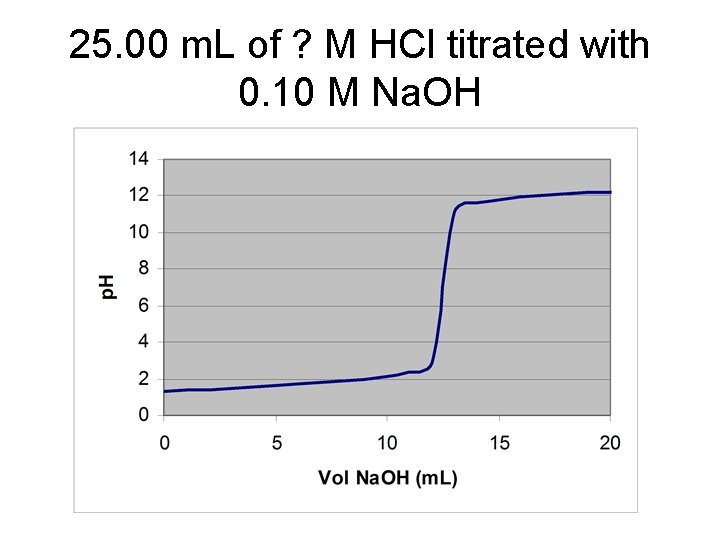
25. 00 m. L of ? M HCl titrated with 0. 10 M Na. OH
![V* = 12. 50 m. L of Na. OH [HCl] = (12. 50 m. V* = 12. 50 m. L of Na. OH [HCl] = (12. 50 m.](http://slidetodoc.com/presentation_image/3eaf37e94b47c27b86099f1a3628e9eb/image-5.jpg)
V* = 12. 50 m. L of Na. OH [HCl] = (12. 50 m. L)(0. 1000 M)/(25. 00 m. L) = 0. 0500 M
![Strong Base with Strong Acid • Starting p. H = 14 – {-log[Base] • Strong Base with Strong Acid • Starting p. H = 14 – {-log[Base] •](http://slidetodoc.com/presentation_image/3eaf37e94b47c27b86099f1a3628e9eb/image-6.jpg)
Strong Base with Strong Acid • Starting p. H = 14 – {-log[Base] • Very Sharp Equivalence Point p. H = 7. 00 • Excess acid p. H = -log[H+]) [H+] = Facid·{(Vacid-V*)/(Vacid+Vsol)]}
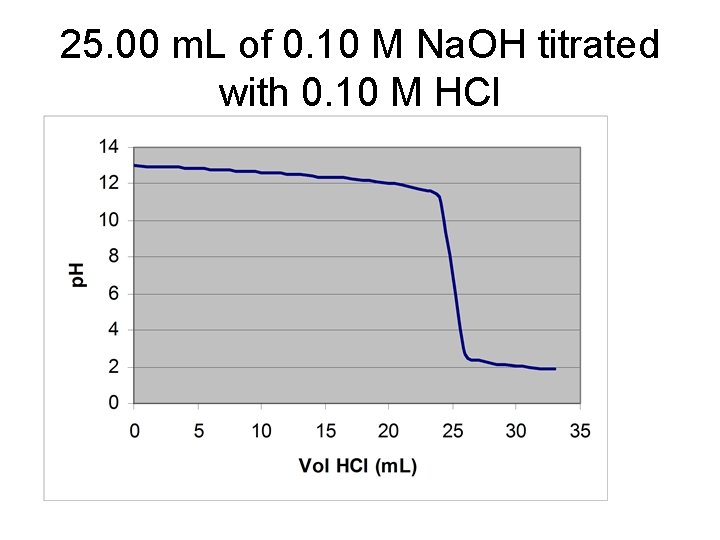
25. 00 m. L of 0. 10 M Na. OH titrated with 0. 10 M HCl
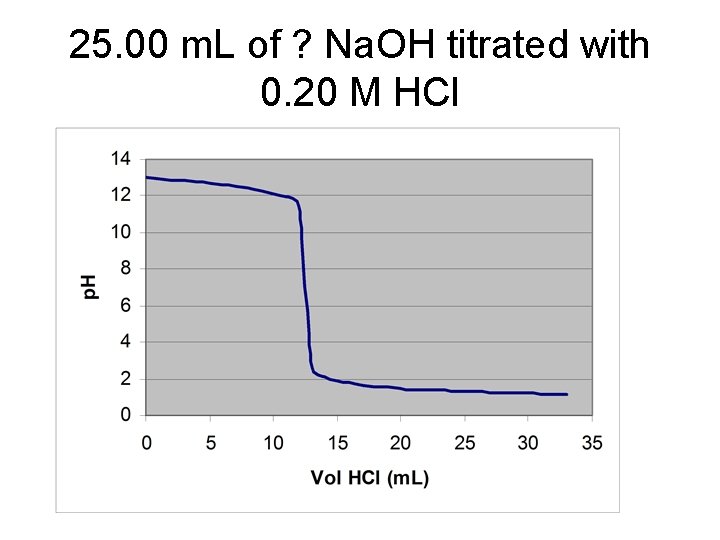
25. 00 m. L of ? Na. OH titrated with 0. 20 M HCl
![V* = 12. 50 m. L of HCl [Na. OH] = (12. 50 m. V* = 12. 50 m. L of HCl [Na. OH] = (12. 50 m.](http://slidetodoc.com/presentation_image/3eaf37e94b47c27b86099f1a3628e9eb/image-9.jpg)
V* = 12. 50 m. L of HCl [Na. OH] = (12. 50 m. L)(0. 2000 M)/(25. 00 m. L) = 0. 1000 M

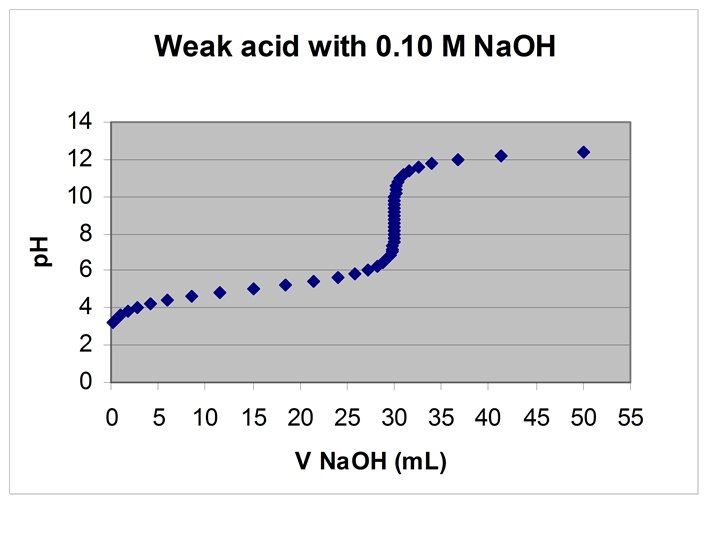
Titrating Monoprotic Weak Acids with Strong Base • Starting p. H: Use weak acid dissociation Ka = [H+]2/(FAcid – [H+]) p. H = -log[H+] HA + OH- → A- + H 2 O • Before the Equivalence Point use H-H eq p. H = p. Ka + log [(FNa. OH·VNa. OH)/(FHA·VHA- FNa. OH·VNa. OH)] ½ way to Equivalence point, p. H = p. Ka
![• At Equivalence point: use weak base dissociation Kb = [OH-]2/(Fbase – [OH-]), • At Equivalence point: use weak base dissociation Kb = [OH-]2/(Fbase – [OH-]),](http://slidetodoc.com/presentation_image/3eaf37e94b47c27b86099f1a3628e9eb/image-11.jpg)
• At Equivalence point: use weak base dissociation Kb = [OH-]2/(Fbase – [OH-]), where Fbase = FHA·VHA/(Vsol+Veq) • Excess base [OH-] = Fbase·{(Vbase-V*)/(Vbase+Vsol)]} p. H = 14 – (-log[OH-])
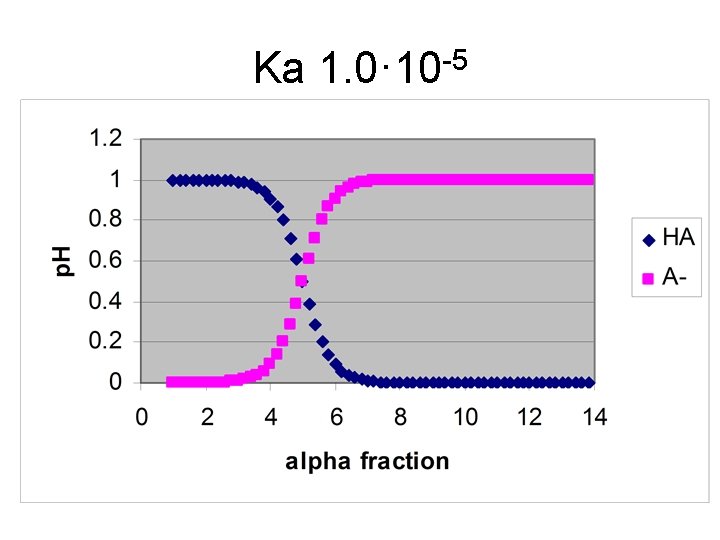
Ka 1. 0· 10 -5
![Example for Ka = 10 -5 • Starting p. H [H+] = ((1∙ 10 Example for Ka = 10 -5 • Starting p. H [H+] = ((1∙ 10](http://slidetodoc.com/presentation_image/3eaf37e94b47c27b86099f1a3628e9eb/image-14.jpg)
Example for Ka = 10 -5 • Starting p. H [H+] = ((1∙ 10 -5∙ 0. 1)1/2 = 0. 0010 M p. H = 3. 00 • ½ way – p. H = p. Ka = 5. 00 • At Equiv. pt [OH-] = ((1∙ 10 -9∙ 0. 05)1/2 = 7. 1· 10 -6 M p. H = 14. 00 – [-log(7. 1· 10 -6)] = 8. 85

Titrating Monoprotic Weak Bases with Strong Acid • Starting p. H: Use weak base dissociation Kb = [OH-]2/(Fbase – [OH-]) p. H = 14. 00 – (-log[OH-]) A- + H+ → HA • Before the Equivalence Point use H-H eq p. H = p. Ka + log [(FA-·VA-- FHCl·VHCl)/ (FHCl·VHCl)] ½ way to Equivalence point, p. H = p. Ka
![• At Equivalence point: use weak acid dissociation Kb = [H+]2/(Facid – [H+]), • At Equivalence point: use weak acid dissociation Kb = [H+]2/(Facid – [H+]),](http://slidetodoc.com/presentation_image/3eaf37e94b47c27b86099f1a3628e9eb/image-16.jpg)
• At Equivalence point: use weak acid dissociation Kb = [H+]2/(Facid – [H+]), where FACID = FA-·VA-/(Vsol+Veq) • Excess HCl [H+] = Facid·{(Vacid-V*)/(Vacid+Vsol)]} p. H = log[H+])
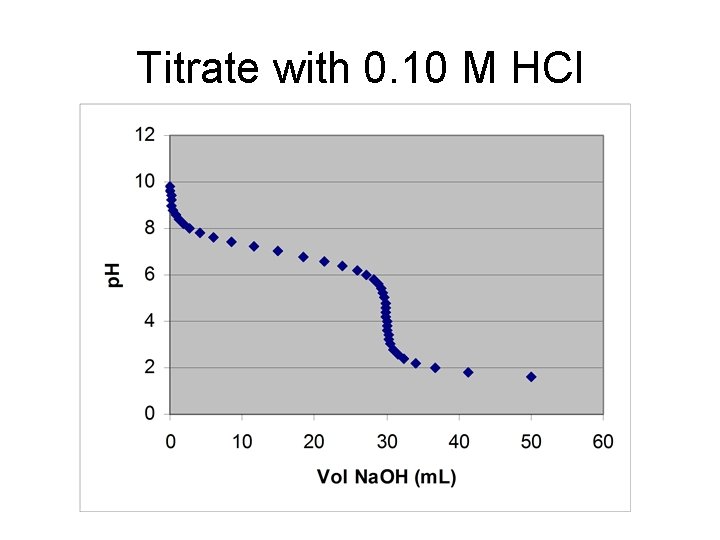
Titrate with 0. 10 M HCl
![Example for Ka = 10 -7 • Starting p. H [OH-] = ((1∙ 10 Example for Ka = 10 -7 • Starting p. H [OH-] = ((1∙ 10](http://slidetodoc.com/presentation_image/3eaf37e94b47c27b86099f1a3628e9eb/image-18.jpg)
Example for Ka = 10 -7 • Starting p. H [OH-] = ((1∙ 10 -7∙ 0. 1)1/2 = 0. 00010 M p. H = 10. 00 • ½ way – p. H = p. Ka = 7. 00 • At Equiv. pt [H-] = ((1∙ 10 -7∙ 0. 05)1/2 = 7. 1· 10 -5 M p. H = 4. 15

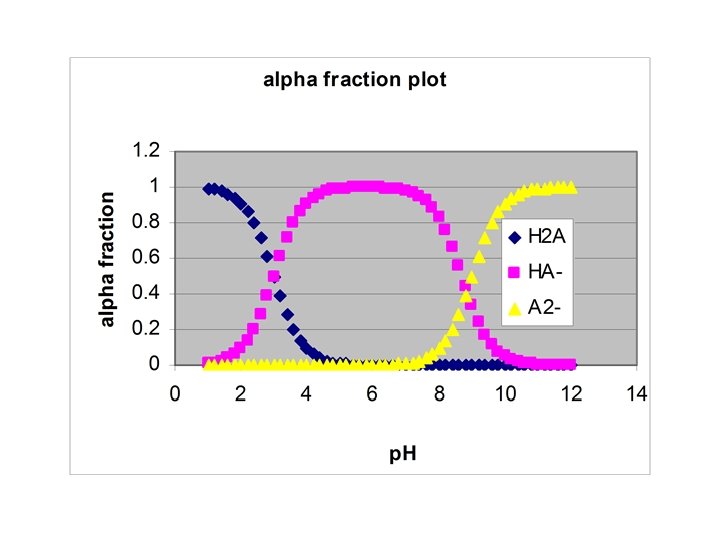
Titration of H 2 A with strong base • Starting p. H – weak acid equilibrium • ½ way to first eq. : p. H = p. Ka 1 • At first equivalence pt: p. H = (p. Ka 1 + p. Ka 2)/2 • ½ way to 2 nd eq. : p. H = p. Ka 2 • At 2 nd equiv. pt. : weak base equilibrium
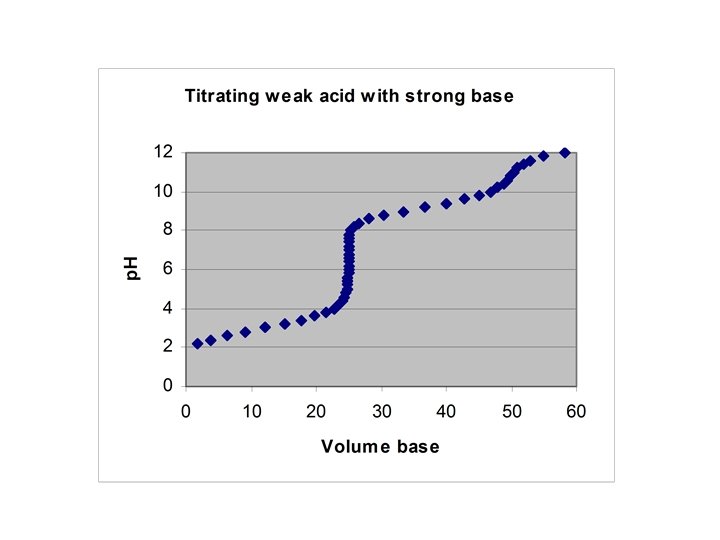
![Example • Starting p. H – [H+] = (10 -3*0. 1)1/2 = 0. 0100 Example • Starting p. H – [H+] = (10 -3*0. 1)1/2 = 0. 0100](http://slidetodoc.com/presentation_image/3eaf37e94b47c27b86099f1a3628e9eb/image-22.jpg)
Example • Starting p. H – [H+] = (10 -3*0. 1)1/2 = 0. 0100 M • ½ way to first eq. : p. H = 3. 00 • At first equivalence pt: p. H = (p. Ka 1 + p. Ka 2)/2 = 6. 00 • ½ way to 2 nd eq. : p. H = 9. 00 • At 2 nd equiv. pt. : [OH-] = (10 -5*0. 05)1/2 = 7. 07∙ 10 -4 M, p. H = 10. 85

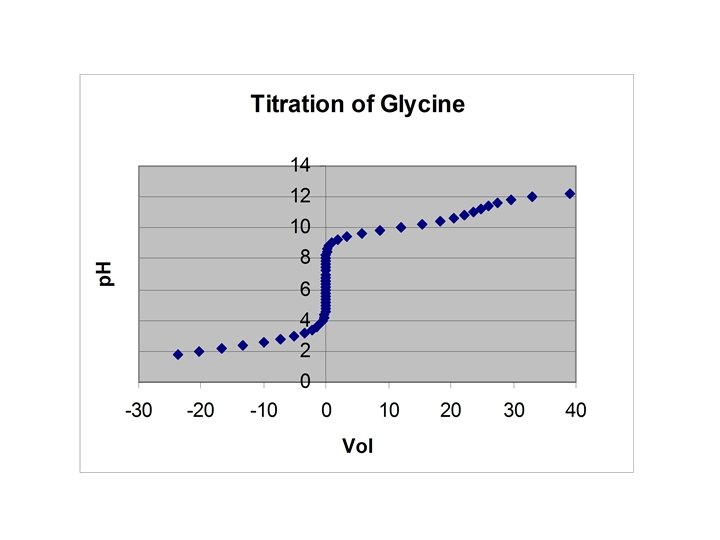
Amino acids • Amphoteric – Can act as an acid and a base • Glycine (GH 2+, GH, G-) p. Ka 1 = 2. 350, p. Ka 2 = 9. 778
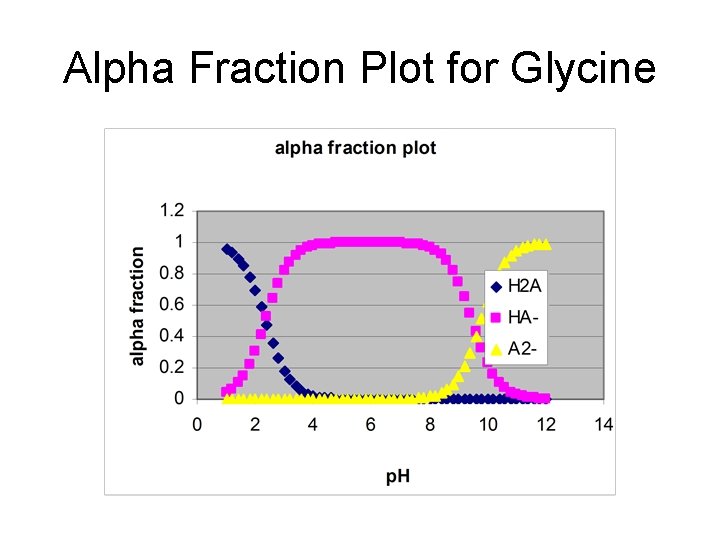
Alpha Fraction Plot for Glycine
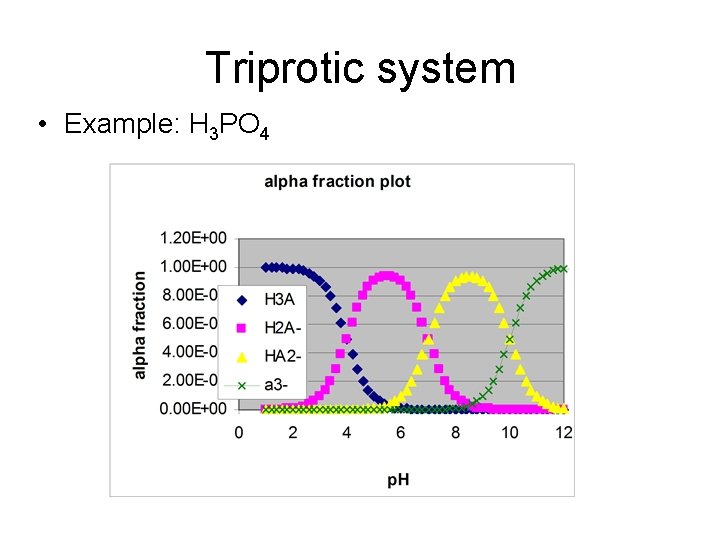
Triprotic system • Example: H 3 PO 4

Titration of a triprotic acid with strong base • Starting p. H – weak acid equilibrium • ½ way to first eq. : p. H = p. Ka 1 • At first equivalence pt: p. H = (p. Ka 1 + p. Ka 2)/2 • ½ way to 2 nd eq. : p. H = p. Ka 2

• At 2 nd equiv. pt. : p. H = (p. Ka 2 + p. Ka 3)/2 • ½ way to third equiv. pt. : p. H = p. Ka 3 • At third equiv pt: weak base equilibrium
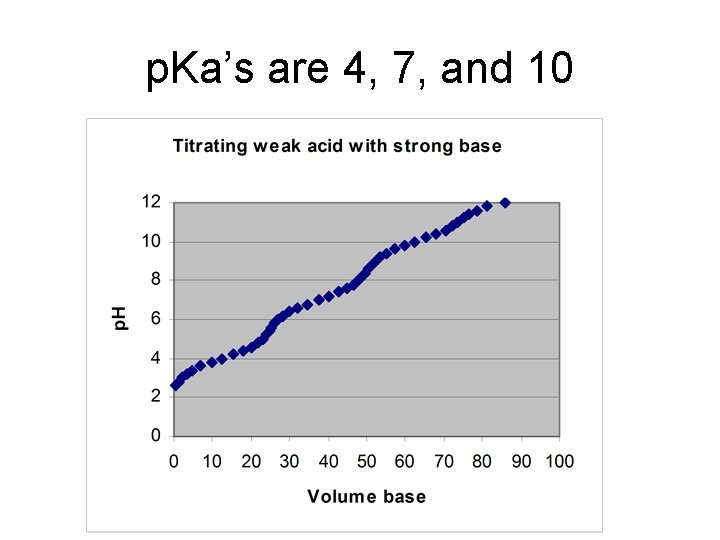
p. Ka’s are 4, 7, and 10
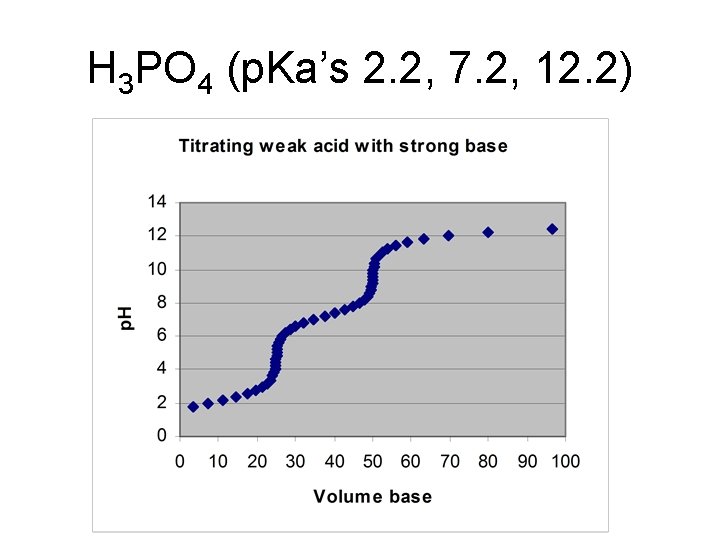
H 3 PO 4 (p. Ka’s 2. 2, 7. 2, 12. 2)
- Slides: 30